Abstract
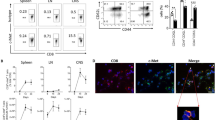
Clinical or laboratory evidence of varicella-zoster virus (VZV) infection has been consistently associated with lower glioma risk in case–control studies, suggesting a protective effect of VZV against glioma. We formulated the following explanatory hypotheses: reactivated VZV preferentially infects and kills gliomas compared to normal parenchyma; and VZV-specific cytotoxic T lymphocytes (CTL) cross-react with gliomas. We established an ex vivo model of VZV infection, which showed that glioma cell lines and primary astrocytes were equally permissive to VZV infection and had similar 15% average decrease in viability upon infection. In co-cultures, the relative growth of glioma cells and astrocytes was not affected by the VZV infection. However, VZV stimulated, but not mock stimulated, peripheral blood mononuclear cells from VZV-seropositive individuals recognized and killed HLA class I-matched glioma cells (mean±SE decrease in viability of 26 ± 12%, p = 0.04), but not matched astrocytes. VZV infection of the glioma cells did not affect the T cell-mediated killing. Taken together, these data suggest that ex vivo VZV infection has similar direct effects on glioma cells and astrocytes. The protective effect of prior VZV infection against the incidence of glioma may be mediated by CTL that recognizes epitopes shared by VZV and glioma cells.




Similar content being viewed by others
References
Ansari A, Li S, Abzug MJ, Weinberg A (2004) Human herpesviruses 6 and 7 and central nervous system infection in children. Emerg Infect Dis 10:1450–1454
Cobbs CS, Harkins L, Samanta M, Gillespie GY, Bharara S, King PH, Nabors LB, Cobbs CG, Britt WJ (2002) Human cytomegalovirus infection and expression in human malignant glioma. Cancer Res 62:3347–3350
Donson AM, Weil MD, Foreman NK (1999) Tamoxifen radiosensitization in human glioblastoma cell lines. J Neurosurg 90:533–536
Fodor PA, Levin MJ, Weinberg A, Sandberg E, Sylman J, Tyler KL (1998) Atypical herpes simplex virus encephalitis diagnosed by PCR amplification of viral DNA from CSF. Neurology 51:554–559
Gilden DH, Kleinschmidt-DeMasters BK, LaGuardia JJ, Mahalingam R, Cohrs RJ (2000) Neurologic complications of the reactivation of varicella-zoster virus. N Engl J Med 342:635–645
Haanpaa M, Dastidar P, Weinberg A, Levin M, Miettinen A, Lapinlampi A, Laippala P, Nurmikko T (1998) CSF and MRI findings in patients with acute herpes zoster. Neurology 51:1405–1411
Ji Q, Perchellet A, Goverman JM (2010) Viral infection triggers central nervous system autoimmunity via activation of CD8+ T cells expressing dual TCRs. Nat Immunol 11:628–634
Levin MJ, Oxman MN, Zhang JH, Johnson GR, Stanley H, Hayward AR, Caulfield MJ, Irwin MR, Smith JG, Clair J, Chan IS, Williams H, Harbecke R, Marchese R, Straus SE, Gershon A, Weinberg A (2008) Varicella-zoster virus-specific immune responses in elderly recipients of a herpes zoster vaccine. J Infect Dis 197:825–835
Loparev VN, Rubtcova E, Seward JF, Levin MJ, Schmid DS (2007) DNA sequence variability in isolates recovered from patients with postvaccination rash or herpes zoster caused by Oka varicella vaccine. J Infect Dis 195:502–510
Mehta SK, Cohrs RJ, Forghani B, Zerbe G, Gilden DH, Pierson DL (2004) Stress-induced subclinical reactivation of varicella zoster virus in astronauts. J Med Virol 72:174–179
Mitchell DA, Xie W, Schmittling R, Learn C, Friedman A, McLendon RE, Sampson JH (2008) Sensitive detection of human cytomegalovirus in tumors and peripheral blood of patients diagnosed with glioblastoma. Neuro Oncol 10:10–18
Patterson-Bartlett J, Levin MJ, Lang N, Schodel FP, Vessey R, Weinberg A (2007) Phenotypic and functional characterization of ex vivo T cell responses to the live attenuated herpes zoster vaccine. Vaccine 25:7087–7093
Quinlivan ML, Ayres K, Ran H, McElwaine S, Leedham-Green M, Scott FT, Johnson RW, Breuer J (2007) Effect of viral load on the outcome of herpes zoster. J Clin Microbiol 45:3909–3914
Tang YW, Espy MJ, Persing DH, Smith TF (1997) Molecular evidence and clinical significance of herpesvirus coinfection in the central nervous system. J Clin Microbiol 35:2869–2872
Wakim LM, Woodward-Davis A, Bevan MJ (2010) Memory T cells persisting within the brain after local infection show functional adaptations to their tissue of residence. Proc Natl Acad Sci USA 107:17872–17879
Watson B, Boardman C, Laufer D, Piercy S, Tustin N, Olaleye D, Cnaan A, Starr SE (1995) Humoral and cell-mediated immune responses in healthy children after one or two doses of varicella vaccine. Clin Infect Dis 20:316–319
Weinberg A, Clark JC, Schneider SA, Forghani B, Levin MJ (1996) Improved detection of varicella zoster infection with a spin amplification shell vial technique and blind passage. Clin Diagn Virol 5:61–65
Weinberg A, Betensky RA, Zhang L, Ray G (1998) Effect of shipment, storage, anticoagulant, and cell separation on lymphocyte proliferation assays for human immunodeficiency virus-infected patients. Clin Diagn Lab Immunol 5:804–807
Weinberg A, Li S, Palmer M, Tyler KL (2002) Quantitative CSF PCR in Epstein-Barr virus infections of the central nervous system. Ann Neurol 52:543–548
Weinberg A, Bloch KC, Li S, Tang YW, Palmer M, Tyler KL (2005) Dual infections of the central nervous system with Epstein-Barr virus. J Infect Dis 191:234–237
Wen PY, Kesari S (2008) Malignant gliomas in adults. N Engl J Med 359:492–507
Wilson A, Sharp M, Koropchak CM, Ting SF, Arvin AM (1992) Subclinical varicella-zoster virus viremia, herpes zoster, and T lymphocyte immunity to varicella-zoster viral antigens after bone marrow transplantation. J Infect Dis 165:119–126
Wollmann G, Tattersall P, van den Pol AN (2005) Targeting human glioblastoma cells: comparison of nine viruses with oncolytic potential. J Virol 79:6005–6022
Wollmann G, Robek MD, van den Pol AN (2007) Variable deficiencies in the interferon response enhance susceptibility to vesicular stomatitis virus oncolytic actions in glioblastoma cells but not in normal human glial cells. J Virol 81:1479–1491
Wrensch M, Lee M, Miike R, Newman B, Barger G, Davis R, Wiencke J, Neuhaus J (1997a) Familial and personal medical history of cancer and nervous system conditions among adults with glioma and controls. Am J Epidemiol 145:581–593
Wrensch M, Weinberg A, Miike R, Weincke J, Masters H, Lee M (1997b) Does prior infection with varicella zoster virus influence brain cancer risk? Amer J Epidemiol 145:594–597
Wrensch M, Weinberg A, Wiencke J, Miike R, Barger G, Kelsey K (2001) Prevalence of antibodies to four herpesviruses among adults with glioma and controls. Am J Epidemiol 154:161–165
Wrensch M, Weinberg A, Wiencke J, Miike R, Sison J, Wiemels J, Barger G, DeLorenze G, Aldape K, Kelsey K (2005) History of chickenpox and shingles and prevalence of antibodies to varicella-zoster virus and three other herpesviruses among adults with glioma and controls. Am J Epidemiol 161:929–938
Acknowledgments
The authors thank Karen Helm for technical support and Drs. Ken Tyler, Margaret Wrensch, Joe Wiemels, John Wincke, and Myron Levin for critical review of the manuscript. This work was supported by P30 CA046934 grant to the University of Colorado Cancer Center and by a subcontract to R01CA52689 (PI Margaret Wrensch). The contents are the sole responsibility of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Canniff, J., Donson, A.M., Foreman, N.K. et al. Cytotoxicity of glioblastoma cells mediated ex vivo by varicella-zoster virus-specific T cells. J. Neurovirol. 17, 448–454 (2011). https://doi.org/10.1007/s13365-011-0048-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13365-011-0048-z