Abstract
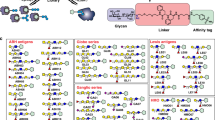
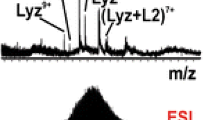
A semiquantitative electrospray ionization mass spectrometry (ESI-MS) binding assay suitable for analyzing mixtures of oligosaccharides, at unknown concentrations, for interactions with target proteins is described. The assay relies on the differences in the ratio of the relative abundances of the ligand-bound and free protein ions measured by ESI-MS at two or more initial protein concentrations to distinguish low affinity (≤103 M–1) ligands from moderate and high affinity (>105 M–1) ligands present in the library and to rank their affinities. Control experiments were performed on solutions of a single chain antibody and a mixture of synthetic oligosaccharides, with known affinities, in the absence and presence of a 40-component carbohydrate library to demonstrate the implementation and reliability of the assay. The application of the assay for screening natural libraries of carbohydrates against proteins is also demonstrated using mixtures of human milk oligosaccharides, isolated from breast milk, and fragments of a bacterial toxin and human galectin 3.

ᅟ






Similar content being viewed by others
References
Arnaud, J., Audfray, A., Imberty, A.: Binding sugars: from natural lectins to synthetic receptors and engineered neolectins. Chem. Soc. Rev. 42, 4798–4813 (2013)
Moran, A.P., Gupta, A., Joshi, L.: Sweet-talk: role of host glycosylation in bacterial pathogenesis of the gastrointestinal tract. Gut 60, 1412–1425 (2011)
Rillahan, C.D., Paulson, J.C.: Glycan microarrays for decoding the glycome. Annu. Rev. Biochem. 80, 797–823 (2011)
Yu, Y., Mishra, S., Song, X.Z., Lasanajak, Y., Bradley, K.C., Tappert, M.M., Air, G.M., Steinhauer, D.A., Halder, S., Cotmore, S., Tattersall, P., Agbandje-McKenna, M., Cummings, R.D., Smith, D.F.: Functional glycomic analysis of human milk glycans reveals the presence of virus receptors and embryonic stem cell biomarkers. J. Biol. Chem. 287, 44784–44799 (2012)
Recognition of carbohydrates in biological systems. Part A: General procedures. In: Lee, Y.C., Lee, R.T. (Eds.) Methods of enzymology, Vol 362, pp 1-625, Academic Press: Amsterdam (2003)
He, X.G., Gerona-Navarro, G., Jaffrey, S.R.: Ligand discovery using small-molecule microarrays. J. Pharmacol. Exp. Ther. 313, 1–7 (2005)
Grant, O.C., Smith, H.M., Firsova, D., Fadda, E., Woods, R.J.: Presentation, presentation, presentation! Molecular-level insight into linker effects on glycan array screening data. Glycobiology 24, 17–25 (2014)
Kitova, E.N., El-Hawiet, A., Schnier, P.D., Klassen, J.S.: Reliable determinations of protein-ligand interactions by direct ESI-MS measurements. Are we there yet? J. Am. Soc. Mass Spectrom. 23, 431–441 (2012)
Wang, W., Kitova, E.N., Klassen, J.S.: Influence of solution and gas phase processes on protein-carbohydrate binding affinities determined by nanoelectrospray Fourier transform ion cyclotron resonance mass spectrometry. Anal. Chem. 75, 4945–4955 (2003)
Shoemaker, G.K., Soya, N., Palcic, M.M., Klassen, J.S.: Temperature-dependent cooperativity in donor-acceptor substrate binding to the human blood group glycosyltransferases. Glycobiology 18, 587–592 (2008)
Soya, N., Shoemaker, G.K., Palcic, M.M., Klassen, J.S.: Comparative study of substrate and product binding to the human ABO(H) blood group glycosyltransferases. Glycobiology 19, 1224–1234 (2009)
Rademacher, C., Shoemaker, G.K., Kim, H.-S., Zheng, R.B., Taha, H., Liu, C., Nacario, R.C., Schriemer, D.C., Klassen, J.S., Peters, T., Lowary, T.L.: Ligand specificity of CS-35, a monoclonal antibody that recognizes mycobacterial lipoarabinomannan: a model system for oligofuranoside–protein recognition. J. Am. Chem. Soc. 129, 10489–10502 (2007)
Jorgensen, T.J.D., Roepstorff, P., Heck, A.J.R.: Direct determination of solution binding constants for noncovalent complexes between bacterial cell wall peptide analogues and vancomycin group antibiotics by electrospray ionization mass spectrometry. Anal. Chem. 70, 4427–4432 (1998)
Jecklin, M.C., Touboul, D., Bovet, C., Wortmann, A., Zenobi, R.: Which electrospray-based ionization method best reflects protein–ligand interactions found in solution? A comparison of ESI, NanoESI, and ESSI for the determination of dissociation constants with mass spectrometry. J. Am. Soc. Mass Spectrom. 19, 332–343 (2008)
Yu, Y.H., Kirkup, C.E., Pi, N., Leary, J.A.: Characterization of noncovalent protein-ligand complexes and associated enzyme intermediates of GlcNAc-6-O-sulfotransferase by electrospray ionization FT-ICR mass spectrometry. J. Am. Soc. Mass Spectrom. 15, 1400–1407 (2004)
Hofstadler, S.A., Sannes-Lowery, K.A.: Applications of ESI-MS in drug discovery: interrogation of noncovalent complexes. Nat. Rev. Drug Discov. 5, 585–595 (2006)
Ganem, B., Henion, J.D.: Going gently into flight: analyzing noncovalent interactions by mass spectrometry. Bioorg. Med. Chem. 11, 311–314 (2003)
Liu, L., Kitova, E.N., Klassen, J.S.: Quantifying protein–fatty acid interactions using electrospray ionization mass spectrometry. J. Am. Soc. Mass Spectrom. 22, 310–318 (2011)
Lin, H., Kitova, E.N., Klassen, J.S.: Measuring positive cooperativity using the direct ESI-MS assay. Cholera toxin B subunit homopentamer binding to GM1 pentasaccharide. J. Am. Soc. Mass Spectrom. 25, 104–110 (2014)
El-Hawiet, A., Shoemaker, G., Daneshfar, R., Kitova, E.N., Klassen, J.S.: Applications of a catch and release electrospray ionization mass spectrometry assay for carbohydrate library screening. Anal. Chem. 84, 50–58 (2012)
El-Hawiet, A., Kitova, E.N., Klassen, J.S.: Quantifying protein interactions with isomeric carbohydrate ligands using a catch and release electrospray ionization-mass spectrometry assay. Anal. Chem. 85, 7637–7644 (2013)
Han, L., Kitova, E.N., Tan, M., Jiang, X., Klassen, J.S.: Identifying carbohydrate ligands of a norovirus P particle using a catch and release electrospray ionization mass spectrometry assay. J. Am. Soc. Mass Spectrom. 25, 111–119 (2014)
Abzalimov, R.R., Dubin, P.L., Kaltashov, I.A.: Glycosaminoglycans as naturally occurring combinatorial libraries: spectrometry-based strategy for characterization of anti-thrombin interaction strategy with low molecular weight heparin and heparin oligomers. Anal. Chem. 79, 6055–6063 (2007)
Cederkvist, F., Zamfir, A.D., Bahrke, S., Eijsink, V.G.H., Sørlie, M., Peter-Katalinic, J., Peter, M.G.: Identification of a high-affinity-binding oligosaccharide by (+) nanoelectrospray quadrupole time-of-flight tandem mass spectrometry of a noncovalent enzyme-ligand complex. Angew. Chem. Int. Ed. 45, 2429–2434 (2006)
Zdanov, A., Li, Y., Bundle, D.R., Deng, S.-J., MacKenzie, C.R., Narang, S.A., Young, N.M., Cygler, M.: Proc. Natl. Acad. Sci. U. S. A. 91, 6423–6427 (1994)
El-Hawiet, A., Kitova, E.N., Kitov, P.I., Eugenio, L., Ng, K.K., Mulvey, G.L., Dingle, T.C., Szpacenko, A., Armstrong, G.D., Klassen, J.S.: Binding of Clostridium difficile toxins to human milk oligosaccharides. Glycobiology 21, 1217–1227 (2011)
Wu, S., Tao, N., German, J.B., Grimm, R., Lebrilla, C.B.: Development of an annotated library of neutral human milk oligosaccharides. J. Proteome Res. 9, 4138–4151 (2010)
Wu, S., Grimm, R., German, J.B., Lebrilla, C.B.: Annotation and structural analysis of sialylated human milk oligosaccharides. J. Proteome Res. 10, 856–868 (2011)
De Leoz, M.L., Wu, S., Strum, J.S., Niñonuevo, M.R., Gaerlan, S.C., Mirmiran, M., German, J.B., Mills, D.A., Lebrilla, C.B., Underwood, M.A.: A quantitative and comprehensive method to analyze human milk oligosaccharide structures in the urine and feces of infants. Anal. Bioanal. Chem. 405, 4089–4105 (2013)
Ninonuevo, M.R., Park, Y., Yin, H., Zhang, J., Ward, R.E., Clowers, B.H., German, J.B., Freeman, S.L., Killeen, K., Grimm, R., Lebrilla, C.B.: A strategy for annotating the human milk glycome. J. Agric. Food Chem. 54, 7471–7480 (2006)
Sun, J., Kitova, E.N., Wang, W., Klassen, J.S.: Method for distinguishing specific and nonspecific protein-ligand complexes in nanoelectrospray ionization mass spectrometry. Anal. Chem. 78, 3010–3018 (2006)
Hirabayashi, J., Hashidate, T., Arata, Y., Nishi, N., Nakamura, T., Hirashima, M., Urashima, T., Oka, T., Futai, M., Muller, W.E., Yagi, F., Kasai, K.: Oligosaccharide specificity of galectins: a search by frontal affinity chromatography. Biochim. Biophys. Acta 1572, 232–254 (2002)
Acknowledgments
The authors acknowledge the Alberta Glycomics Center and the Natural Sciences and Engineering Research Council of Canada for funding, and Professor D. Bundle (University of Alberta) for generously providing some of the oligosaccharides used in this study, Professor K. Ng (University of Calgary) for the gift of TcdB fragment, and Professor C. Cairo (University of Alberta) for providing the fragment of human galectin 3.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 551 kb)
Rights and permissions
About this article
Cite this article
Kitova, E.N., El-Hawiet, A. & Klassen, J.S. Screening Carbohydrate Libraries for Protein Interactions Using the Direct ESI-MS Assay. Applications to Libraries of Unknown Concentration. J. Am. Soc. Mass Spectrom. 25, 1908–1916 (2014). https://doi.org/10.1007/s13361-014-0964-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13361-014-0964-2