Abstract
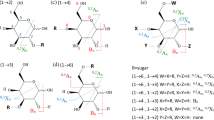
A systematic approach is described that can pinpoint the stereo-structures (sugar identity, anomeric configuration, and location) of individual sugar units within linear oligosaccharides. Using a highly modified mass spectrometer, dissociation of linear oligosaccharides in the gas phase was optimized along multiple-stage tandem dissociation pathways (MSn, n = 4 or 5). The instrument was a hybrid triple quadrupole/linear ion trap mass spectrometer capable of high-efficiency bidirectional ion transfer between quadrupole arrays. Different types of collision-induced dissociation (CID), either on-resonance ion trap or beam-type CID could be utilized at any given stage of dissociation, enabling either glycosidic bond cleavages or cross-ring cleavages to be maximized when wanted. The approach first involves optimizing the isolation of disaccharide units as an ordered set of overlapping substructures via glycosidic bond cleavages during early stages of MSn, with explicit intent to minimize cross-ring cleavages. Subsequently, cross-ring cleavages were optimized for individual disaccharides to yield key diagnostic product ions (m/z 221). Finally, fingerprint patterns that establish stereochemistry and anomeric configuration were obtained from the diagnostic ions via CID. Model linear oligosaccharides were derivatized at the reducing end, allowing overlapping ladders of disaccharides to be isolated from MSn. High confidence stereo-structural determination was achieved by matching MSn CID of the diagnostic ions to synthetic standards via a spectral matching algorithm. Using this MSn (n = 4 or 5) approach, the stereo-structures, anomeric configurations, and locations of three individual sugar units within two pentasaccharides were successfully determined.

ᅟ




Similar content being viewed by others
References
Apweiler, R., Hermjakob, H., Sharon, N.: On the frequency of protein glycosylation, as deduced from analysis of the SWISS-PROT database. Biochim. Biophys. Acta 1473, 4–8 (1999)
Eichler, J., Michael, W.W.A.: Post-translational protein modification in Archaea. Microbiol. Mol. Biol. Rev. 69, 393–425 (2005)
Mescher, M.F., Strominger, J.L.: Purification and characterization of a prokaryotic glucoprotein from the cell envelope of Halobacterium salinarium. J. Biol. Chem. 251, 2005–2014 (1976)
Abu-Qarn, M., Eichler, J., Sharon, N.: Not just for Eukarya anymore: protein glycosylation in bacteria and archaea. Curr. Opin. Struct. Biol. 18, 544–550 (2008)
Perkel, J.M.: Life science technologies: Glycoproteomics: the sweet smell of we’re-getting-there. Science 331, 95–97 (2011)
Geyer, H., Geyer, R.: Strategies for analysis of glycoprotein glycosylation. Biochim. Biophys. Acta Gen. Subj. 1764, 1853–1869 (2006)
Duus, J., Gotfredsen, C.H., Bock, K.: Carbohydrate structural determination by NMR spectroscopy: modern methods and limitations. Chem. Rev. 100, 4589–4614 (2000)
Fellenberg, M., Coksezen, A., Meyer, B.: Characterization of picomole amounts of oligosaccharides from glycoproteins by 1H NMR spectroscopy. Angew. Chem. Int. Ed. 49, 2630–2633 (2010)
Tseng, K., Hedrick, J.L., Lebrilla, C.B.: Catalog-library approach for the rapid and sensitive structural elucidation of oligosaccharides. Anal. Chem. 71, 3747–3754 (1999)
Zaia, J.: Mass spectrometry of oligosaccharides. Mass Spectrom. Rev. 23, 161–227 (2004)
Reinhold, V.N., Reinhold, B.B., Costello, C.E.: Carbohydrate molecular weight profiling, sequence, linkage, and branching data: ES-MS and CID. Anal. Chem. 67, 1772–1784 (1995)
Peltier, J.M., Maclean, D.B., Szarek, W.A.: Determination of the glycosidic linkage in peracetylated disaccharides comprised of D-glucopyranose units by use of desorption electron-ionization mass-spectrometry. Rapid Commun. Mass Spectrom. 5, 446–449 (1991)
Asam, M.R., Glish, G.L.: Tandem mass spectrometry of alkali cationized polysaccharides in a quadrupole ion trap. J. Am. Soc. Mass Spectrom. 8, 987–995 (1997)
Polfer, N.C., Valle, J.J., Moore, D.T., Oomens, J., Eyler, J.R., Bendiak, B.: Differentiation of isomers by wavelength-tunable infrared multiple-photon dissociation-mass spectrometry: application to glucose-containing disaccharides. Anal. Chem. 78, 670–679 (2006)
Dongré, A.R., Wysocki, V.H.: Linkage position determination of lithium cationized disaccharides by surface-induced dissociation tandem mass spectrometry. Org. Mass Spectrom. 29, 700–702 (1994)
Domon, B., Müller, D.R., Richter, W.J.: High performance tandem mass spectrometry for sequence, branching and interglycosidic linkage analysis of peracetylated oligosaccharides. Biol. Mass Spectrom. 19, 390–392 (1990)
Sheeley, D.M., Reinhold, V.N.: Structural characterization of carbohydrate sequence, linkage, and branching in a quadrupole ion trap mass spectrometer: neutral oligosaccharides and N-linked glycans. Anal. Chem. 70, 3053–3059 (1998)
Chai, W., Lawson, A., Piskarev, V.: Branching pattern and sequence analysis of underivatized oligosaccharides by combined MS/MS of singly and doubly charged molecular ions in negative-ion electrospray mass spectrometry. J. Am. Soc. Mass Spectrom. 13, 670–679 (2002)
Hofmeister, G.E., Zhou, Z., Leary, J.A.: Linkage position determination in lithium-cationized disaccharides: tandem mass spectrometry and semiempirical calculations. J. Am. Chem. Soc. 113, 5964–5970 (1991)
Xie, Y., Lebrilla, C.B.: Infrared multiphoton dissociation of alkali metal-coordinated oligosaccharides. Anal. Chem. 75, 1590–1598 (2003)
Reilly, J.P.: Ultraviolet photofragmentation of biomolecular ions. Mass Spectrom. Rev. 28, 425–447 (2009)
Adamson, J.T., Håkansson, K.: Electron capture dissociation of oligosaccharides ionized with alkali, alkaline earth, and transition metals. Anal. Chem. 79, 2901–2910 (2007)
Zhao, C., Xie, B., Chan, S.-Y., Costello, C., O’Connor, P.: Collisionally activated dissociation and electron capture dissociation provide complementary structural information for branched permethylated oligosaccharides. J. Am. Soc. Mass Spectrom. 19, 138–150 (2008)
Wolff, J., Laremore, T., Aslam, H., Linhardt, R., Amster, I.J.: Electron-induced dissociation of glycosaminoglycan tetrasaccharides. J. Am. Soc. Mass Spectrom. 19, 1449–1458 (2008)
Wolff, J., Amster, I.J., Chi, L., Linhardt, R.: Electron detachment dissociation of glycosaminoglycan tetrasaccharides. J. Am. Soc. Mass Spectrom. 18, 234–244 (2007)
Yu, X., Huang, Y., Lin, C., Costello, C.E.: Energy-dependent electron activated dissociation of metal-adducted permethylated oligosaccharides. Anal. Chem. 84, 7487–7494 (2012)
Gennaro, L.A., Harvey, D.J., Vouros, P.: Reversed-phase ion-pairing liquid chromatography/ion trap mass spectrometry for the analysis of negatively charged, derivatized glycans. Rapid Commun. Mass Spectrom. 17, 1528–1534 (2003)
Domon, B., Mueller, D.R., Richter, W.J.: Tandem mass-spectrometric analysis of fixed-charge derivatized oligosaccharides. Org. Mass Spectrom. 29, 713–719 (1994)
Bendiak, B., Fang, T.T.: Assignment of the stereochemistry and anomeric configuration of structurally informative product ions derived from disaccharides: infrared photodissociation of glycosyl-glycolaldehydes in the negative ion mode. Carbohydr. Res. 345, 2390–2400 (2010)
Laine, R.A.: A calculation of all possible oligosaccharide isomers both branched and linear yields 1.05 × 1012 structures for a reducing hexasaccharide - the isomer-barrier to development of single-method saccharide sequencing or synthesis systems. Glycobiology 4, 759–767 (1994)
Gaucher, S.P., Leary, J.A.: Determining anomericity of the glycosidic bond in Zn(II)-diethylenetriamine–disaccharide complexes using MSn in a quadrupole ion trap. J. Am. Soc. Mass Spectrom. 10, 269–272 (1999)
Smith, G., Leary, J.A.: Differentiation of stereochemistry of glycosidic bond configuration: tandem mass spectrometry of diastereomeric cobalt-glucosyl-glucose disaccharide complexes. J. Am. Soc. Mass Spectrom. 7, 953–957 (1996)
Mulroney, B., Traeger, J.C., Stone, B.A.: Determination of both linkage position and anomeric configuration in underivatized glucopyranosyl disaccharides by electrospray mass spectrometry. J. Mass Spectrom. 30, 1277–1283 (1995)
Guan, B., Cole, R.B.: MALDI linear-field reflectron TOF post-source decay analysis of underivatized oligosaccharides: determination of glycosidic linkages and anomeric configurations using anion attachment. J. Am. Soc. Mass Spectrom. 19, 1119–1131 (2008)
Simoes, J., Domingues, P., Reis, A., Nunes, F.M., Coimbra, M.A., Domingues, R.M.: Identification of anomeric configuration of underivatized reducing glucopyranosyl-glucose Disaccharides by tandem mass Spectrometry and Multivariate analysis. Anal. Chem. 79, 5896–5905 (2007)
Gaucher, S.P., Leary, J.A.: Stereochemical differentiation of mannose, glucose, galactose, and talose using zinc(II) diethylenetriamine and ESI-ion trap mass spectrometry. Anal. Chem. 70, 3009–3014 (1998)
Salpin, J.Y., Tortajada, J.: Structural characterization of hexoses and pentoses using lead cationization. An electrospray ionization and tandem mass spectrometric study. J. Mass Spectrom. 37, 379–388 (2002)
Zhu, X.Y., Sato, T.: The distinction of underivatized monosaccharides using electrospray ionization ion trap mass spectrometry. Rapid Commun. Mass Spectrom. 21, 191–198 (2007)
Brown, D.J., Stefan, S.E., Berden, G., Steill, J.D., Oomens, J., Eyler, J.R., Bendiak, B.: Direct evidence for the ring opening of monosaccharide anions in the gas phase: photodissociation of aldohexoses and aldohexoses derived from disaccharides using variable-wavelength infrared irradiation in the carbonyl stretch region. Carbohydr. Res. 346, 2469–2481 (2011)
Augusti, D.V., Carazza, F., Augusti, R., Tao, W.A., Cooks, R.G.: Quantitative chiral analysis of sugars by electrospray ionization tandem mass spectrometry using modified amino acids as chiral reference compounds. Anal. Chem. 74, 3458–3462 (2002)
Ashline, D., Singh, S., Hanneman, A., Reinhold, V.: Congruent strategies for carbohydrate sequencing. 1. Mining structural details by MSn. Anal. Chem. 77, 6250–6262 (2005)
Ashline, D.J., Lapadula, A.J., Liu, Y.-H., Lin, M., Grace, M., Pramanik, B., Reinhold, V.N.: Carbohydrate structural isomers analyzed by sequential mass spectrometry. Anal. Chem. 79, 3830–3842 (2007)
Fang, T.T., Bendiak, B.: The stereochemical dependence of unimolecular dissociation of monosaccharide-glycolaldehyde anions in the gas phase: a basis for assignment of the stereochemistry and anomeric configuration of monosaccharides in oligosaccharides by mass spectrometry via a key discriminatory product ion of disaccharide fragmentation, m/z 221. J. Am. Chem. Soc. 129, 9721–9736 (2007)
Viseux, N., de Hoffmann, E., Domon, B.: Structural assignment of permethylated oligosaccharide subunits using sequential tandem mass spectrometry. Anal. Chem. 70, 4951–4959 (1998)
Garozzo, D., Giuffrida, M., Impallomeni, G., Ballistreri, A., Montaudo, G.: Determination of linkage position and identification of the reducing end in linear oligosaccharides by negative ion fast atom bombardment mass spectrometry. Anal. Chem. 62, 279–286 (1990)
Dallinga, J.W., Heerma, W.: Reaction mechanism and fragment ion structure determination of deprotonated small oligosaccharides, studied by negative ion fast atom bombardment (tandem) mass spectrometry. Biol. Mass Spectrom. 20, 215–231 (1991)
Carroll, J.A., Willard, D., Lebrilla, C.B.: Energetics of cross-ring cleavages and their relevance to the linkage determination of oligosaccharides. Anal. Chim. Acta. 307, 431–447 (1995)
Fang, T.T., Zirrolli, J., Bendiak, B.: Differentiation of the anomeric configuration and ring form of glucosyl-glycolaldehyde anions in the gas phase by mass spectrometry: isomeric discrimination between m/z 221 anions derived from disaccharides and chemical synthesis of m/z 221 standards. Carbohydr. Res. 342, 217–235 (2007)
Konda, C., Bendiak, B., Xia, Y.: Differentiation of the stereochemistry and anomeric configuration for 1-3 linked disaccharides via tandem mass spectrometry and 18O-labeling. J. Am. Soc. Mass Spectrom. 23, 347–358 (2012)
Harvey, D.J.: Electrospray mass spectrometry and fragmentation of N-linked carbohydrates derivatized at the reducing terminus. J. Am. Soc. Mass Spectrom. 11, 900–915 (2000)
Londry, F.A., Hager, J.: Mass selective axial ion ejection from a linear quadrupole ion trap. J. Am. Soc. Mass Spectrom. 14, 1130–1147 (2003)
Zhang, Z.: Prediction of low-energy collision-induced dissociation spectra of peptides. Anal. Chem. 76, 3908–3922 (2004)
Domon, B., Costello, C.E.: A systematic nomenclature for carbohydrate fragmentations in FAB-MS/MS spectra of glycoconjugates. Glycoconj. J. 5, 397–409 (1988)
Thomson, B.A.: Apparatus and method for MSnth in a tandem mass spectrometer system. U.S. Patent 7 145, 133–B142 (2006)
Xia, Y., Thomson, B.A., McLuckey, S.A.: Bidirectional ion transfer between quadrupole arrays: MSn ion/ion reaction experiments on a quadrupole/time-of-flight tandem mass spectrometer. Anal. Chem. 79, 8199–8206 (2007)
Thomson, B.A., Jolliffe, C.L.: Spectrometer with axial field. U.S. Patent 5 847, 386 (1998)
Loboda, A., Krutchinsky, A., Loboda, O., McNabb, J., Spicer, V., Ens, W., Standing, K.: Novel linad II electrode geometry for creating an axial field in a mulitpole ion guide. Eur. J. Mass Spectrom. 6, 531–536 (2000)
Collings, B.A.: Fragmentation of ions in a low pressure linear ion trap. J. Am. Soc. Mass Spectrom. 18, 1459–1466 (2007)
Morris, M., Pierre, T., Boyd, R.K.: Characterization of a high-pressure quadrupole collision cell for low-energy collision-induced dissociation. J. Am. Soc. Mass Spectrom. 5, 1042–1063 (1994)
Acknowledgements
The authors thank Y. Du and Professor Z. Ouyang for conducting spectral similarity score calculations and Dr. L. J. Campbell for helpful discussions. C.K. and Y.X. thank the support from Purdue Research Foundation. B.B. acknowledges financial support from NSF CHE-0137986 and CHE-0718007.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 716 kb)
Rights and permissions
About this article
Cite this article
Konda, C., Londry, F.A., Bendiak, B. et al. Assignment of the Stereochemistry and Anomeric Configuration of Sugars within Oligosaccharides Via Overlapping Disaccharide Ladders Using MSn . J. Am. Soc. Mass Spectrom. 25, 1441–1450 (2014). https://doi.org/10.1007/s13361-014-0881-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13361-014-0881-4