Abstract
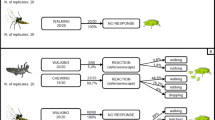
Female parasitoids distinguish between host-infested and intact plants using chemical cues; however, the contribution of intact plants to host searching of parasitoids has not been investigated so far. Here, we tested how host-searching behavior of the parasitoid wasp, Cotesia kariyai (Watanabe) (Hymenoptera: Braconidae), was affected by intact maize plants in a wind tunnel. To determine the best color for material to create a plant model, we observed flight responses of female wasps to paper plant models of four different colors. Wasps tended to land more frequently on green models than other models. Therefore, a green paper model was used for subsequent experiments. In a no-choice test, female wasps showed higher landing rates on a paper plant model treated with herbivore-induced plant volatiles (HIPVs) than on an intact plant. Moreover, in two-choice tests, wasps preferred the plant model with HIPVs over an intact plant with HIPVs. Intact plants seem to deter C. kariyai females. Our findings suggest that information from intact plants also contributes to the host-searching behavior of females in the natural environment.


Similar content being viewed by others
References
Aartsma Y, Bianchi FJJA, van der Werf W, Poelman EH, Dicke M (2017) Herbivore-induced plant volatiles and tritrophic interactions across spatial scales. New Phytol 216:1054–1063. https://doi.org/10.1111/nph.14475
Aartsma Y, Leroy B, van der Werf W, Dicke M, Poelman EH, Bianchi FJJA (2019) Intraspecific variation in herbivore-induced plant volatiles influences the spatial range of plant–parasitoid interactions. Oikos 128:77–86. https://doi.org/10.1111/oik.05151
Afsheen S, Wang X, Li R, Zhu C, Lou Y (2008) Differential attraction of parasitoids in relation to specificity of kairomones from herbivores and their by-products. Insect Sci 15:381–397. https://doi.org/10.1111/j.1744-7917.2008.00225.x
Fujiwara C, Takabayashi J, Yano S (2000) Effects of host-food plant species on parasitization rates of Mythimna separata (Lepidoptera: Noctuidae) by a parasitoid, Cotesia kariyai (Hymenoptera: Braconidae). Appl Entomol Zool 35:131–136. https://doi.org/10.1303/aez.2000.131
Fukushima J, Kainoh Y, Honda H, Takabayashi J (2001) Learning of host-infested plant volatiles in the larval parasitoid Cotesia kariyai. Entomol Exp Appl 99:341–346. https://doi.org/10.1046/j.1570-7458.2001.00833.x
Godfray HCJ (1994) Parasitoids behavioral and evolutionary ecology. Princeton University Press, Princeton
Gohole LS, Overholt WA, Khan ZR, Vet LEM (2003) Role of volatiles emitted by host and non-host plants in the foraging behaviour of Dentichasmias busseolae, a pupal parasitoid of the spotted stemborer Chilo partellus. Entomol Exp Appl 107:1–9. https://doi.org/10.1046/j.1570-7458.2003.00030.x
Gohole LS, Overholt WA, Khan ZR, Vet LEM (2005) Close-range host searching behavior of the stemborer parasitoids Cotesiasesamiae and Dentichasmiasbusseolae: influence of a non-host plant Melinis minutiflora. J Insect Behav 18:149–169. https://doi.org/10.1007/s10905-005-0472-0
Hatano E, Kunert G, Michaud JP, Weisser WW (2008) Chemical cues mediating aphid location by natural enemies. Eur J Entomol 105:797–806
Henneman ML, Dyreson EG, Takabayashi J, Raguso RA (2002) Response to walnut olfactory and visual cues by the parasitic wasp Diachasmimorpha juglandis. J Chem Ecol 28:2221–2224. https://doi.org/10.1023/A:1021097215572
Horikoshi M, Takabayashi J, Yano S, Yamaoka R, Ohsaki N, Sato Y (1997) Cotesia glomerata female wasps use fatty acids from plant–herbivore complex in host searching. J Chem Ecol 23:1505–1515. https://doi.org/10.1023/B:JOEC.0000006418.49537.5d
Hou M, Takabayashi J, Kainoh Y (2005) Effect of leaf age on flight response of a parasitic wasp Cotesia kariyai (Hymenoptera: Braconidae) to a plant–herbivore complex. Appl Entomol Zool 40:113–117. https://doi.org/10.1303/aez.2005.113
Ichiki RT, Kainoh Y, Yamawaki Y, Nakamura S (2011) The parasitoid fly Exorista japonica uses visual and olfactory cues to locate herbivore-infested plants. Entomol Exp Appl 138:175–183. https://doi.org/10.1111/j.1570-7458.2010.01091.x
Kawamata R, Sato Y, Suzuki M, Kainoh Y (2018) Color preference and associative color learning in a parasitoid wasp, Ascogaster reticulata (Hymenoptera: Braconidae). J Insect Behav 31:523–534. https://doi.org/10.1007/s10905-018-9696-7
Kuramitsu K, Vicencio EJM, Kainoh Y (2019) Differences in food plant species of the polyphagous herbivore Mythimna separata (Lepidoptera: Noctuidae) influence host searching behavior of its larval parasitoid, Cotesia kariyai (Hymenoptera: Braconidae). Arthropod Plant Interact 13:49–55. https://doi.org/10.1007/s11829-018-9659-0
Martin WR, Nordlund DA, Nettles WC (1990) Response of parasitoid Eucelatoria bryani to selected plant material in an olfactometer. J Chem Ecol 16:499–508. https://doi.org/10.1007/BF01021781
McCormick AC, Unsicker SB, Gershenzon J (2012) The specificity of herbivore-induced plant volatiles in attracting herbivore enemies. Trends Plant Sci 17:303–310. https://doi.org/10.1016/j.tplants.2012.03.012
Meiners T, Peri E (2013) Chemical ecology of insect parasitoids: essential elements for developing effective biological control programmes. In: Wajnberg E, Colazza S (eds) Chemical ecology of insect parasitoids. Wiley-Blackwell, Chichester, pp 193–224
Ozawa R, Ohara Y, Shiojiri K, Uchida T, Kakibuchi K, Kugimiya S, Uefune M, Takabayashi J (2018) Uninfested plants and honey enhance the attractiveness of a volatile blend to a parasitoid Cotesia vestalis. J Appl Entomol 142:978–984. https://doi.org/10.1111/jen.12537
Pareja M, Moraes MCB, Clark SJ, Birkett MA, Powell W (2007) Response of the aphid parasitoid Aphidius funebris to volatiles from undamaged and aphid-infested Centaurea nigra. J Chem Ecol 33:695–710. https://doi.org/10.1007/s10886-007-9260-y
Quicke DLJ (2015) The braconid and ichneumonid parasitoid wasps: biology, systematics, evolution and ecology. John Wiley & Sons, Chichester
R Core Team (2015) R: a language and environment for statistical computing, R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. Accessed 1 Dec 2018
Romeis J, Shanower TG, Zebitz CPW (1997) Volatile plant infochemicals mediate plant preference of Trichogramma chilonis. J Chem Ecol 23:2455–2465. https://doi.org/10.1023/B:JOEC.0000006659.06960.ed
Takabayashi J, Noda T, Takahashi S (1985) Effects of kairomones in the host searching behavior of Apanteles kariyai Watanabe (Hymenoptera: Braconidae), a parasitoid of the common armyworm, Pseudaletia separata Walker (Lepidoptera: Noctuidae). I. Presence of arresting stimulants produced by the host larvae. Appl Entomol Zool 20:484–489
Takabayashi J, Noda T, Takahashi S (1991) Plants produce attractants for Apanteles kariyai, a parasitoid of Pseudaletia separata; cases of “communication” and “misunderstanding” in parasitoid-plant interactions. Appl Entomol Zool 26:237–243
Takabayashi J, Takahashi S, Dicke M, Posthumus MA (1995) Developmental stage of herbivore Pseudaletia separata affects production of herbivore-induced synomone by corn plants. J Chem Ecol 21:273–287. https://doi.org/10.1007/BF02036717
Thanikkul P, Piyasaengthong N, Menezes-Netto AC, Taylor D, Kainoh Y (2017) Effects of quantitative and qualitative differences in volatiles from host- and non-host-infested maize on the attraction of the larval parasitoid Cotesia kariyai. Entomol Exp Appl 163:60–69. https://doi.org/10.1111/eea.12555
Turlings TCJ, Erb M (2018) Tritrophic interactions mediated by herbivore-induced plant volatiles: mechanisms, ecological relevance, and application potential. Annu Rev Entomol 63:433–452. https://doi.org/10.1146/annurev-ento-020117-043507
Uefune M, Kugimiya S, Shimada T, Takabayashi J (2013) Starvation and herbivore-induced plant volatiles affect the color preferences of parasitic wasps. Biocontrol 58:187–193. https://doi.org/10.1007/s10526-012-9483-x
Vet L, Dicke M (1992) Ecology of infochemical use by natural enemies in a tritrophic context. Annu Rev Entomol 37:141–172. https://doi.org/10.1146/annurev.en.37.010192.001041
Vinson SB (1976) Host selection by insect parasitoids. Annu Rev Entomol 21:109–133. https://doi.org/10.1146/annurev.en.21.010176.000545
Wäschke N, Meiners T, Rostas M (2013) Foraging strategies of parasitoids in complex chemical environments. In: Wajnberg E, Colazza S (eds) Chemical ecology of insect parasitoids. Wiley-Blackwell, Chichester, pp 37–63
Yamawaki Y, Kainoh Y (2005) Visual recognition of the host in the parasitoid fly Exorista japonica. Zool Sci 22:563–570. https://doi.org/10.2108/zsj.22.563
Acknowledgements
We thank Prof. Hiroshi Honda, Prof. DeMar Taylor, and Assoc. Prof. Seiichi Furukawa for their helpful comments and advice on this study. We also thank Prof. Junji Takabayashi for supplying the colony of Cotesia kariyai. We are grateful to Ms. Rumi Yoshimura for her help and support in conducting this study. This research did not receive any grants from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
13355_2020_669_MOESM1_ESM.pptx
Supplementary file1. Online Resource 1 Reflectance spectra of colored papers, infested plants, and intact plants used in these experiments. (PPTX 102 kb)
Rights and permissions
About this article
Cite this article
Isono, K., Kuramitsu, K. & Kainoh, Y. Deterrent effects of intact plants on host-searching behavior of parasitoid wasps. Appl Entomol Zool 55, 199–204 (2020). https://doi.org/10.1007/s13355-020-00669-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13355-020-00669-8