Abstract
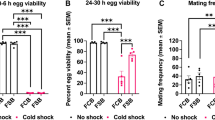
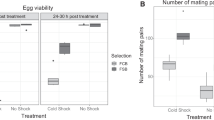
Tolerance to extreme temperatures is one of the most important components of adaptation to environments in insects. However, the genetic mechanisms of tolerance to long-term heat stress are mostly unknown. In this study, we investigated the genetic variation of susceptibility to long-term heat stress among the isofemale strains of Drosophila prolongata Singh & Gupta. High rates of pupal lethality were observed at 25 °C, showing that D. prolongata is more susceptible to heat stress than other Drosophila species. Susceptibility to heat stress was significantly different among the isofemale strains, suggesting that intraspecific genetic variation is involved in the reduced heat tolerance of D. prolongata. Unexpectedly, the tertiary sex ratio was biased to females at temperatures higher than 20 °C, indicating that the males were more susceptible to heat stress. These results demonstrated that D. prolongata is useful for genetic analysis for elucidating the molecular mechanisms of heat tolerance in insects.


Similar content being viewed by others
References
Bettencourt BR, Feder ME (2001) Hsp70 duplication in the Drosophila melanogaster species group: how and when did two become five? Mol Biol Evol 18(7):1272–1282. doi:10.1093/oxfordjournals.molbev.a003912
Calabria G, Dolgova O, Rego C, Castaneda LE, Rezende EL, Balanya J, Pascual M, Sorensen JG, Loeschcke V, Santos M (2012) Hsp70 protein levels and thermotolerance in Drosophila subobscura: a reassessment of the thermal co-adaptation hypothesis. J Evol Biol 25(4):691–700. doi:10.1111/j.1420-9101.2012.02463.x
Chen H, Xu XL, Li YP, Wu JX (2014) Characterization of heat shock protein 90, 70 and their transcriptional expression patterns on high temperature in adult of Grapholita molesta (Busck). Insect Sci 21(4):439–448. doi:10.1111/1744-7917.12057
Crosthwaite JC, Sobek S, Lyons DB, Bernards MA, Sinclair BJ (2011) The overwintering physiology of the emerald ash borer, Agrilus planipennis fairmaire (Coleoptera: Buprestidae). J Insect Physiol 57(1):166–173. doi:10.1016/j.jinsphys.2010.11.003
Dobzhansky T (1935) Fecundity in Drosophila pseudoobscura at different temperatures. J Exp Zool 71:449–464
Hurst GDD, Johnson AP, von der Schulenburg JHG, Fuyama Y (2000) Male-killing Wolbachia in Drosophila: a temperature-sensitive trait with a threshold bacterial density. Genetics 156(2):699–709
Ichiki R, Takasu K, Shima H (2003) Effects of temperature on immature development of the parasitic fly Bessa parallela (Meigen) (Diptera: Tachinidae). Appl Entomol Zool 38(4):435–439. doi:10.1303/aez.2003.435
Kellermann V, Overgaard J, Hoffmann AA, Flojgaard C, Svenning JC, Loeschcke V (2012) Upper thermal limits of Drosophila are linked to species distributions and strongly constrained phylogenetically. Proc Natl Acad Sci USA 109(40):16228–16233. doi:10.1073/pnas.1207553109
Kudo A, Takamori H, Watabe H, Ishikawa Y, Matsuo T (2015) Variation in morphological and behavioral traits among isofemale strains of Drosophila prolongata (Diptera: Drosophilidae). Entomol Sci 18(2):221–229. doi:10.1111/ens.12116
Moiroux J, Brodeur J, Boivin G (2014) Sex ratio variations with temperature in an egg parasitoid: behavioural adjustment and physiological constraint. Anim Behav 91:61–66. doi:10.1016/j.anbehav.2014.02.021
Omkar, Pervez A (2004) Temperature-dependent development and immature survival of an aphidophagous ladybeetle Propylea dissecta (Mulsant). J Appl Entomol 128(7):510–514. doi:10.1111/j.1439-0418.2004.00881.510-514
Pakyari H, Fathipour Y, Enkegaard A (2011) Effect of temperature on life table parameters of predatory thrips Scolothrips longicornis (Thysanoptera: Thripidae) fed on twospotted spider mites (Acari: Tetranychidae). J Econ Entomol 104(3):799–805. doi:10.1603/EC10144
Parsell DA, Lindquist S (1993) The function of heat-shock proteins in stress tolerance—degradation and reactivation of damaged proteins. Annu Rev Genet 27:437–496. doi:10.1146/annurev.ge.27.120193.002253
Petavy G, David JR, Gibert P, Moreteau B (2001) Viability and rate of development at different temperatures in Drosophila: a comparison of constant and alternating thermal regimes. J Therm Biol 26(1):29–39. doi:10.1016/S0306-4565(00)00022-X
R Development Core Team (2008) R: a language and environment for statistical computing. R foundation for statistical computing, vienna, Austria. ISBN 3-900051-07-0, http://www.R-project.org
Singh BK, Gupta JP (1977) Two new and two unrecorded species of the genus Drosophila fallen (Diptera: Drosophilidae) from Shillong, Meghalaya, India. Proc Zool Soc 30:31–38
Takahashi KH, Okada Y, Teramura K (2011) Genome-wide deficiency screen for the genomic regions responsible for heat resistance in Drosophila melanogaster. BMC Genet 12:57. doi:10.1186/1471-2156-12-57
Toda MJ (1991) Drosophilidae (Diptera) in Myanmar (Burma) VII. The Drosophila melanogaster species-group, excepting the D. montium species-subgroup. Orient Insects 25:69–94
Tungjitwitayakul J, Tatun N, Vajarasathira B, Sakurai S (2015) Expression of heat shock protein genes in different developmental stages and after temperature stress in the maize weevil (Coleoptera: Curculionidae). J Econ Entomol 108(3):1313–1323. doi:10.1093/jee/tov051
Werren JH, Baldo L, Clark ME (2008) Wolbachia: master manipulators of invertebrate biology. Nat Rev Microbiol 6(10):741–751. doi:10.1038/nrmicro1969
Wharton DA (2011) Cold tolerance of New Zealand alpine insects. J Insect Physiol 57(8):1090–1095. doi:10.1016/j.jinsphys.2011.03.004
Acknowledgments
This work was supported by JSPS KAKENHI Grant Number 26660263 to TM.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hitoshi, Y., Ishikawa, Y. & Matsuo, T. Intraspecific variation in heat tolerance of Drosophila prolongata (Diptera: Drosophilidae). Appl Entomol Zool 51, 515–520 (2016). https://doi.org/10.1007/s13355-016-0425-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13355-016-0425-4