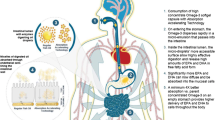
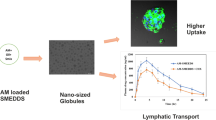
Abstract
This study explored the design of supersaturable self-microemulsifying drug delivery systems (S-SMEDDS) to address poor solubility and oral bioavailability of a novel benzimidazole derivative anticancer drug (BI). Firstly, self-microemulsifying drug delivery systems SMEDDS made of Miglyol® 812, Kolliphor® RH40, Transcutol® HP, and ethanol were prepared and loaded with the BI drug. Upon dispersion, the systems formed neutrally charged droplets of around 20 nm. However, drug precipitation was observed following incubation with simulated gastric fluid (pH 1.2). Aiming at reducing this precipitation and enhancing drug payload, supersaturable systems were then prepared by adding 1% hydroxypropyl cellulose as precipitation inhibitor. Supersaturable systems maintained a higher amount of drug in a supersaturated state in gastric medium compared with conventional formulations and were stable in simulated intestinal medium (pH 6.8). In vitro cell studies using Caco-2 cell line showed that these formulations reduced in a transient manner the transepithelial electrical resistance of the monolayers without toxicity. Accordingly, confocal images revealed that the systems accumulated at tight junctions after a 2 h exposure. In vivo pharmacokinetic studies carried out following oral administration of BI-loaded S-SMEDDS, SMEDDS, and free drug to healthy mice showed that supersaturable systems promoted drug absorption compared with the other formulations. Overall, these data highlight the potential of using the supersaturable approach as an alternative to conventional SMEDDS for improving oral systemic absorption of lipophilic drugs.
Graphical abstract








Similar content being viewed by others
References
Filipski KJ, Varma MV, El-Kattan AF, Ambler CM, Ruggeri RB, Goosen TC, et al. Intestinal targeting of drugs: rational design approaches and challenges. Curr Top Med Chem. 2013;13:776–802.
Rosso A, Lollo G, Chevalier Y, Troung N, Bordes C, Bourgeois S, et al. Development and structural characterization of a novel nanoemulsion for oral drug delivery. Colloids Surf A Physicochem Eng Asp. 2020;593:124614.
Lollo G, Gonzalez-Paredes A, Garcia-Fuentes M, Calvo P, Torres D, Alonso MJ. Polyarginine nanocapsules as a potential oral peptide delivery carrier. J Pharm Sci. 2017;106:611–8.
Gupta S, Kesarla R, Omri A. Formulation strategies to improve the bioavailability of poorly absorbed drugs with special emphasis on self-emulsifying systems. ISRN Pharm. 2013;2013:1–16.
Dumont C, Bourgeois S, Fessi H, Jannin V. Lipid-based nanosuspensions for oral delivery of peptides, a critical review. Int J Pharm. 2018;541:117–35.
Buya AB, Ucakar B, Beloqui A, Memvanga PB, Préat V. Design and evaluation of self-nanoemulsifying drug delivery systems (SNEDDSs) for senicapoc. Int J Pharm. 2020;580:119180.
Desai P, Thakkar A, Ann D, Wang J, Prabhu S. Loratadine self-microemulsifying drug delivery systems (SMEDDS) in combination with sulforaphane for the synergistic chemoprevention of pancreatic cancer. Drug Deliv Transl Res. 2019;9:641–51.
Patel P, Pailla SR, Rangaraj N, Cheruvu HS, Dodoala S, Sampathi S. Quality by design approach for developing lipid-based nanoformulations of gliclazide to improve oral bioavailability and anti-diabetic activity. AAPS Pharm Sci Tech. 2019;20:45.
Pouton CW. Lipid formulations for oral administration of drugs: Non-emulsifying, self-emulsifying and ‘self-microemulsifying’ drug delivery systems. Eur J Pharm Sci. 2000;11:93–8.
Ruckenstein E. The origin of thermodynamic stability of microemulsions. Chem Phys Lett. 1978;57:517–21.
Mukherjee T, Plakogiannis FM. Development and oral bioavailability assessment of a supersaturated self-microemulsifying drug delivery system (SMEDDS) of albendazole. J Pharm Pharmacol. 2010;62:1112–20.
Dokania S, Joshi AK. Self-microemulsifying drug delivery system (SMEDDS)-challenges and road ahead. Drug Deliv. 2015;22:675–90.
Sawatdee S, Atipairin A, Yoon AS, Srichana T, Changsan N, Suwandecha T. Formulation development of albendazole-loaded self-microemulsifying chewable tablets to enhance dissolution and bioavailability. Pharmaceutics. 2019;11.
Chatterjee B, Hamed Almurisi S, Ahmed Mahdi Dukhan A, Mandal UK, Sengupta P. Controversies with self-emulsifying drug delivery system from pharmacokinetic point of view. Drug Deliv. 2016;23:3639–52.
Nasr A, Gardouh A, Ghorab M. Novel solid self-nanoemulsifying drug delivery system (S-SNEDDS) for oral delivery of olmesartan medoxomil: Design, formulation, pharmacokinetic and bioavailability evaluation. Pharmaceutics. 2016;8.
Park H, Ha E, Kim M. Current status of supersaturable self-emulsifying drug delivery systems. Pharmaceutics. 2020;12:365.
Larsen AT, Ohlsson AG, Polentarutti B, Barker RA, Phillips AR, Abu-Rmaileh R, et al. Oral bioavailability of cinnarizine in dogs: Relation to SNEDDS droplet size, drug solubility and in vitro precipitation. Eur J Pharm Sci. 2013;48:339–50.
Raut S, Karzuon B, Atef E. Using in situ Raman spectroscopy to study the drug precipitation inhibition and supersaturation mechanism of Vitamin E TPGS from self-emulsifying drug delivery systems (SEDDS). J Pharm Biomed Anal. 2015;109:121–7.
Quan G, Niu B, Singh V, Zhou Y, Wu C-Y, Pan X, et al. Supersaturable solid self-microemulsifying drug delivery system: precipitation inhibition and bioavailability enhancement. Int J Nanomedicine. 2017;12:8801–11.
Xu S, Dai W-G. Drug precipitation inhibitors in supersaturable formulations. Int J Pharm. 2013;453:36–43.
Boyd BJ, Bergström CAS, Vinarov Z, Kuentz M, Brouwers J, Augustijns P, et al. Successful oral delivery of poorly water-soluble drugs both depends on the intraluminal behavior of drugs and of appropriate advanced drug delivery systems. Eur J Pharm Sci. 2019;137:104967.
Lee DR, Ho MJ, Choi YW, Kang MJ. A polyvinylpyrrolidone-based supersaturable self-emulsifying drug delivery system for enhanced dissolution of cyclosporine A. Polymers (Basel). 2017;9:124.
Gao P, Rush BD, Pfund WP, Huang T, Bauer JM, Morozowich W, et al. Development of a supersaturable SEDDS (S-SEDDS) formulation of paclitaxel with improved oral bioavailability. J Pharm Sci. 2003;92:2386–98.
Renno T, Coste-Invernizzi I, Giraud S, Lebecque S. Benzoimidazole derivatives as anticancer agents. PATENT WO/2018/054989A1. France; 2018.
Scheffe H. The simplex-centroid design for experiments with mixtures. J R Stat Soc Ser B. 1963;25:235–63.
Cornell JA. Experiments with mixtures: designs, models, and the analysis of mixture data. Third Edit. New York: Wiley & Sons; 2002.
Guerreiro F, Pontes JF, Rosa da Costa AM, Grenha A. Spray-drying of konjac glucomannan to produce microparticles for an application as antitubercular drug carriers. Powder Technol. 2019;342:246–52.
Sambuy Y, De Angelis I, Ranaldi G, Scarino ML, Stammati A, Zucco F. The Caco-2 cell line as a model of the intestinal barrier: influence of cell and culture-related factors on Caco-2 cell functional characteristics. Cell Biol Toxicol. 2005;21:1–26.
Beloqui A, des Rieux A, Préat V. Mechanisms of transport of polymeric and lipidic nanoparticles across the intestinal barrier. Adv Drug Deliv Rev. Elsevier B.V. 2016;106:242–55.
Lee Y-C, Dalton C, Regler B, Harris D. Drug solubility in fatty acids as a formulation design approach for lipid-based formulations: a technical note. Drug Dev Ind Pharm. 2018;44:1551–6.
Stella VJ. Chemical Drug Stability in Lipids, Modified Lipids, and Polyethylene Oxide-Containing Formulations. Pharm Res. 2013;30:3018–28.
Constantinides PP, Scalart J-P. Formulation and physical characterization of water-in-oil microemulsions containing long- versus medium-chain glycerides. Int J Pharm. 1997;158:57–68.
Jing B, Wang Z, Yang R, Zheng X, Zhao J, Tang S, et al. Enhanced oral bioavailability of felodipine by novel solid self-microemulsifying tablets. Drug Dev Ind Pharm. 2016;42:506–12.
Tung N-T, Tran C-S, Pham T-M-H, Nguyen H-A, Nguyen T-L, Chi S-C, et al. Development of solidified self-microemulsifying drug delivery systems containing l-tetrahydropalmatine: Design of experiment approach and bioavailability comparison. Int J Pharm. 2018;537:9–21.
Oberle RL, Moore TJ, Krummel DAP. Evaluation of mucosal damage of surfactants in rat jejunum and colon. J Pharmacol Toxicol Methods. 1995;33:75–81.
Siqueira Jørgensen SD, Rades T, Mu H, Graeser K, Müllertz A. Exploring the utility of the Chasing Principle: influence of drug-free SNEDDS composition on solubilization of carvedilol, cinnarizine and R3040 in aqueous suspension. Acta Pharm Sin B. 2019;9:194–201.
Kiss L, Walter FR, Bocsik A, Veszelka S, Ózsvári B, Puskás LG, et al. Kinetic analysis of the toxicity of pharmaceutical excipients cremophor EL and RH40 on endothelial and epithelial cells. J Pharm Sci. 2013;102:1173–81.
Ogino M, Yakushiji K, Suzuki H, Shiokawa K, Kikuchi H, Seto Y, et al. Enhanced pharmacokinetic behavior and hepatoprotective function of ginger extract-loaded supersaturable self-emulsifying drug delivery systems. J Funct Foods. 2018;40:156–63.
Kraut JA, Mullins ME. Toxic alcohols. In: Campion EW, editor. N Engl J Med. 2018;378:270–80.
Vertzoni M, Augustijns P, Grimm M, Koziolek M, Lemmens G, Parrott N, et al. Impact of regional differences along the gastrointestinal tract of healthy adults on oral drug absorption: An UNGAP review. Eur J Pharm Sci. 2019;134:153–75.
Liu Y, Wang X, Ren W, Chen Y, Yu Y, Zhang J, et al. Novel albendazole-chitosan nanoparticles for intestinal absorption enhancement and hepatic targeting improvement in rats. J Biomed Mater Res Part B Appl Biomater. 2013;101B:998–1005.
Lee J-H, Kim H, Cho Y, Koo T-S, Lee G. Development and evaluation of raloxifene-hydrochloride-loaded supersaturatable SMEDDS containing an acidifier. Pharmaceutics. 2018;10:78.
Sakloetsakun D, Dünnhaupt S, Barthelmes J, Perera G, Bernkop-Schnürch A. Combining two technologies: Multifunctional polymers and self-nanoemulsifying drug delivery system (SNEDDS) for oral insulin administration. Int J Biol Macromol. 2013;61:363–72.
Food and Drug Administration. Dimethyl Sulfoxide (DMSO) [Internet]. Available from: https://www.fda.gov/.
Parmar N, Singla N, Amin S, Kohli K. Study of cosurfactant effect on nanoemulsifying area and development of lercanidipine loaded (SNEDDS) self nanoemulsifying drug delivery system. Colloids Surf B Biointerfaces. 2011;86:327–38.
Jaisamut P, Wiwattanawongsa K, Graidist P, Sangsen Y, Wiwattanapatapee R. Enhanced oral bioavailability of curcumin using a supersaturatable self-microemulsifying system incorporating a hydrophilic polymer; in vitro and in vivo investigations. AAPS PharmSciTech AAPS PharmSciTech. 2018;19:730–40.
Li S, Madan P, Lin S. Effect of ionization of drug on drug solubilization in SMEDDS prepared using Capmul MCM and caprylic acid. Asian J Pharm Sci. 2017;12:73–82.
International Organization for Standardization. ISO 10993–1 Biological Evaluation of Medical Devices—Part, vol. 5. Geneva Switz: Tests for in Vitro Cytotoxicity; 2009.
Aktas Y, Celik Tekeli M, Celebi N. Development and characterization of exendin-4 loaded self-nanoemulsifying system and in vitro evaluation on Caco-2 cell line. J Microencapsul. 2020;37:41–51.
Beloqui A, Memvanga PB, Coco R, Reimondez-Troitiño S, Alhouayek M, Muccioli GG, et al. A comparative study of curcumin-loaded lipid-based nanocarriers in the treatment of inflammatory bowel disease. Colloids Surf B Biointerfaces. 2016;143:327–35.
Nottingham E, Sekar V, Mondal A, Safe S, Rishi AK, Singh M. The Role of Self-Nanoemulsifying Drug Delivery Systems of CDODA-Me in Sensitizing Erlotinib-Resistant Non-Small Cell Lung Cancer. J Pharm Sci. 2020;109:1867–82.
Funding
The research leading to these results has received funding from National Research Agency (ANR), HyDNano project (ANR-18-CE18-0025-01), the PHC Pessoa Programme between ANR and Fundação para a Ciência e Tecnologia (FCT): NanoSpeed, (N° 42306YB), and from FCT project UID/Multi/04326/2019.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All animal experiments were approved by the local animal ethics of University Claude Bernard Lyon 1 and carried out in compliance with current French guidelines (authorisation number 10386).
Consent for publication
All authors have been personally and actively involved in substantial work leading to the paper, will take public responsibility for its content, and given their consent for publication. All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by A.R., J.P., E.R., V.A., C.B., E.A., M.L, S.A., I.C., T.R. and S.G. Manuscript was written by A.R. and G.L. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rosso, A., Almouazen, E., Pontes, J. et al. Supersaturable self-microemulsifying delivery systems: an approach to enhance oral bioavailability of benzimidazole anticancer drugs. Drug Deliv. and Transl. Res. 11, 675–691 (2021). https://doi.org/10.1007/s13346-021-00904-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-021-00904-x