Abstract
Leukemia stem cells (LSCs) are responsible for leukemia initiation and targeting LSCs is one strategy to treat this disease. This study aims to target LSCs using multi-siRNA loaded antibodies modified with mesoporous silica nanoparticles (MSNs). Here, both siRNAGLI1 and siRNASMO were loaded in an anti-CD34 antibody modified with MSNs, and then, the MSN@siRNAGLI1@Antibody + MSNs@siRNASMO@Antibody cocktail was used to target LSCs. Expression levels of BCL-2 in LSCs were significantly reduced whereas Bax expression was significantly increased after treatment with nano-drug carriers. In addition, these nano-drug carriers also effectively induced the apoptosis of LSCs. The MSNs@siRNAGLI1@Antibody + MSNs@siRNASMO@Antibody cocktail significantly inhibited LSCs. In short, we constructed two target MSN nano-drug carriers where loaded siRNAs can be used in a chemotherapeutic drug cocktail to improve the treatment of leukemia.
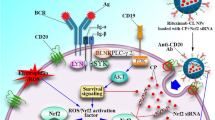
Graphical abstract







Similar content being viewed by others
Data availability
The work described has not been submitted elsewhere for publication and thereby not under simultaneous consideration by another journal.
References
Briot T, Roger E, Thepot S, Lagarce F. Advances in treatment formulations for acute myeloid leukemia. Drug Discov Today. 2018;23:1936-49.
Kubasch AS, Wehner R, Bazzurri S, Tunger A, Stasik S, Garzarolli M, et al. Clinical, molecular, and immunological responses to pembrolizumab treatment of synchronous melanoma and acute myeloid leukemia. Blood advances. 2018;2:1187–90.
Lupi SM, Rodriguez YBA, Cervino G, Todaro C, Rizzo S. Long-term effects of acute myeloid leukemia treatment on the oral system in a pediatric patient. Open Dent J. 2018;12:230–7.
Horne GA, Jackson L, Helgason V, Holyoake TL. Stem cell guardians - old and new perspectives in LSC biology. Curr Drug Targets. 2017;18:405–13.
Kim SW, Khang D. Multiple cues on the physiochemical, mesenchymal, and intracellular trafficking interactions with nanocarriers to maximize tumor target efficiency. Int J Nanomedicine. 2015;10:3989–4008.
Pelosi E, Castelli G, Testa U. Targeting LSCs through membrane antigens selectively or preferentially expressed on these cells. Blood Cells Mol Dis. 2015;55:336–46.
Chandra V, Das T, Gulati P, Biswas NK, Rote S, Chatterjee U, et al. Hedgehog signaling pathway is active in GBM with GLI1 mRNA expression showing a single continuous distribution rather than discrete high/low clusters. PLoS ONE. 2015;10:e0116390.
Gao L, Li S, Li Y. Exendin-4 promotes the osteogenic differentiation of osteoblasts via the Hedgehog/Gli1 signaling pathway. Am J Transl Res. 2018;10:315–24.
Li XW, Li JF, Qu Y, Cai Q, Ji J, Nie H, et al. Hedgehog signaling pathway activates in gastric carcinoma and promotes the proliferation through GLI1 in MKN28 cell. Zhonghua Wei Chang Wai Ke Za Zhi. 2009;12:603–6.
Shahi MH, Rey JA, Castresana JS. The sonic hedgehog-GLI1 signaling pathway in brain tumor development. Expert Opin Ther Targets. 2012;16:1227–38.
Xu X, Su B, Xie C, Wei S, Zhou Y, Liu H, et al. Sonic hedgehog-Gli1 signaling pathway regulates the epithelial mesenchymal transition (EMT) by mediating a new target gene, S100A4, in pancreatic cancer cells. PLoS ONE. 2014;9:e96441.
Courthion H, Mugnier T, Rousseaux C, Moller M, Gurny R, Gabriel D. Self-assembling polymeric nanocarriers to target inflammatory lesions in ulcerative colitis. J Control Release. 2018;275:32–9.
Jensen LB, Griger J, Naeye B, Varkouhi AK, Raemdonck K, Schiffelers R, et al. Comparison of polymeric siRNA nanocarriers in a murine LPS-activated macrophage cell line: gene silencing, toxicity and off-target gene expression. Pharm Res. 2012;29:669–82.
Li X, Qian Y, Liu T, Hu X, Zhang G, You Y, et al. Amphiphilic multiarm star block copolymer-based multifunctional unimolecular micelles for cancer targeted drug delivery and MR imaging. Biomaterials. 2011;32:6595–605.
Moghimi SM. Chemical camouflage of nanospheres with a poorly reactive surface: towards development of stealth and target-specific nanocarriers. Biochim Biophys Acta. 2002;1590:131–9.
Zheng F, Zhang P, Xi Y, Chen X, He Z, Meng T, et al. Hierarchical nanocarriers for precisely regulating the therapeutic process via dual-mode controlled drug release in target tumor cells. ACS Appl Mater Interfaces. 2017;9:36655–64.
Guo J, Gao J, Li Z, Gong Y, Man X, Jin J, et al. Adenovirus vector-mediated Gli1 siRNA induces growth inhibition and apoptosis in human pancreatic cancer with Smo-dependent or Smo-independent Hh pathway activation in vitro and in vivo. Cancer Lett. 2013;339:185–94.
Zhou Q, Wu HF, Wei Y, Zhu WL. Effect of Smo SiRNA-mediated hedgehog signaling pathway inhibition on palatal fusion. Biomed Environ Sci. 2016;29:594–8.
Hom C, Lu J, Liong M, Luo H, Li Z, Zink JI, et al. Mesoporous silica nanoparticles facilitate delivery of siRNA to shutdown signaling pathways in mammalian cells. Small. 2010;6:1185–90.
Li X, Xie QR, Zhang J, Xia W, Gu H. The packaging of siRNA within the mesoporous structure of silica nanoparticles. Biomaterials. 2011;32:9546–56.
PDQ Adult Treatment Editorial Board. Adult Acute Myeloid Leukemia Treatment (PDQ®): Patient Version. PDQ Cancer Information Summaries. Bethesda (MD); 2002.
Castejon N, Cappelleri JC, Cuervo J, Lang K, Mehta P, Mokgokong R, et al. Social preferences for health states associated with acute myeloid leukemia for patients undergoing treatment in the United Kingdom. Health Qual Life Outcomes. 2018;16:66.
Im HJ. Current treatment for pediatric acute myeloid leukemia. Blood Research. 2018;53:1–2.
Jiang X, Distinguishing CML. LSCs from HSCs using CD26. Blood. 2014;123:3851–2.
Kovacsovics TJ, Mims A, Salama ME, Pantin J, Rao N, Kosak KM, et al. Combination of the low anticoagulant heparin CX-01 with chemotherapy for the treatment of acute myeloid leukemia. Blood Adv. 2018;2:381–9.
Weisberg E, Sattler M, Manley PW, Griffin JD. Spotlight on midostaurin in the treatment of FLT3-mutated acute myeloid leukemia and systemic mastocytosis: design, development, and potential place in therapy. OncoTargets Ther. 2018;11:175–82.
Sawant RR, Sawant RM, Kale AA, Torchilin VP. The architecture of ligand attachment to nanocarriers controls their specific interaction with target cells. J Drug Target. 2008;16:596–600.
Funding
This work was supported by the National Natural Science Foundation of China (NO.81802429).
Author information
Authors and Affiliations
Contributions
Yuan Zhang: Conceptualization, Methodology, Experimental operation, Writing and Reviewing; Rui Cai, HaiJun Li, Yu Duan, Yan Zhang, Wei Jing, ShaoGang Lv, Xi Chu, Zheng Cao and Lei Yang plays equally to the Methodology, Experimental operation and Writing. Liang Ming was the mentor of the manuscript concept.
Corresponding author
Ethics declarations
Conflict of interest
All the listed authors have approved the manuscript and its submission and the authors have declared that no conflict of interest exists.
Ethical approval and consent to participate
No animal or human studies were carried out by the authors for this article.
Consent for publication
The copyright to the English-language article is transferred to Drug Delivery and Translational Research effective if and when the article is accepted for publication.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, Y., Cai, R., Li, H. et al. Construction of a target MSNs drugcarrier loaded with siRNAGLI1 and siRNASMO aim at hedgehog signal pathway and the pharmacodynamic study of drug-carriers in the treatment of leukemia stem cells. Drug Deliv. and Transl. Res. 12, 2463–2473 (2022). https://doi.org/10.1007/s13346-020-00893-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-020-00893-3