Abstract
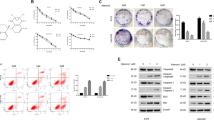
Previous evidence has shown that the increased expression of aurora kinase is closely related to melanoma progression and is an important therapeutic target in melanoma. Danusertib is an inhibitor of aurora kinase, and recent studies have shown that danusertib treatment induces autophagy in several types of cancer. Interestingly, autophagy plays a dual function in cancer as a pro-survival and anti-survival factor. In this study, we investigated the role of danusertib on the induction of autophagy in melanoma and determined the impact of autophagy induction on its anticancer activity against melanoma. Our results showed that danusertib can significantly inhibit melanoma growth by inducing cell cycle arrest and apoptosis. In addition, we demonstrated that danusertib treatment significantly inhibits the oncogenic Akt/mTOR signaling pathway and induces autophagy in melanoma cells. Furthermore, we identified that the inhibition of autophagy can enhance the inhibitory effects of danusertib on melanoma growth. Thus, the induction of autophagy by danusertib appears to be a survival mechanism in melanoma cells that may counteract its anticancer effects. These findings suggest a novel strategy to enhance the anticancer efficacy of danusertib in melanoma by blocking autophagy.









Similar content being viewed by others
References
Lo JA, Fisher DE. The melanoma revolution: from UV carcinogenesis to a new era in therapeutics. Science. 2014;346:945–9.
Ma J, Guo W, Li C. Ubiquitination in melanoma pathogenesis and treatment. Cancer Med. 2017;6:1362–77.
Chen J, Peng C, Lei L, Zhang J, Zeng W, Chen X. Nuclear envelope-distributed CD147 interacts with and inhibits the transcriptional function of RING1 and promotes melanoma cell motility. PLoS One. 2017;12:e0183689.
Jemal A, Siegel R, Ward E, Murray T, Xu J, Smigal C, et al. Cancer statistics, 2006. CA Cancer J Clin. 2006;56:106–30.
Bonet C, Giuliano S, Ohanna M, Bille K, Allegra M, Lacour JP, et al. Aurora B is regulated by the mitogen-activated protein kinase/extracellular signal-regulated kinase (MAPK/ERK) signaling pathway and is a valuable potential target in melanoma cells. J Biol Chem. 2012;287:29887–98.
Chang X, Zhang H, Lian S, Zhu W. miR-137 suppresses tumor growth of malignant melanoma by targeting aurora kinase A. Biochem Biophys Res Commun. 2016;475:251–6.
Pacchierotti F, Adler ID, Eichenlaub-Ritter U, Mailhes JB. Gender effects on the incidence of aneuploidy in mammalian germ cells. Environ Res. 2007;104:46–69.
Rong R, Jiang LY, Sheikh MS, Huang Y. Mitotic kinase Aurora-A phosphorylates RASSF1A and modulates RASSF1A-mediated microtubule interaction and M-phase cell cycle regulation. Oncogene. 2007;26:7700–8.
Dar AA, Zaika A, Piazuelo MB, Correa P, Koyama T, Belkhiri A, et al. Frequent overexpression of Aurora kinase A in upper gastrointestinal adenocarcinomas correlates with potent antiapoptotic functions. Cancer. 2008;112:1688–98.
Chefetz I, Holmberg JC, Alvero AB, Visintin I, Mor G. Inhibition of Aurora-A kinase induces cell cycle arrest in epithelial ovarian cancer stem cells by affecting NFkB pathway. Cell Cycle. 2011;10:2206–14.
Fenouille N, Tichet M, Dufies M, Pottier A, Mogha A, Soo JK, et al. The epithelial-mesenchymal transition (EMT) regulatory factor SLUG (SNAI2) is a downstream target of SPARC and AKT in promoting melanoma cell invasion. PLoS One. 2012;7:e40378.
Porcelli L, Guida G, Quatrale AE, Cocco T, Sidella L, Maida I, et al. Aurora kinase B inhibition reduces the proliferation of metastatic melanoma cells and enhances the response to chemotherapy. J Transl Med. 2015;13:26.
Liu Y, Hawkins OE, Su YJ, Vilgelm AE, Sobolik T, Thu YM, et al. Targeting aurora kinases limits tumour growth through DNA damage-mediated senescence and blockade of NF-?B impairs this drug-induced senescence. EMBO Mol Med. 2013;5:149–66.
Xie L, Meyskens FL Jr. The pan-Aurora kinase inhibitor, PHA-739358, induces apoptosis and inhibits migration in melanoma cell lines. Melanoma Res. 2013;23:102–13.
Li JP, Yang YX, Liu QL, Zhou ZW, Pan ST, He ZX, et al. The pan-inhibitor of Aurora kinases danusertib induces apoptosis and autophagy and suppresses epithelial-to-mesenchymal transition in human breast cancer cells. Drug Des Devel Ther. 2015;9:1027–62.
Yuan CX, Zhou ZW, Yang YX, He ZX, Zhang X, Wang D, et al. Danusertib, a potent pan-Aurora kinase and ABL kinase inhibitor, induces cell cycle arrest and programmed cell death and inhibits epithelial to mesenchymal transition involving the PI3K/Akt/mTOR-mediated signaling pathway in human gastric cancer AGS and NCI-N78 cells. Drug Des Devel Ther. 2015;9:1293–318.
Zi D, Zhou ZW, Yang YJ, Huang L, Zhou ZL, He SM, et al. Danusertib induces apoptosis, cell cycle arrest, and autophagy but inhibits epithelial to mesenchymal transition involving PI3K/Akt/mTOR signaling pathway in human ovarian cancer cells. Int J Mol Sci. 2015;16:27228–51.
He SJ, Shu LP, Zhou ZW, Yang T, Duan W, Zhang X, et al. Inhibition of Aurora kinases induces apoptosis and autophagy via AURKB/p70S6K/RPL15 axis in human leukemia cells. Cancer Lett. 2016;382:215–30.
Levine B, Kroemer G. Autophagy in the pathogenesis of disease. Cell. 2008;132:27–42.
Mizushima N, Levine B, Cuervo AM, Klionsky DJ. Autophagy fights disease through cellular self-digestion. Nature. 2008;451:1069–75.
Dhawan P, Singh AB, Ellis DL, Richmond A. Constitutive activation of Akt/protein kinase B in melanoma leads to up-regulation of nuclear factor-kappaB and tumor progression. Cancer Res. 2002;62:7335–42.
Levine B, Yuan J. Autophagy in cell death: an innocent convict? J Clin Invest. 2005;115:2679–88.
Caporali S, Alvino E, Lacal PM, Levati L, Giurato G, Memoli D, et al. Targeting the PI3K/AKT/mTOR pathway overcomes the stimulating effect of dabrafenib on the invasive behavior of melanoma cells with acquired resistance to the BRAF inhibitor. Int J Oncol. 2016;49:1164–74.
Acknowledgments
The authors acknowledge the First-Class Discipline Construction Founded Project of Ningxia Medical University (NXYLXK2017A05) and the School of Clinical Medicine and Ningxia Medical University 2018 university-level scientific research project (XM2018152).
Author information
Authors and Affiliations
Contributions
Yuan-Yuan Shang and Weichao Li conceived and designed this study. Yuan-Yuan Shang and Nan Yu Xu were responsible for doing the main experimental. Li Xia, Ying-Yao Yu, Chun-mei Ma, and Ya-Ning Jiao were responsible for construction of mice model. Yuan-Yuan Shang, Yun-feng Li, and Jie Dang were jointly involving in extracting data and writing the manuscript.
Corresponding author
Ethics declarations
This research was approved by the Research Ethics Board of the General Hospital of Ningxia Medical University.
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shang, YY., Yu, N., Xia, L. et al. Augmentation of danusertib’s anticancer activity against melanoma by blockage of autophagy. Drug Deliv. and Transl. Res. 10, 136–145 (2020). https://doi.org/10.1007/s13346-019-00668-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-019-00668-5