Abstract
Since fulminant type 1 diabetes was reported as a distinct subtype of type 1 diabetes in 2000, the Committee on Type 1 diabetes, Japan Diabetes Society has continuously recruited patients and conducted genomic research to elucidate the genetic basis of fulminant type 1 diabetes. The contribution of the human leukocyte antigen complex (HLA) to genetic susceptibility to fulminant type 1 diabetes was compared with that of other subtypes in 2009. The alleles and haplotypes associated with fulminant type 1 diabetes were found to be different from acute-onset and slowly progressive type 1 diabetes. DRB1*15:01-DQB1*06:02, a protective haplotype against acute-onset type 1 diabetes, does not provide protection against fulminant type 1 diabetes and DRB1*08:02-DQB1*03:02, a susceptible haplotype to acute-onset type 1 diabetes, does not confer susceptibility to fulminant type 1 diabetes. Recently, the first genome-wide association study (GWAS) of fulminant type 1 diabetes was performed in Japanese individuals. A strong association was observed with multiple single nucleotide polymorphisms (SNPs) in the HLA region, and the strongest association was observed with rs9268853 in the class II DR region. In addition, 11 SNPs outside the HLA region showed some evidence of association with the disease. In particular, rs11170445 in CSAD/lnc-ITGB7-1 on chromosome 12q13.13 showed an association at a genome-wide significance level. Fine mapping revealed that rs3782151 in CSAD/lnc-ITGB7-1 showed the lowest P value. CSAD/lnc-ITGB7-1 was found to be strongly associated with susceptibility to fulminant, but not classical, autoimmune type 1 diabetes, implicating this locus in the distinct phenotype of fulminant type 1 diabetes.






Similar content being viewed by others
References
Imagawa A, Hanafusa T, Miyagawa J, Matsuzawa Y. A novel subtype of type 1 diabetes mellitus characterized by a rapid onset and an absence of diabetes related antibodies. Osaka IDDM study group. N Engl J Med. 2000;342(5):301–7.
Imagawa A, Hanafusa T. Fulminant type 1 diabetes: a novel clinical entity requiring special attention by all medical practitioners. Nat Clin Prac Endocrinol Metab. 2007;3:36–45.
Gianani R, Eisenbarth GS. The stages of type 1A diabetes: 2005. Immunol Rev. 2005;204:232–49.
Bakay M, Pandey R, Grant SFA, Hakonarson H. The genetic contribution to type 1 diabetes. Curr Diab Rep. 2019;19:116.
Karvonen M, Viik-Kajander M, Moltchanova E, Libman I, LaPorte R, Tuomilehto J. Incidence of childhood type 1 diabetes worldwide. Diabetes mondiale (DiaMond) project group. Diabetes Care. 2000;23:1516–26.
Ikegami H, Awata T, Kawasaki E, Kobayashi T, Maruyama T, Nakanishi K, Shimada A, Amemiya S, Kawabata Y, Kurihara S, Tanaka S, Kanazawa Y, Mochizuki M, Ogihara T. Japanese study group on type 1 diabetes genetics: the association of CTLA4 polymorphism with type 1 diabetes is concentrated in patients complicated with autoimmune thyroid disease: a multi-center collaborative study in Japan. J Clin Endocrinol Metab. 2006;91:1087–92.
Ikegami H, Kawabata Y, Noso S, Fujisawa T, Ogihara T. Genetics of type 1 diabetes in Asian and Caucasian populations. Diabetes Res Clin Pract. 2007;77(Suppl. 1):S116–S12121.
Cho YM, Kim JT, Ko KS, Koo BK, Yang SW, Park MH, Lee HK, Park KS. Fulminant type 1 diabetes in Korea: high prevalence among patients with adult-onset type 1 diabetes. Diabetologia. 2007;50:2276–9.
Moreau C, Drui D, Arnault-Ouary G, Charbonnel B, Chaillous L, Cariou B. Fulminant type 1 diabetes in Caucasians: a report of three cases. Diabetes Metab. 2008;34:529–32.
Rish N. Assessing the role of HLA-linked and unlinked determinants of disease. Am J Hum Genet. 1987;40:1–14.
Ikegami H, Oihara T. Genetics of insulin-dependent diabetes mellitus. Endocrine J. 1996;43:605–13.
Noble JA, Valdes AM, Cook M, Klitz W, Thomson G, Erlich HA. The role of HLA class II genes in insulin-dependent diabetes mellitus: molecular analysis of 180 Caucasian, multiplex families. Am J Hum Genet. 1996;59:1134–48.
Lambert AP, Gillespie KM, Thomson G, Cordell HJ, Todd JA, Gale EA, Bingley PJ. Absolute risk of childhood-onset type 1 diabetes defined by human leukocyte antigen class II genotype: a population-based study in the United Kingdom. J Clin Endocrinol Metab. 2004;89:4037–43.
Todd J, Bell JI, MacDevitt HO. HLA-DQ beta gene contributes to susceptibility and resistance to insulin-dependent diabetes mellitus. Nature. 1987;329:599–604.
Ikegami H, Kawaguchi Y, Yamato E, Kuwata S, Tokunaga K, Noma Y, Shima K, Ogiharaet T. Analysis by the polymerase chain reaction of histocompatibility leukocyte antigen-DR9-linked susceptibility to insulin-dependent diabetes mellitus. J Clin Endocrinol Metab. 1992;75:1381–5.
Kawabata Y, Ikegami H, Kawaguchi Y, Fujisawa T, Shintani M, Ono M, Nishino M, Uchigata Y, Lee I, Ogihara T. Asian-specific HLA haplotypes reveal heterogeneity of the contribution of HLA-DR and -DQ haplotypes to susceptibility to type 1 diabetes. Diabetes. 2002;51:545–51.
Thomson G, Valdes AM, Noble JA, et al. Relative predispositional effects of HLA class II DRB1-DQB1 haplotypes and genotypes on type 1 diabetes: a meta-analysis. Tissue Antigens. 2007;70:110–27.
Ikegami H, Noso S, Babaya N, Hiromine Y, Kawabata Y. Genetic basis of type 1 diabetes: similarities and differences between East and West. Rev Diabet Stud. 2008;5:64–72.
Kawabata Y, Ikegami H, Awata T, Imagawa A, Maruyama T, Kawasaki E, Tanaka S, Shimada A, Osawa H, Kobayashi T, Hanafusa T, Tokunaga K, Makino H. Committee on type 1 diabetes, Japan diabetes society. Diabetologia. 2009;52:2513–21.
Tsutsumi C, Imagawa A, Ikegami H, Makino H, Kobayashi T, Hanafusa T. Japan diabetes society committee on type 1 diabetes mellitus research. Class II HLA genotype in fulminant type 1 diabetes: a nationwide survey with reference to glutamic acid decarboxylase antibodies. J Diabetes Investig. 2012;3:62–9.
Imagawa A, Hanafusa T. Fulminant type 1 diabetes–an important subtype in East Asia. Diabetes Metab Res Rev. 2011;27:959–64.
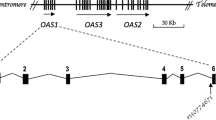
Kawabata Y, Nishida N, Awata T, Kawasaki E, Imagawa A, Shimada A, Osawa H, Tanaka S, Takahashi K, Nagata M, Yasuda H, Uchigata Y, Kajio H, Makino H, Yasuda K, Kobayashi T, Hanafusa T, Tokunaga K, Ikegami H. Genome-wide association study confirming a strong effect of HLA and identifying variants in CSAD/lnc-ITGB7–1 on chromosome 12q13.13 associated with susceptibility to fulminant type 1 diabetes. Diabetes. 2019;68:665–75.
Evangelou M, Smyth DJ, Fortune MD, Burren OS, Walker NM, Guo H, Onengut-Gumuscu S, Chen W-M, Concannon P, Rich SR, Todd JA, Wallace C. A method for gene-based pathway analysis using genome wide association study summary statistics reveals nine new type 1 diabetes associations. Genet Epidemiol. 2014;38:661–70.
Sirdah MM. Protective and therapeutic effectiveness of taurine in diabetes mellitus: a rationale for antioxidant supplementation. Diabetes Metab Syndr. 2015;9:55–64.
Lambert IH, Kristensen DM, Holm JB, Mortensen OH. Physiological role of taurine–from organism to organelle. Acta Physiol (Oxf). 2015;213:191–21212.
Huxtable RJ. Physiological actions of taurine. Physiol Rev. 1992;72:101–63.
Marcinkiewicz J, Kontny E. Taurine and inflammatory diseases. Amino Acids. 2014;46:7–20.
Kim C, Cha Y-N. Taurine chloramine produced from taurine under inflammation provides anti-inflammatory and cytoprotective effects. Amino Acids. 2014;46:89–100.
Fakruddin M, Wei F, Suzuki T, et al. Defective mitochondrial tRNA taurine modification activates global proteostress and leads to mitochondrial disease. Cell Rep. 2018;22:482–96.
Ohsawa Y, Hagiwara H, Nishimatsu S, et al. Taurine supplementation for prevention of stroke-like episodes in MELAS: a multicenter, open-label, 52-week phase III trial. J Neurol Neurosurg Psychiatry. 2019;90:529–36.
Arany E, Strutt B, Romanus P, Remacle C, Reusens B, Hill DJ. Taurine supplement in early life altered islet morphology, decreased insulitis and delayed the onset of diabetes in non-obese diabetic mice. Diabetologia. 2004;47:1831–7.
Lin S, Yang J, Wu G, Liu M, Lv Q, Yang Q, Hu J. Inhibitory effects of taurine on STZ-induced apoptosis of pancreatic islet cells. Adv Exp Med Biol. 2013;775:287–97.
Volders PJ, Verheggen K, Menschaert G, Vandepoele K, Martens L, Vandesompele J, Mestdagh P. An update on LNCipedia: a database for annotated human lncRNA sequences. Nucleic Acids Res. 2015;43:D174–D180180.
Westra HJ, Peters MJ, Esko T, et al. Systematic identification of trans eQTLs as putative drivers of known disease associations. Nat Genet. 2013;45:1238–43.
Narahara M, Higasa K, Nakamura S, Tabara Y, Kawaguchi T, Ishii M, Matsubara K, Matsuda F, Yamada R. Large-scale East-Asian eQTL mapping reveals novel candidate genes for LD mapping and the genomic landscape of transcriptional effects of sequence variants. PLoS ONE. 2014;9:e100924.
Higasa K, Miyake N, Yoshimura J, et al. Human genetic variation database, a reference database of genetic variations in the Japanese population. J Hum Genet. 2016;61:547–53.
Berlin C, Berg EL, Briskin MJ, Andrew DP, Kilshaw PJ, Holzmann B, Weissman IL, Hamann A, Butcher EC. Alpha 4 beta 7 integrin mediates lymphocyte binding to the mucosal vascular addressin MAdCAM-1. Cell. 1993;74:185–95.
Streeter PR, Berg EL, Rouse BT, Bargatze RF, Butcher EC. A tissue-specific endothelial cell molecule involved in lymphocyte homing. Nature. 1988;331:41–6.
Hänninen A, Taylor C, Streeter PR, Stark LS, Sarte JM, Shizuru JA, Simell O, Michie SA. Vascular addressins are induced on islet vessels during insulitis in nonobese diabetic mice and are involved in lymphoid cell binding to islet endothelium. J Clin Invest. 1993;92:2509–15.
Faveeuw C, Gagnerault MC, Lepault F. Expression of homing and adhesion molecules in infiltrated islets of Langerhans and salivary glands of nonobese diabetic mice. J Immunol. 1994;152:5969–78.
Yang XD, Sytwu HK, McDevitt HO, Michie SA. Involvement of beta 7 integrin and mucosal addressin cell adhesion molecule-1 (MAdCAM-1) in the development of diabetes in obese diabetic mice. Diabetes. 1997;46:1542–7.
Acknowledgements
The authors thank all of the participants in the project, the members of the Study Group on Type 1 Diabetes Genetics and the Committee on Type 1 diabetes, Japan Diabetes Society.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial conflicts of interest to disclose for this manuscript.
Ethics policy
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Kawabata, Y., Ikegami, H. Genetics of fulminant type 1 diabetes. Diabetol Int 11, 315–322 (2020). https://doi.org/10.1007/s13340-020-00468-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13340-020-00468-0