Abstract
Aim
We investigated the association of visceral adiposity with glycated albumin (GA) as well as GA/hemoglobin A1c (HbA1c) in type 2 diabetes.
Methods
One hundred twenty-three patients (68 males, 55 females) with type 2 diabetes were enrolled in this cross-sectional study. Visceral fat area (VFA) was determined using an abdominal dual bioelectrical impedance analysis (dual BIA) instrument. The relationship of VFA with GA and GA/HbA1c was analyzed.
Results
Simple regression analysis showed that BMI was inversely correlated with GA as well as GA/HbA1c, but not with HbA1c, while VFA had a significant correlation with GA and GA/HbA1c. Furthermore, multiple regression analysis revealed VFA as an independent contributor to GA/HbA1c. These results suggest that visceral adiposity is a primary factor associated with GA and HbA1c level discrepancy in patients with type 2 diabetes.
Conclusions
GA is a useful indicator for glycemic control, while visceral obesity should also be taken into consideration in type 2 diabetes cases.



Similar content being viewed by others
References
Guthrow CE, Morris MA, Day JF, Thorpe SR, Baynes JW. Enhanced nonenzymatic glucosylation of human serum albumin in diabetes mellitus. Proc Natl Acad Sci USA. 1979;76:4258–61.
Kohzuma T, Koga M. Lucica GA-L glycated albumin assay kit: a new diagnostic test for diabetes mellitus. Mol Diagn Ther. 2010;14:49–51.
Koenig RJ, Peterson CM, Jones RL, Saudek C, Lehrman M, Cerami A. Correlation of glucose regulation and hemoglobin AIc in diabetes mellitus. N Engl J Med. 1976;295:417–20.
Shima K, Ito N, Abe F, Hirota M, Yano M, Yamamoto Y, et al. High-performance liquid chromatographic assay of serum glycated albumin. Diabetologia. 1988;31:627–31.
Jeffcoate SL. Diabetes control and complications: the role of glycated haemoglobin, 25 years on. Diabetes Med. 2004;21:657–65.
Sakuma N. Converse contributions of fasting and postprandial glucose to HbA1c and glycated albumin. Diabetol Int. 2011;2:167–72.
Hiramatsu Y, Shimizu I, Omori Y, Nakabayashi M, Group JGAS. Determination of reference intervals of glycated albumin and hemoglobin A1c in healthy pregnant Japanese women and analysis of their time courses and influencing factors during pregnancy. Endocr J. 2012;59:145–51.
Inaba M, Okuno S, Kumeda Y, Yamada S, Imanishi Y, Tabata T, et al. Glycated albumin is a better glycemic indicator than glycated hemoglobin values in hemodialysis patients with diabetes: effect of anemia and erythropoietin injection. J Am Soc Nephrol. 2007;18:896–903.
Austin GE, Mullins RH, Morin LG. Non-enzymic glycation of individual plasma proteins in normoglycemic and hyperglycemic patients. Clin Chem. 1987;33:2220–4.
Schleicher ED, Olgemoller B, Wiedenmann E, Gerbitz KD. Specific glycation of albumin depends on its half-life. Clin Chem. 1993;39:625–8.
Koga M, Murai J, Saito H, Matsumoto S, Kasayama S. Effects of thyroid hormone on serum glycated albumin levels: study on non-diabetic subjects. Diabetes Res Clin Pract. 2009;84:163–7.
Okada T, Nakao T, Matsumoto H, Nagaoka Y, Tomaru R, Iwasawa H, et al. Influence of proteinuria on glycated albumin values in diabetic patients with chronic kidney disease. Intern Med. 2011;50:23–9.
Koga M, Kasayama S, Kanehara H, Bando Y. CLD (chronic liver diseases)-HbA1C as a suitable indicator for estimation of mean plasma glucose in patients with chronic liver diseases. Diabetes Res Clin Pract. 2008;81:258–62.
Nishimura R, Kanda A, Sano H, Matsudaira T, Miyashita Y, Morimoto A, et al. Glycated albumin is low in obese, non-diabetic children. Diabetes Res Clin Pract. 2006;71:334–8.
Miyashita Y, Nishimura R, Morimoto A, Matsudaira T, Sano H, Tajima N. Glycated albumin is low in obese, type 2 diabetic patients. Diabetes Res Clin Pract. 2007;78:51–5.
Koga M, Matsumoto S, Saito H, Kasayama S. Body mass index negatively influences glycated albumin, but not glycated hemoglobin, in diabetic patients. Endocr J. 2006;53:387–91.
Koga M, Otsuki M, Matsumoto S, Saito H, Mukai M, Kasayama S. Negative association of obesity and its related chronic inflammation with serum glycated albumin but not glycated hemoglobin levels. Clin Chim Acta. 2007;378:48–52.
Smalley KJ, Knerr AN, Kendrick ZV, Colliver JA, Owen OE. Reassessment of body mass indices. Am J Clin Nutr. 1990;52:405–8.
Frankenfield DC, Rowe WA, Cooney RN, Smith JS, Becker D. Limits of body mass index to detect obesity and predict body composition. Nutrition. 2001;17:26–30.
Roubenoff R, Dallal GE, Wilson PW. Predicting body fatness: the body mass index vs estimation by bioelectrical impedance. Am J Public Health. 1995;85:726–8.
Segal KR, Dunaif A, Gutin B, Albu J, Nyman A, Pi-Sunyer FX. Body composition, not body weight, is related to cardiovascular disease risk factors and sex hormone levels in men. J Clin Invest. 1987;80:1050–5.
Kobayashi H, Nakamura T, Miyaoka K, Nishida M, Funahashi T, Yamashita S, et al. Visceral fat accumulation contributes to insulin resistance, small-sized low-density lipoprotein, and progression of coronary artery disease in middle-aged non-obese Japanese men. Jpn Circ J. 2001;65:193–9.
Kim K, Valentine RJ, Shin Y, Gong K. Associations of visceral adiposity and exercise participation with C-reactive protein, insulin resistance, and endothelial dysfunction in Korean healthy adults. Metabolism. 2008;57:1181–9.
Wang F, Ma X, Hao Y, Yang R, Ni J, Xiao Y, et al. Serum glycated albumin is inversely influenced by fat mass and visceral adipose tissue in Chinese with normal glucose tolerance. PLoS One. 2012;7:e51098.
American Diabetes A. Standards of medical care in diabetes—2014. Diabetes Care. 2014;37(Suppl 1):S14–80.
Yamazaki Y, Emoto M, Morioka T, Kawano N, Lee E, Urata H, et al. Clinical impact of the leptin to soluble leptin receptor ratio on subclinical carotid atherosclerosis in patients with type 2 diabetes. J Atheroscler Thromb. 2013;20:186–94.
Matsuo S, Imai E, Horio M, Yasuda Y, Tomita K, Nitta K, et al. Revised equations for estimated GFR from serum creatinine in Japan. Am J Kidney Dis. 2009;53:982–92.
Demura S, Sato S, Kitabayashi T. Percentage of total body fat as estimated by three automatic bioelectrical impedance analyzers. J Physiol Anthropol Appl Hum Sci. 2004;23:93–9.
Ida M, Hirata M, Odori S, Mori E, Kondo E, Fujikura J, et al. Early changes of abdominal adiposity detected with weekly dual bioelectrical impedance analysis during calorie restriction. Obesity (Silver Spring). 2013;21:E350–3.
Yamakage H, Ito R, Tochiya M, Muranaka K, Tanaka M, Matsuo Y, et al. The utility of dual bioelectrical impedance analysis in detecting intra-abdominal fat area in obese patients during weight reduction therapy in comparison with waist circumference and abdominal CT. Endocr J. 2014;61:807–19.
Morigami H, Morioka T, Yamazaki Y, Imamura S, Numaguchi R, Asada M, et al. Visceral adiposity is preferentially associated with vascular stiffness rather than thickness in men with type 2 diabetes. J Atheroscler Thromb. 2016;23:1067–79.
Inoue K, Tsujimoto T, Yamamoto-Honda R, Goto A, Kishimoto M, Noto H, et al. A newer conversion equation for the correlation between HbA1c and glycated albumin. Endocr J. 2014;61:553–60.
Salas-Salvado J, Bullo M, Garcia-Lorda P, Figueredo R, Del Castillo D, Bonada A, et al. Subcutaneous adipose tissue cytokine production is not responsible for the restoration of systemic inflammation markers during weight loss. Int J Obes (Lond). 2006;30:1714–20.
Alexopoulos N, Katritsis D, Raggi P. Visceral adipose tissue as a source of inflammation and promoter of atherosclerosis. Atherosclerosis. 2014;233:104–12.
Bertin E, Nguyen P, Guenounou M, Durlach V, Potron G, Leutenegger M. Plasma levels of tumor necrosis factor-alpha (TNF-alpha) are essentially dependent on visceral fat amount in type 2 diabetic patients. Diabetes Metab. 2000;26:178–82.
Kumagai S, Kishimoto H, Zou B. The leptin to adiponectin ratio is a good biomarker for the prevalence of metabolic syndrome, dependent on visceral fat accumulation and endurance fitness in obese patients with diabetes mellitus. Metab Syndr Relat Disord. 2005;3:85–94.
Tessari P, Kiwanuka E, Millioni R, Vettore M, Puricelli L, Zanetti M, et al. Albumin and fibrinogen synthesis and insulin effect in type 2 diabetic patients with normoalbuminuria. Diabetes Care. 2006;29:323–8.
Acknowledgements
We thank the patients for their participation in this study. Additionally, we are grateful for the staff of Osaka City University Hospital for their help with recruiting the patients, and collecting and recording clinical information.
Author information
Authors and Affiliations
Contributions
SM performed all of the processes involved in this study. KM managed all of the processes involved in this study, and wrote and edited the drafts of the manuscript. MI, ME, and KM conceived the study, and participated in its design and coordination. TH, YK, YY, TM, KM, SF, AS, TS, and ME recruited the patients. All co-authors contributed to discussions related to and were involved in drafting or critically revising the manuscript. Each author has read the final version of the manuscript and approved its submission for publishing.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical approval
All procedures employed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national), and with the Helsinki Declaration of 1964 and later versions.
Informed consent
Written informed consent was obtained from all subjects. The ethical review board of Osaka City University Graduate School of Medicine approved the study protocol (Approval date: not applicable. no. 308).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
13340_2020_431_MOESM1_ESM.pptx
Correlation between VFA and, GA (a)(b) or GA/HbAb1c (c)(d) separately in males and females. Correlation of VFA with (a) GA in male, (b) GA in female, (c) GA/HbA1c in male, and (d) GA/HbA1c in female (PPTX 72 kb)
13340_2020_431_MOESM2_ESM.pptx
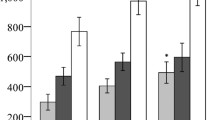
Comparison analysis between actual measured HbA1c and that calculated with a formula using GA. Non-obese (a) BMI <25 kg/m2, (c) VFA <100 cm2, and obese (b) BMI ≥25 kg/m2, (d) VFA ≥100 cm2, patients. Data are presented as mean with SD (PPTX 60 kb)
13340_2020_431_MOESM3_ESM.pptx
Cumulative response curve of VFA. A cut-off value for prediction of a -2SD of calculated HbA1c to measured HbA1c was shown to be 121 cm2 (PPTX 64 kb)
About this article
Cite this article
Maruo, S., Motoyama, K., Hirota, T. et al. Visceral adiposity is associated with the discrepancy between glycated albumin and HbA1c in type 2 diabetes. Diabetol Int 11, 368–375 (2020). https://doi.org/10.1007/s13340-020-00431-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13340-020-00431-z