Abstract
Aim
The accuracy of HbA1c values measured by use of a new portable device (A1CNow+) was evaluated by comparison with values measured by high-performance liquid chromatography (HPLC).
Materials and methods
Venous blood samples were obtained from 144 subjects (111 diabetic and 33 non-diabetic subjects). HbA1c was measured by use of both the new device and HPLC for the same blood samples. The linearity of the results and the correlation between the two methods were evaluated.
Results
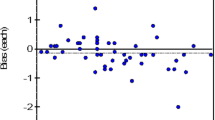
HbA1c measured by HPLC (HbA1c-HPLC) ranged between 4.4 and 12.3 % whereas values measured by the new device (HbA1c-new device) ranged between 4.5 and 12.7 %. There was a strong positive correlation between HbA1c-HPLC (Y) and HbA1c-new device (X) (Y = 0.951X + 0.197, r = 0.989; p < 0.001; 95 % confidence interval of the slope 0.928–0.975; 95 % confidence interval of the intercept 0.015–0.379).
Conclusion
HbA1c results obtained by use of the new device correlated strongly with those measured by HPLC, indicating the new device could be useful for clinical use.



Similar content being viewed by others
Abbreviations
- NGSP:
-
National Glycohemoglobin Standardization Program
- HPLC:
-
High-performance liquid chromatography
- JDS:
-
Japan Diabetes Society
- POCT:
-
Point-of-care test
References
Sacks DB, Bruns DE, Goldstein DE, et al. Guideline and recommendations for laboratory analysis and management of diabetes mellitus. Clin Chem. 2002;48:436–72.
American Diabetes Association. Standards of medical care in diabetes. Diabetes Care. 2004;27(Suppl 1):S15–35.
DCCT Research group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin dependent diabetes mellitus. New Engl J Med. 1993;329:977–86.
Ohkubo Y, Kishikawa H, Araki E, et al. Intensive insulin therapy prevents the progression of diabetic microvascular complications in Japanese patients with non-insulin-dependent diabetes mellitus: a randomized prospective 6-year study. Diabetes Res Clin Pract. 1995;28:103–17.
UK Prospective Diabetes Study (UKPDS) Group. Intensive blood glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352:837–53.
Little RR, Rohlfing CL, Wiedmeyer HM, et al. The National Glycohemoglobin Standardization Program: a five-year progress report. Clin Chem. 2001;47:1985–92.
The International Expert Committee. International expert committee report on the role of the A1C assay in the diagnosis of diabetes. Diabetes Care. 2009;32:1–8.
The Committee of the Japan Diabetes Society on the Diagnostic Criteria of Diabetes Mellitus. Report of the Committee on the Classification and Diagnostic Criteria of Diabetes Mellitus. J Diabetes Invest 2010;1: 212–28.
Goldstein DE, Little RR, Wiedmeyer HM, et al. Glycated hemoglobin: methodologies and clinical applications. Clin Chem 1986; 32(suppl.):B64–B70.
Little RR, Wiedmeyer HM, England JD, et al. Interlaboratory standardization of measurements of glycohemoglobins. Clin Chem. 1992;38:2472–8.
Shima K, Endo J, Oimomi M, et al. Interlaboratory difference in HbA1c measurement in Japan—a report of the committee on an interlaboratory standardization of HbA1c, determination, the Japan Diabetes Society. J Japan Diabetes Soc. 1994;37:855–64.
Tominaga M, Makino H, Yoshino G, et al. Report of the committee on standardization of laboratory testing related to diabetes mellitus of the Japan Diabetes Society: determination of reference intervals of hemoglobin A1c (IFCC) and glycoalbumin in the Japanese population. J Japan Diabetes Soc. 2006;49:825–33.
Vogeser M, Parhofer KG. Head-to-head comparison of an automated immunometric and an automated HPLC method for the quantification of HbA1c. Clin Biochem. 2008;41:1410–2.
Tran DV, Lyon AW, Higgins TN, et al. Use of serial patient hemoglobin A1c differences to determine long-term imprecision of immunoassay and high-performance liquid chromatography analyzers. J Diabetes Sci Technol. 2009;3:424–8.
Bode BW, Irvin BR, Pierce JA, et al. Advances in hemoglobin A1c point of care technology. J Diabetes Sci Technol. 2007;1:405–11.
Leal S, Soto-Rowen M. Usefulness of point-of-care testing in the treatment of diabetes in an underserved population. J Diabetes Sci Technol. 2009;3:672–6.
Bayer diabetes care, A1CNow. http://a1cnow.com.
Gremmels HD, Richter A, Watzke I. Evaluation of the hemoglobin A1c-analyzer TOSOH HLC-723 G7. Clin Lab. 2003;49:243–50.
Kashiwagi A, Kasuga M, Araki E, et al. Committee on the Standardization of Diabetes Mellitus-Related Laboratory Testing of the Japan Diabetes Society. International clinical harmonization of glycated hemoglobin in Japan: From Japan Diabetes Society to National Glycohemoglobin Standardization Program values. Diabetol Int. 2012;3:8–10.
Kashiwagi A, Kasuga M, Araki E, et al. Committee on the Standardization of Diabetes Mellitus-Related Laboratory Testing of the Japan Diabetes Society. International clinical harmonization of glycated hemoglobin in Japan. International clinical harmonization of glycated hemoglobin in Japan: From Japan Diabetes Society to National Glycohemoglobin Standardization Program values. J Diabetes Invest. 2012;3:39–40.
Little RR, Roberts WL. A review of variant hemoglobins interfering with hemoglobin A1c measurement. J Diabetes Sci Technol. 2009;3:446–51.
Cagliero E, Levina EV, Nathan DM. Immediate feedback of HbA1c levels improves glycemic control in type 1 and insulin-treated type 2 diabetic patients. Diabetes Care. 1999;22:1785–9.
Ferenczi A, Reddy K, Lorber DL. Effect of immediate hemoglobin A1c results on treatment decisions in office practice. Endocr Pract. 2001;7:85–8.
Schwartz KL, Monsur JC, Bartoces MG, et al. Correlation of same-visit HbA1c test with laboratory-based measurements: a MetroNet study. BMC Fam Pract. 2005;6:28.
Lenters-Westra E, Slingerland RJ. Six of eight hemoglobin A1c point-of-care instruments do not meet the general accepted analytical performance criteria. Clin Chem. 2010;55:44–52.
Acknowledgments
A1CNow+® units were kindly provided by Bayer HealthCare LLC (New York, USA).
Conflict of interest
The authors declare that A1CNow+® units were provided by Bayer HealthCare LLC (New York, USA).
Author information
Authors and Affiliations
Corresponding author
Additional information
S. Shimoda and T. Maeda contributed equally to this work.
About this article
Cite this article
Shimoda, S., Maeda, T., Furukawa, N. et al. Evaluation of a new device for measurement of hemoglobin A1c for Japanese subjects. Diabetol Int 4, 112–116 (2013). https://doi.org/10.1007/s13340-012-0102-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13340-012-0102-y