Abstract
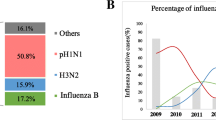
Influenza A viruses are highly adaptable and are the main pathogen behind winter time morbidity. The present study reports the molecular and phylogenetic characterization of A(H1N1)pdm09 and H3N2 isolates from Haryana, India during 2015 influenza outbreak. A total of 144 nasopharyngeal samples were collected from Post Graduate Institute of Medical Sciences, Rohtak, Haryana, India form September 2014 to February 2016. The samples were screened for influenza A subtypes; A(H1N1)pdm09 and H3N2 by using real-time RT-PCR. Virus isolation and hemagglutinin gene sequencing studies were performed for selected positive samples. Out of 24 (16.6%) Influenza A positive samples, 13 (54.2%) and 11 (45.8%) were subtyped into A(H1N1)pdm09 and H3N2, respectively by real-time RT-PCR. Genetic analysis of A(H1N1)pdm09 isolates revealed the presence of key mutations (P100S, S202T and S220T) in HA gene as compare to reference strain A/California/07/2009 and these isolates were grouped in clade 6B.1 and 6B.2. All A(H3N2) isolates were clustered in clade 3C.2a and revealed specific amino acid substitutions of N161S and P214S in their HA genes in comparison to the reference strain A/Texas/50/2012. The HA gene sequences of all isolates showed 97–98% of nucleotide sequence similarity with their respective reference strains. Influenza A(H1N1)pdm09 and H3N2 isolates were drifted significantly from their respective vaccines strains of 2015–2016 and were more closely related to recommended vaccine strains for flu season 2017–2018. The study supports the need of routine influenza surveillance and continuous monitoring of the genetic changes in the major antigenic sites of these viruses.


Similar content being viewed by others
References
Alonso WJ, Yu C, Viboud C, Richard SA, Schuck-Paim C, Simonsen L, Mello WA, Miller MA. A global map of hemispheric influenza vaccine recommendations based on local patterns of viral circulation. Sci Rep. 2015;5:17214. https://doi.org/10.1038/srep17214.
Caton AJ, Brownlee GG, Yewdell JW, Gerhard W. The antigenic structure of the influenza virus A/PR/8/34 hemagglutinin (H1 subtype). Cell. 1982;31:417–27. https://doi.org/10.1016/0092-8674(82)90135-0.
Clark AM, DeDiego ML, Anderson CS, Wang J, Yang H, Nogales A, et al. Antigenicity of the 2015–2016 seasonal H1N1 human influenza virus HA and NA proteins. PLoS ONE. 2017;12:e0188267. https://doi.org/10.1371/journal.pone.0188267.
Connor RJ, Kawaoka Y, Webster RG, Paulson JC. Receptor specificity in human, avian and equine H2 and H3 influenza virus isolates. Virology. 1994;205:17–23. https://doi.org/10.1006/viro.1994.1615.
Edgar RC. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucl Acids Res. 2004;32:1792–7. https://doi.org/10.1093/nar/gkh340.
Eisfeld AJ, Neumann G, Kawaoka Y. Influenza A virus isolation, culture and identification. Nat Protoc. 2014;9:2663–81. https://doi.org/10.1038/nprot.2014.180.
Elderfield RA, Watson SJ, Godlee A, Adamson WE, Thompson CI, Dunning J, et al. Accumulation of human-adapting mutations during circulation of A(H1N1)pdm09 influenza virus in humans in the United Kingdom. J Virol. 2014;88:13269–83. https://doi.org/10.1128/JVI.01636-14.
Guillebaud J, Héraud JM, Razanajatovo NH, Livinski AA, Alonso WJ. Both hemispheric influenza vaccine recommendations would have missed near half of the circulating viruses in Madagascar. Influenza Other Respir Viruses. 2017;11:473–8. https://doi.org/10.1111/irv.12517.
Katoh K, Standley DM. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol. 2013;30:772–80. https://doi.org/10.1093/molbev/mst010.
Matrosovich M, Tuzikov A, Bovin N, Gambaryan A, Klimov A, Castrucci MR, Donatelli I, Kawaoka Y. Early alterations of the receptor-binding properties of H1, H2, and H3 avian influenza virus hemagglutinins after their introduction into mammals. J Virol. 2000;74:8502–12. https://doi.org/10.1128/JVI.74.18.8502-8512.2000.
Matrosovich MN, Matrosovich TY, Gray T, Roberts NA, Klenk HD. Human and avian influenza viruses target different cell types in cultures of human airway epithelium. Proc Natl Acad Sci USA. 2004;101:4620–4. https://doi.org/10.1073/pnas.0308001101.
Mukherjee A, Nayak MK, Dutta S, Panda S, Satpathi BR, Chawla-Sarkar M. Genetic characterization of circulating 2015 A(H1N1)pdm09 influenza viruses from Eastern India. PLoS ONE. 2016;11:e0168464. https://doi.org/10.1371/journal.pone.0168464.
Nakamura K, Shirakura M, Fujisaki S, Kishida N, Burke DF, Smith DJ, et al. Characterization of influenza A(H1N1)pdm09 viruses isolated from Nepalese and Indian outbreak patients in early 2015. Influenza Other Respir Viruses. 2017;11:399-03. https://doi.org/10.1111/irv.12469.
Parida M, Dash PK, Kumar JS, Joshi G, Tandel K, Sharma S, et al. Emergence of influenza A (H1N1)pdm09 genogroup 6B and drug resistant virus, India, January to May 2015. Euro Surveill. 2016;21:6–11. https://doi.org/10.2807/1560-7917.ES.2016.21.5.30124.
Ping J, Li C, Deng G, Jiang Y, Tian G, Zhang S, Bu Z, Chen H. Single-amino-acid mutation in the HA alters the recognition of H9N2 influenza virus by a monoclonal antibody. Biochem Biophys Res Commun. 2008;371:168–71. https://doi.org/10.1016/j.bbrc.2008.04.045.
Potdar VA, Chadha MS, Jadhav SM, Mullick J, Cherian SS, Mishra AC. Genetic characterization of the influenza A pandemic (H1N1) 2009 virus isolates from India. PLoS ONE. 2010;5:e9693. https://doi.org/10.1371/journal.pone.0009693.
Raymond FL, Caton AJ, Cox NJ, Kendal AP, Brownlee GG. The antigenicity and evolution of influenza H1 haemagglutinin, from 1950–1957 and 1977–1983: two pathways from one gene. Virology. 1986;148:275–87. https://doi.org/10.1016/0042-6822(86)90325-9.
Resende PC, Motta FC, Born PS, Miranda M, Siqueira MM. Whole-genome sequences of influenza A(H3N2) viruses isolated from Brazilian patients with mild illness during the 2014 season. Mem Inst Oswaldo Cruz. 2015;110:148–50. https://doi.org/10.1590/0074-02760140412.
Rogers GN, Paulson JC, Daniels RS, Skehel JJ, Wilson IA, Wiley DC. Single amino acid substitutions in influenza haemagglutinin change receptor binding specificity. Nature. 1983;304:76–8. https://doi.org/10.1038/304076a0.
Scholtissek C. Source for influenza pandemics. Eur J Epidemiol. 1994;10:455–8. https://doi.org/10.1007/BF01719674.
Sharma V, Chaudhry D, Kaushik S. Evaluation of clinical applicability of reverse transcription-loop-mediated isothermal amplification assay for detection and subtyping of Influenza A viruses. J Virol Methods. 2018;253:18–25. https://doi.org/10.1016/j.jviromet.2017.12.005.
Shaw ML, Palese P. Orthomyxoviridae. In: Knipe DM, Howley PM, editors. Fields virology. 6th ed. Philadelphia: Lippincott Williams & Wilkins; 2013. p. 1151–85.
Skehel JJ, Stevens DJ, Daniels RS, Douglas AR, Knossow M, Wilson IA, et al. A carbohydrate side chain on hemagglutinins of Hong Kong influenza viruses inhibits recognition by a monoclonal antibody. Proc Natl Acad Sci USA. 1984;81:1779–83. https://doi.org/10.1073/pnas.81.6.1779.
Suzuki Y, Ito T, Suzuki T, Holland RE Jr, Chambers TM, Kiso M, et al. Sialic acid species as a determinant of the host range of influenza A viruses. J Virol. 2000;74:11825–31. https://doi.org/10.1128/JVI.74.24.11825-11831.2000.
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol. 2013;30:2725–9. https://doi.org/10.1093/molbev/mst197.
Tumpey TM, García-Sastre A, Taubenberger JK, Palese P, Swayne DE, Basler CF. Pathogenicity and immunogenicity of influenza viruses with genes from the 1918 pandemic virus. Proc Natl Acad Sci USA. 2004;101:3166–71. https://doi.org/10.1073/pnas.0308391100.
Webster RG, Govorkova EA. Continuing challenges in influenza. Ann N Y Acad Sci. 2014;1323:115–39. https://doi.org/10.1111/nyas.12462.
Wiley DC, Wilson IA, Skehel JJ. Structural identification of the antibody-binding sites of Hong Kong influenza haemagglutinin and their involvement in antigenic variation. Nature. 1981;289:373–8. https://doi.org/10.1038/289373a0.
Zhong J, Liang L, Huang P, Zhu X, Zou L, Yu S, et al. Genetic mutations in influenza H3N2 viruses from a 2012 epidemic in Southern China. Virol J. 2013;10:345. https://doi.org/10.1186/1743-422X-10-345.
Acknowledgements
Authors are thankful to Council of Scientific & Industrial Research, New Delhi, India for providing financial support in Fellowship to Vikrant Sharma and to Post Graduate Institute of Medical Sciences (PGIMS), Rohtak, Haryana, India for providing clinical samples for the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human samples were in accordance with the ethical standards of the Human Ethical Committee (HEC) of Maharshi Dayanand University, Rohtak, Haryana, India (Letter No. PHY/13/362).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sharma, V., Sharma, M., Dhull, D. et al. Phylogenetic analysis of the hemagglutinin gene of influenza A(H1N1)pdm09 and A(H3N2) virus isolates from Haryana, India. VirusDis. 30, 336–343 (2019). https://doi.org/10.1007/s13337-019-00532-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13337-019-00532-7