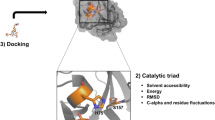
Abstract
In 2011, the FDA approved boceprevir as a hepatitis C virus (HCV) NS3 serine protease inhibitor. The sustained virological response rate for treatment with this approved compound is considerably low. Patients have not responded as much as expected to boceprevir therapy. In this in silico study, modified boceprevir compounds are suggested and tested on wild-type HCV NS3 protease and 19 mutated HCV NS3 proteases using molecular docking. Results reveal the superiority of two of the proposed modified compounds to boceprevir. One of which appears to be more potent than boceprevir itself concerning activity against wild-type NS3 and most of the examined mutated NS3 proteases.





Similar content being viewed by others
References
Abdel-Al A, El-Ahwany E, Zoheiry M, Hassan M, Ouf A, Abu-Taleb H, Rahim AA, El-Talkawy MD, Zada S. miRNA-221 and miRNA-222 are promising biomarkers for progression of liver fibrosis in HCV Egyptian patients. Virus Res. 2018;253:135–9.
Asselah T. Daclatasvir plus sofosbuvir for HCV infection: an oral combination therapy with high antiviral efficacy. J Hepatol. 2014;61(2):435–8.
Berman H, Henrick K, Nakamura H. Announcing the worldwide Protein Data Bank. Nat Struct Mol Biol. 2003;10(12):980.
Bogen S, Saksena AK, Arasappan A, Gu H, Njoroge FG, Girijavallabhan V, Pichardo J, Butkiewicz N, Prongay A, Madison V. Bioorg: Hepatitis C virus NS3-4A serine protease inhibitors: Use of a P2–P1 cyclopropyl alanine combination for improved potency. Med. Chem. Lett. 2005;15:4515–9.
Bogen SL, Arasappan A, Velazquez F, Blackman M, Huelgas R, Pan W, Siegel E, Nair LG, Venkatraman S, Guo Z, Doll R, Shih N, Njoroge FG. Discovery of potent sulfonamide P4-capped ketoamide second generation inhibitors of hepatitis C virus NS3 serine protease with favorable pharmacokinetic profiles in preclinical species. Bioorg Med Chem. 2010;18:1854–65.
Cao Y, Bao Y, Xia W, Wu H, Wei F, Zhang Y, Zhang R, Xu X. Resistance-associated mutations to HCV protease inhibitors naturally pre-existed in HIV/HCV coinfected, treatment-naive patients. Clin Res Hepatol Gastroenterol. 2016;40(5):597–604.
Chen KX, Bancha V, Weiying Y, Mousumi S, Francisco V, Tin-Yau C, Srikanth V, Anilkumar GN, Zeng Q, Bennet F. Structure-activity relationship (SAR) Development and discovery of potent indole-based inhibitors of the Hepatitis C Virus (HCV) NS5B Polymerase. J Med Chem. 2012;55:754–65.
Choo QL, Kuo G, Weiner AJ, Overby LR, Bradley DW, Houghton M. Isolation of a cDNA clone derived from a blood-borne non-A, non-B viral hepatitis genome. Science (New York, NY). 1989;244(4902):359–62.
Das D, Hong J, Chen SH, Wang G, Beigelman L, Seiwert SD, Buckman BO. Recent advances in drug discovery of benzothiadiazine and related analogs as HCV NS5B polymerase inhibitors. Bioorg Med Chem. 2011;19(16):4690–703.
Elfiky AA, Elshemey WM. IDX-184 is a superior HCV direct-acting antiviral drug: a QSAR study. Med Chem Res. 2016;25:1005–8.
Elfiky AAE, Wael M, Gawad Wissam A. 2′-Methylguanosine prodrug (IDX-184), phosphoramidate prodrug (Sofosbuvir), diisobutyryl prodrug (R7128) are better than their parent nucleotides and ribavirin in Hepatitis C virus inhibition: a molecular modeling study. J Comput Theor Nanosci. 2015;12(3):376–86.
Ezat AA, El-Bialy NS, Mostafa HI, Ibrahim MA. Molecular docking investigation of the binding interactions of macrocyclic inhibitors with HCV NS3 protease and its mutants (R155K, D168A and A156V). Protein J. 2014;33(1):32–47.
Ezat AA, Mostafa HI, El-Bialy N, Saleh NA, Ibrahim MA. Computational approaches to study peptidomimetic and macrocyclic Hepatitis C virus NS3 protease inhibitors. J Comput Theor Nanosci. 2015;12(1):52–9.
Ganta NM, Gedda G, Rathnakar B, Satyanarayana M, Bhaskar Yamajala B, Mohamed Jawed Ahsan MJ, Jadav SS, Balaraju T. A review on HCV inhibitors: significance of non-structural polyproteins. Eur J Med Chem. 2019;164:576–601.
Halfon P, Locarnini S. Hepatitis C virus resistance to protease inhibitors. J Hepatol. 2011;55(1):192–206.
Kirschberg TA, Squires NH, Yang H, Corsa AC, Tian Y, Tirunagari N, Sheng XC, Kim CU. Novel, sulfonamide linked inhibitors of the hepatitis C virus NS3 protease. Bioorg Med Chem Lett. 2014;24(3):969–72.
Matthew AN, Leidner F, Newton A, Petropoulos CJ, Huang W, Ali A, Kurtyilmaz N, Schiffer CA. Molecular mechanism of resistance in a clinically significant double-mutant variant of HCV NS3/4A protease. Structure. 2018;26(1360–1372):e5.
Muegge I. A knowledge-based scoring function for protein–ligand interactions: probing the reference state. Perspect Drug Discov Des. 2000;20:99–114.
Nagpal N, Goyal S, Wahi D, Jain R, Jamal S, Singh A, Rana P, Grover A. Molecular principles behind Boceprevir resistance due to mutations in hepatitis C NS3/4A protease. Gene. 2015;570(1):115–21.
Saleh NA, Elshemey WM. Structure-based drug design of novel peptidomimetic cellulose derivatives as HCV-NS3 protease inhibitors. Life Sci. 2017;187:58–63.
Saleh NA, Elfiky AA, Ezat AA, Elshemey WM, Ibrahim M. The electronic and quantitative structure activity relationship properties of modified telaprevir compounds as HCV NS3 protease inhibitors. J Comput Theor Nanosci. 2014;11(2):544–8.
Saleh NA, Ezat AA, Elfiky AA, Elshemey WM, Ibrahim M. Theoretical study on modified boceprevir compounds as NS3 protease inhibitors. J Comput Theor Nanosci. 2015;12(3):371–5.
Salentin S, Schreiber S, Haupt VJ, Adasme MF, Schroeder M. PLIP: fully automated protein–ligand interaction profiler. Nucleic Acids Res. 2015;43(W1):W443–7.
Sarrazin C, Hézode C, Zeuzem S, Pawlotsky J-M. Antiviral strategies in hepatitis C virus infection. J Hepatol. 2012;56:S88–100.
Sorbo MC, Cento V, Di Maio VC, Howe AY, Garcia F, Perno CF, Ceccherini-Silberstein F. Hepatitis C virus drug resistance associated substitutions and their clinical relevance: update 2018. Drug Resist Updates. 2018;37:17–39.
Stewart JJP. Optimization of parameters for semiempirical methods. J Comput Chem. 1991;12(3):320–41.
Yang PL, Gao M, Lin K, Liu Q, Villareal VA. Anti-HCV drugs in the pipeline. Curr Opin Virol. 2011;1(6):607–16.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that there are no conflicts of interest related to this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ezat, A.A., Elfiky, A.A., Elshemey, W.M. et al. Novel inhibitors against wild-type and mutated HCV NS3 serine protease: an in silico study. VirusDis. 30, 207–213 (2019). https://doi.org/10.1007/s13337-019-00516-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13337-019-00516-7