Abstract
Background
Cytochrome P450 (CYP) enzymes are one of the main sources of variability in drug metabolic clearance. Information on their abundance levels is therefore crucial to optimize scaling factors for in vitro–in vivo extrapolation (IVIVE) to predict metabolic clearance.
Objective
This study aims to quantify the abundance data of hepatic drug-metabolizing CYP enzymes in East Asian subjects reported from various sources in the literature using meta-analysis.
Method
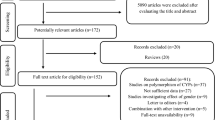
We conducted a meta-analysis on the abundance of drug-metabolizing CYP enzymes in the liver of East Asian adults. Eligible reports were identified based on predefined criteria—(1) individual liver microsomal samples, and (2) absolute protein abundance data from normal tissues of East Asian adult subjects. Subgroup and sensitivity analyses were also performed.
Results
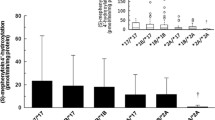
Among the 11 CYP isoforms analyzed in East Asian subjects, CYP3A5 and CYP3A4 had the highest protein levels. In particular, the number of studies and the liver sample used to quantify the abundance of CYP3A4 were the largest. Of the isoforms involved, CYP2J2 and CYP2B6 had the lowest abundance level, i.e., <5 pmol/ mg of microsomal protein. For enzymes with abundance values available in both Chinese and Japanese subjects (CYP1A2, CYP2C9, CYP3A4, and CYP3A5), the abundance level of each CYP isoform appeared to be higher in Chinese than in Japanese subjects. The most distinct difference was observed in CYP3A5 abundance.
Conclusion
The current meta-analysis shows that the abundance levels of CYP enzymes appear to vary greatly among different East Asian individuals who have similar ethnic backgrounds and food habits. The pooled data of CYP abundance can be used as preliminary reference values along with the associated variations for the projections of pharmacokinetics through physiologically based pharmacokinetic (PBPK) approaches.


Similar content being viewed by others
References
Zanger UM, Schwab M. Cytochrome P450 enzymes in drug metabolism: regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol Ther. 2013;138(1):103–41.
Barter ZE, Tucker GT, Rowland-Yeo K. Differences in cytochrome p450-mediated pharmacokinetics between Chinese and Caucasian populations predicted by mechanistic physiologically based pharmacokinetic modelling. Clin Pharmacokinet. 2013;52(12):1085–100.
Li GF, Yu G, Liu HX, Zheng QS. Ethnic-specific in vitro–in vivo extrapolation and physiologically based pharmacokinetic approaches to predict cytochrome P450-mediated pharmacokinetics in the Chinese population: opportunities and challenges. Clin Pharmacokinet. 2014;53(2):197–202.
Zhou L, Sharma P, Yeo KR, Higashimori M, Xu H, Al-Huniti N, et al. Assessing pharmacokinetic differences in Caucasian and East Asian (Japanese, Chinese and Korean) populations driven by CYP2C19 polymorphism using physiologically-based pharmacokinetic modelling. Eur J Pharm Sci. 2019;139:105061.
Li GF, Zheng QS, Yu Y, Zhong W, Zhou HH, Qiu F, et al. Impact of ethnicity-specific hepatic microsomal scaling factor, liver weight, and cytochrome P450 (CYP) 1A2 content on physiologically based prediction of CYP1A2-mediated pharmacokinetics in young and elderly Chinese Adults. Clin Pharmacokinet. 2019;58(7):927–41.
Yu G, Zhou HH, Zheng QS, Li GF. Interethnic scaling of fraction unbound of a drug in plasma and volume of distribution: an analysis of extrapolation from Caucasians to Chinese. Eur J Clin Pharmacol. 2019;75(4):543–51.
Zhang HF, Wang HH, Gao N, Wei JY, Tian X, Zhao Y, et al. Physiological content and intrinsic activities of 10 cytochrome P450 isoforms in human normal liver microsomes. J Pharmacol Exp Ther. 2016;358(1):83–93.
Shu Y, Cheng ZN, Liu ZQ, Wang LS, Zhu B, Huang SL, et al. Interindividual variations in levels and activities of cytochrome P-450 in liver microsomes of Chinese subjects. Acta Pharmacol Sin. 2001;22(3):283–8.
Shu Y, Wang LS, Xiao WM, Wang W, Huang SL, Zhou HH. Probing CYP2C19 and CYP3A4 activities in Chinese liver microsomes by quantification of 5-hydroxyomeprazole and omeprazole sulphone. Acta Pharmacol Sin. 2000;21(8):753–8.
Xiao K, Gao J, Weng SJ, Fang Y, Gao N, Wen Q, et al. CYP3A4/5 activity probed with testosterone and midazolam: correlation between two substrates at the microsomal and enzyme levels. Mol Pharm. 2019;16(1):382–92.
Tateishi T, Nakura H, Asoh M, Watanabe M, Tanaka M, Kumai T, et al. A comparison of hepatic cytochrome P450 protein expression between infancy and postinfancy. Life Sci. 1997;61(26):2567–74.
Imaoka S, Yamada T, Hiroi T, Hayashi K, Sakaki T, Yabusaki Y, et al. Multiple forms of human P450 expressed in Saccharomyces cerevisiae. Systematic characterization and comparison with those of the rat. Biochem Pharmacol. 1996;51(8):1041–50.
Inoue K, Yamazaki H, Imiya K, Akasaka S, Guengerich FP, Shimada T. Relationship between CYP2C9 and 2C19 genotypes and tolbutamide methyl hydroxylation and S-mephenytoin 4′-hydroxylation activities in livers of Japanese and Caucasian populations. Pharmacogenetics. 1997;7(2):103–13.
Yasumori T, Nagata K, Yang SK, Chen LS, Murayama N, Yamazoe Y, et al. Cytochrome P450 mediated metabolism of diazepam in human and rat: involvement of human CYP2C in N-demethylation in the substrate concentration-dependent manner. Pharmacogenetics. 1993;3(6):291–301.
Chen LS, Yasumori T, Yamazoe Y, Kato R. Hepatic microsomal tolbutamide hydroxylation in Japanese: in vitro evidence for rapid and slow metabolizers. Pharmacogenetics. 1993;3(2):77–85.
Shimada T, Tsumura F, Yamazaki H, Guengerich FP, Inoue K. Characterization of (+/−)-bufuralol hydroxylation activities in liver microsomes of Japanese and Caucasian subjects genotyped for CYP2D6. Pharmacogenetics. 2001;11(2):143–56.
Inoue K, Yamazaki H, Shimada T. Characterization of liver microsomal 7-ethoxycoumarin O-deethylation and chlorzoxazone 6-hydroxylation activities in Japanese and Caucasian subjects genotyped for CYP2E1 gene. Arch Toxicol. 2000;74(7):372–8.
Tateishi T, Watanabe M, Moriya H, Yamaguchi S, Sato T, Kobayashi S. No ethnic difference between Caucasian and Japanese hepatic samples in the expression frequency of CYP3A5 and CYP3A7 proteins. Biochem Pharmacol. 1999;57(8):935–9.
Yamaori S, Yamazaki H, Iwano S, Kiyotani K, Matsumura K, Honda G, et al. CYP3A5 Contributes significantly to CYP3A-mediated drug oxidations in liver microsomes from Japanese subjects. Drug Metab Pharmacokinet. 2004;19(2):120–9.
Shimada T, Yamazaki H, Mimura M, Inui Y, Guengerich FP. Interindividual variations in human liver cytochrome P-450 enzymes involved in the oxidation of drugs, carcinogens and toxic chemicals: studies with liver microsomes of 30 Japanese and 30 Caucasians. J Pharmacol Exp Ther. 1994;270(1):414–23.
Nakasa H, Komiya M, Ohmori S, Rikihisa T, Kiuchi M, Kitada M. Characterization of human liver microsomal cytochrome P450 involved in the reductive metabolism of zonisamide. Mol Pharmacol. 1993;44(1):216–21.
Yamazaki H, Okayama A, Imai N, Guengerich FP, Shimizu M. Inter-individual variation of cytochrome P4502J2 expression and catalytic activities in liver microsomes from Japanese and Caucasian populations. Xenobiotica. 2006;36(12):1201–9.
Li GF, Yu G, Li Y, Zheng Y, Zheng QS, Derendorf H. Quantitative estimation of plasma free drug fraction in patients with varying degrees of hepatic impairment: a methodological evaluation. J Pharm Sci. 2018;107(7):1948–56.
Ladumor MK, Thakur A, Sharma S, Rachapally A, Mishra S, Bobe P, et al. A repository of protein abundance data of drug metabolizing enzymes and transporters for applications in physiologically based pharmacokinetic (PBPK) modelling and simulation. Sci Rep. 2019;9(1):9709.
Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5:13.
Wan X, Wang W, Liu J, Tong T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol. 2014;14:135.
Achour B, Barber J, Rostami-Hodjegan A. Expression of hepatic drug-metabolizing cytochrome p450 enzymes and their intercorrelations: a meta-analysis. Drug Metab Dispos. 2014;42(8):1349–56.
Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. 2002;21(11):1539–58.
Kuehl P, Zhang J, Lin Y, Lamba J, Assem M, Schuetz J, et al. Sequence diversity in CYP3A promoters and characterization of the genetic basis of polymorphic CYP3A5 expression. Nat Genet. 2001;27(4):383–91.
Guo Y, Lucksiri A, Dickinson GL, Vuppalanchi RK, Hilligoss JK, Hall SD. Quantitative prediction of CYP3A4- and CYP3A5-mediated drug interactions. Clin Pharmacol Ther. 2019.
Barter ZE, Perrett HF, Yeo KR, Allorge D, Lennard MS, Rostami-Hodjegan A. Determination of a quantitative relationship between hepatic CYP3A5*1/*3 and CYP3A4 expression for use in the prediction of metabolic clearance in virtual populations. Biopharm Drug Dispos. 2010;31(8–9):516–32.
Li GF, Zheng QS. Modeling drug disposition and drug–drug interactions through hypothesis-driven physiologically based pharmacokinetics: a reversal translation perspective. Eur J Drug Metab Pharmacokinet. 2018;43(3):369–71.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Guo Yu was supported in part by the National Natural Science Foundation of China (no. 81603209) and Jiangsu Provincial Science Fund for Distinguished Young Scholars (BK20200005) for the conduct of this study. The funding organization played no role in the preparation, review, or approval of the manuscript.
Conflict of interest
Xiao-Xiao An, Yichao Yu, Guo-Fu Li, Guo Yu have no conflicts of interest that are directly relevant to the contents of this manuscript.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable.
Code availability
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
An, XX., Yu, Y., Li, GF. et al. Abundance and Associated Variations of Cytochrome P450 Drug-Metabolizing Enzymes in the Liver of East Asian Adults: A Meta-Analysis. Eur J Drug Metab Pharmacokinet 46, 225–233 (2021). https://doi.org/10.1007/s13318-020-00667-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13318-020-00667-9