Abstract
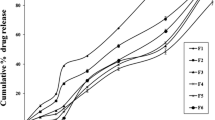
Present research deals with the development of compression-coated flurbiprofen colon-targeted tablets to retard the drug release in the upper gastro intestinal system, but progressively release the drug in the colon. Flurbiprofen core tablets were prepared by direct compression method and were compression coated using sodium alginate and Eudragit S100. The formulation is optimized based on the in vitro drug release study and further evaluated by X-ray imaging and pharmacokinetic studies in healthy humans for colonic delivery. The optimized formulation showed negligible drug release (4.33 ± 0.06 %) in the initial lag period followed by progressive release (100.78 ± 0.64 %) for 24 h. The X-ray imaging in human volunteers showed that the tablets reached the colon without disintegrating in the upper gastrointestinal tract. The C max of colon-targeted tablets was 12,374.67 ng/ml at T max 10 h, where as in case of immediate release tablets the C max was 15,677.52 ng/ml at T max 3 h, that signifies the ability of compression-coated tablets to target the colon. Development of compression-coated tablets using combination of time-dependent and pH-sensitive approaches was suitable to target the flurbiprofen to colon.






Similar content being viewed by others
References
Asghar LF, Chandran S (2006) Multiparticulate formulation approach to colon-specific drug delivery: current perspectives. J Pharm Sci 9:327–338
Asghar LF, Chure CB, Chandran S (2009) Colon specific delivery of indomethacin: effect of incorporating pH sensitive polymers in xanthan gum compression coated Bases. AAPS Pharm Sci Tech 10:418–429
Ashford M, Fell J, Attwood D, Sharma H, Woodhead P (2003) An evaluation of pectin as a carrier for drug targeting to the colon. J Con Rel 26:213–220
Aurora J, Talwar N, Pathak V (2006) Colonic drug delivery challenges and opportunities-an overview. Eur Gastro Rev 1:1–6
Chaudhary A, Tiwari N, Jain V, Singh R (2011) Microporous bilayer osmotic tablet for colon-specific delivery. Eur J Pharm Biopharm 78:134–140
Cheng G, Feng A, Zou M (2004) Time and pH dependent colon specific drug delivery for orally administered diclofencac sodium and 5-amino salicylic acid. World J Gastroenterol 10:1769–1774
Chickpetty SM, Baswaraj R, Nanjwade BK (2010) Studies on development of novel combined time and pH dependent solventless compression coated delivery systems for colonic delivery of diclofenac sodium. Asian J Pharm Clin Res 3:110–113
Chourasia MK, Jain SK (2003) Pharmaceutical approaches to colon targeted drug delivery systems. J Pharm Pharmaceut Sci 6:33–66
Davaran S, Rashidi MR, Hashemi M (2001) Synthesis and characterization of methacrylic derivatives of 5-amino salicylic acid with pH-sensitive swelling properties. AAPS Pharm Sci Tech 2:1–6
El-Kamel AH, Abdel-Aziz AM, Fatani AJ, El-Subbagh HI (2008) Oral colon targeted delivery systems for treatment of inflammatory bowel diseases: synthesis, in vitro and in vivo assessment. Int J Pharm 358:248–255
Ghimire M, Hodges LA, Band J, Lindsay B, Mahony BO, McInnes FJ, Mullen AB, Stevens HNE (2011) Correlation between in vitro and in vivo erosion behaviour of erodible tablets using gamma scintigraphy. Eur J Pharm Biopharm 77:148–157
Gibaldi M, Perrier D (1990) Pharmacokinetics. Marcel Dekker, New York
Grahnen A (1984) Design of bioavailability studies. Pharm Int 5:100–103
Hashem FM, Shaker DS, Nasr M, Saad IE, Ragaey R (2011) Guar gum and hydroxy propyl methylcellulose compressed coated tablets for colonic drug delivery: in vitro and in vivo evaluation in healthy human volunteers. Drug Disc Ther 5:90–95
ICH Q1A (2003) Stability testing guidelines: stability testing of new drug substances and products. CPMP/ICH/380/95, 1–13
Krishnaiah YSR, Satyanarayana S, Ramaprasad YV, Narasimharao S (1998) Evaluation of guar gum as a compression coat for drug targeting to colon. Int J Pharm 171:137–146
Krishnaiah YSR, Satyanarayana S, Dinesh Kumar B, Karthikeyan RS, Bhaskar P (2003a) In vivo pharmacokinetics in human volunteers: oral administered guar gum-based colon-targeted 5-fluorouracil tablets. Eur J Pharm Sci 19:355–362
Krishnaiah YSR, Raju VP, Kumar DB, Satyanarayana V, Karthikeyan RS, Bhaskar P (2003b) Pharmacokinetic evaluation of guar gum-based colon-targeted drug delivery systems of mebendazole in healthy volunteers. J Con Rel 88:95–103
Kumar M, Ali A, Kaldhone P, Shirode A, Kadam VJ (2010) Platform technologies for colon targeted drug delivery system. J Pharm Res 3:543–547
Mandal S, Basu SK, Sa B (2010) Ca2+ ion cross-linked interpenetrating network matrix tablets of polyacrylamide-grafted-sodium alginate and sodium alginate for sustained release of diltiazem hydrochloride. Carb Polym 82:867–873
Maroni A, Zema L, Curto MDD, Foppoli A, Gazzaniga A (2012) Oral colon delivery of insulin with the aid of functional adjuvants. Adv Drug Del Rev 64:540–556
Mathews BR (1999) Regulatory aspects of stability testing in Europe. Drug Dev Ind Pharm 25:831–856
Orlu M, Cevher E, Araman A (2006) Design and evaluation of colon-specific drug delivery system containing flubiprofen microsponges. Int J Pharm 318:103–117
Philip AK, Dubey RK, Pathak K (2008) Optimizing delivery of flurbiprofen to the colon using a targeted prodrug approach. J Pharm Pharmacol 60:607–613
Shukla RK, Tiwari A (2012) Carbohydrate polymers: applications and recent advances in delivering drugs to the colon. Carb Polym 88:399–416
Sinha VR, Bhinje JR, Kumaria R, Kumar M (2006) Development of pulsatile systems for targeted drug delivery of celecoxib for prophylaxis of colorectal cancer. Drug Del 13:221–225
Talukder RM, Fassihi R (2008) Development and in vitro evaluation of a colon-specific controlled release drug delivery system. J Pharm Pharmacol 60:1297–1303
Tugcu-Demiroz F, Acarturk F, Takka S, Konus-Boyunaga O (2004) In vitro and In vivo evaluation of mesalazine-guar gum matrix tablets for colonic drug delivery. J Drug Targ 12:105–112
Valluru R, Siddaramaiah T, Pramod M (2008) Influence of natural polymer coating on novel colon targeting drug delivery system. J Mater Sci 19:2131–2136
Veerareddy PR, Vemula SK (2012) Formulation, evaluation and pharmacokinetics of colon targeted pulsatile system of flurbiprofen. J Drug Targ 20:703–714
Vemula SK, Veerareddy PR (2009) Different approaches to design and evaluation of colon specific drug delivery systems. Int J Pharm Tech 1:1–35
Vemula SK, Veerareddy PR (2011) Fast disintegrating tablets of flurbiprofen: formulation and characterization. Lat Am J Pharm 30:1135–1141
Vemula SK, Veerareddy PR (2013) Formulation, evaluation and pharmacokinetics of ketorolac tromethamine time-dependent colon targeted drug delivery system. Exp Opin Drug Del 10(1):33–45
Vincent HL, Suman KM (2002) Drug delivery-oral colon-specific. In: Swarbick J, Boylan CJ (eds) Encyclopedia of pharmaceutical technology. Marcel Dekker, New York, pp 871–885
Vyas SP, Roop KK (2006) Controlled drug delivery concepts and advances, 1st edn. Vallabh Prakashan, Delhi
Wang X, Yu J, Tang X (2007) In vitro release and pharmacokinetics of flurbiprofen sustained-release capsules containing coated pellets. Asian J Pharm Sci 2:77–84
Wu B, Shun N, Wei X, Lu Y, Wu W (2007a) Biphasic release of indomethacin from HPMC/pectin/calcium compression coated tablet: I. Characterization and mechanistic study. Eur J Pharm Biopharm 67:707–714
Wu B, Shun N, Wei X, Wu W (2007b) Characterization of 5-fluorouracil release from hydroxypropyl methylcellulose compression-coated tablets. Pharm Dev Tech 12:203–210
Acknowledgments
The authors acknowledge FDC Limited, Mumbai, India and Matrix laboratories, Hyderabad, India for gift sample of Flurbiprofen, Sodium alginate and Eudragit S100. The authors also thank the Principal and Management, Jangaon Institute of Pharmaceutical Sciences for providing the facilities.
Conflict of interest
The authors report no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vemula, S.K., Veerareddy, P.R. & Devadasu, V.R. Pharmacokinetics of colon-specific pH and time-dependent flurbiprofen tablets. Eur J Drug Metab Pharmacokinet 40, 301–311 (2015). https://doi.org/10.1007/s13318-014-0210-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13318-014-0210-0