Abstract
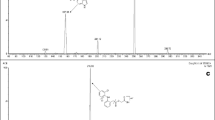
A highly sensitive and rapid method for the analysis of pantoprazole in human plasma using liquid chromatography coupled to tandem electrospray ionization mass spectrometry was developed. The procedure involves a simple protein precipitation method with methyl alcohol and separation by RP-HPLC. Detection was performed by positive ion electrospray ionization in multiple reaction monitoring mode, monitoring the transitions m/z 384.1 → 200.0 and m/z 346.1 → 198.0, for quantification of pantoprazole and IS, respectively. The standard calibration curves showed good linearity within the range of 5–5,000 ng mL−1. The lower limit of quantitation (LLOQ) was about 5 ng mL−1. The extractive recovery of pantoprazole from the biological matrix was more than 77.58% and the matrix effect was complied with relevant provision. The intra-day accuracy of the drug containing serum samples was more than 92.19% with a precision of 0.79–5.36%. The inter-day accuracy was 85.49% or more, with a precision of 0.91–12.67%. Intra and inter-day accuracy of the assay at four concentrations were 97.9–98.2% with a precision of 4.2–13.9%. This method offered good precision and accuracy and was successfully applied to the pharmacokinetic and bioequivalence studies of 40 mg of enteric-coated pantoprazole in 20 healthy Chinese volunteers.




Similar content being viewed by others
References
Cente for Drug Evaluation and Research (CDER)-Food and Drug Administration (FDA) (2002) Guidance for industry: food-effect bioavailability and fed bioequivalence studies
Committee for Proprietary Medicinal Products (CPMP) (1999) Note for guidance on modified release oral and transdermic dosage forms: section II (pharmacokinetic and clinical evaluation). CPMP/EWP/280/96
Filipe A, Almeida S, Franco Spínola AC, Neves R, Trabelsi F, Torns A et al (2008) Bioequivalence study of two enteric-coated formulations of pantoprazole in healthy volunteers under fed. J Arzneimittel-Forschung (Drug Research) 58(9):451–456
Food and Drug Administration (FDA) (2008) Guidance on pantoprazole sodium, May 2008
Ling J, Wang H, Shentu JZH (2005) Determination of pantoprazole in human plasma by HPLC-MS.J. Chin J Mod Appl Pharm 22(4):306–308
Osmair P, Oliveira CH, Barrientos-Astigarraga RE, Rezende VM, Mendes GD et al (2004) Determination of pantoprazole in human plasma by LC-MS-MS using lansoprazole as internal standard. J Arzneimittel-Forschung (Drug Research) 54(6):314–319
SFDA guidance for industry. Bioavailability studies for orally administered drug-products (2005) General considerations. China Department of Health and Human Services, State Food and Drug administration, Centre for Drug Evaluation and Research 2005. http://www.sfda.gov.cn/WS01/CL0001/
Wang LX, Li JH, Li YB (2006) Bioequivalence of pantoprazole enteric capsules in human plasma. J Clin Med Pract 10(3):30–32
Xu HY, Yang L, Zhang P, Shen T, Zhao HQ (2008) Determination of pantoprazole in human plasma by HPLC.J. J Shenyang Pharm Univ 25(1):44–47
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, Y., Ding, MJ., Ma, J. et al. Quantification of pantoprazole in human plasma using LC-MS/MS for pharmacokinetics and bioequivalence study. Eur J Drug Metab Pharmacokinet 35, 147–155 (2011). https://doi.org/10.1007/s13318-010-0018-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13318-010-0018-5