Abstract
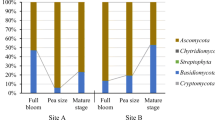
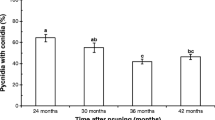
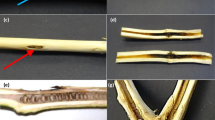
Grapevine trunk diseases cause substantial economic loss worldwide. Grapevines are susceptible to infection by the causal agents when pruning during dormancy coincides with rainfall. The ascospores of Eutypa lata, the causal agent of the trunk disease Eutypa dieback, are released by fruiting bodies following rainfall and infect fresh wounds in woody tissue. Wound-colonising microbes have the potential to compete with the pathogen but the effect on such microbes of exposure of wounds on grapevines to rainfall has not been investigated. The presence of microbes in grapevine pruning wounds, both exposed to and sheltered from rainfall, was investigated at weekly intervals for one month following pruning. Surface disinfected tissue was cultured and the predominant microbes identified by morphology and by Sanger sequencing. Epicoccum nigrum, Aureobasidium pullulans, Alternaria and Cladosporium spp. were the predominant fungi, E. lata was not isolated and bacteria were isolated only rarely. The number of microbes found in rainfall-exposed wounds was greater than in those protected from rainfall and, of the above-mentioned, Epicoccum nigrum was predominant. Three of these species competed for space with E. lata when co-cultured in vitro.









Similar content being viewed by others
Data availability
The data will be available in Henderson's PhD thesis in due course.
References
Abdulkareem MK, Kimber RBE, Scott ES (2019) Interactions between Ascochyta fabae and Cercospora zonata, fungal pathogens of faba bean. Australas Plant Pathol 48:271–280
Ayres MR, Wicks TJ, Scott ES, Sosnowski MR (2017) Developing pruning wound protection strategies for managing Eutypa dieback. Aust J Grape Wine Res 23:103–111
Biggs AR (1990) Managing wound-associated diseases by understanding wound healing in the bark of woody plants. J Arboric 16:108–112
Billones-Baaijens R, Ayres M, Savocchia S, Sosnowski M (2017) Trunk disease: monitoring inoculum dispersal by grapevine trunk disease pathogens using spore traps. Wine Vitic J 32(4):46–50
Bruez E, Baumgartner K, Bastien S, Travadon R, Guerin-Dubrana L, Rey P (2016) Various fungal communities colonise the functional wood tissues of old grapevines externally free from grapevine trunk disease symptoms. Aust J Grape Wine Res 22:288–295
Bonada M, Edwards EJ, McCarthy MG, Sepúlveda GC, Petrie PR (2020) Impact of low rainfall during dormancy on vine productivity and development. Aust J Grape Wine Res 26:325–342
Carter MV (1991) The status of Eutypa lata as a pathogen. Monograph - Phytopathol Paper 32. CAB International Wallingford, England.
Carter MV, Moller J (1970) Duration of susceptibility of apricot pruning wounds to infection by Eutypa armeniacae. Aust J Agric Res 21:915–920
Carter MV, Price TV (1974) Biological control of Eutypa armeniacae. II Studies of interaction between E. armeniacae and Fusarium lateritium and their relative sensitivities to benomyl chemicals. Aust J Agric Res 25:105–119
Chapuis L, Richard L, Dubos B (1998) Variation in susceptibility of grapevine pruning wound to infection by Eutypa lata in south western France. Plant Pathol 47:463–472
de Cal A, Larena I, Linan M, Torres R, Lamarca N, Usall J, Domenichini P, Bellini A, de Eribe XO, Melgarejo P (2009) Population dynamics of Epicoccum nigrum, a biocontrol agent against brown rot in stone fruit. J Appl Microbiol 106:592–605
Del Frari G, Gobbi A, Aggerbeck MR, Oliveira H, Hansen LH, Ferriera B (2019) Fungicides and the grapevine wood mycobiome: a case study on tracheomycotic ascomycete Phaeomoniella chlamydospora reveals potential for two novel control strategies. Front Plant Sci 10:1405. https://doi.org/10.3389/fpls.2019.01405
Elena G, Sosnowski MR, Ayres MR, Lecomte P, Benetreau C, Garcia-Figueres F, Luque J (2015) Effect of the inoculum dose of three grapevine trunk pathogens on the infection of artificially inoculated pruning wounds. Phytopathol Mediterr 54:345–354
Ferreira JHS, Matthee FN, Thomas AC (1991) Biological control of Eutypa lata on grapevine by an antagonistic strain of Bacillus subtilis. Phytopathology 81:283–287
Gramaje D, Urbez-Torres JR, Sosnowski MR (2018) Managing grapevine trunk diseases with respect to epidemiology and etiology: current strategies and future prospects. Plant Dis 102:12–39
Halleen F, Fourie PH, Lombard PJ (2010) Protection of grapevine pruning wounds against Eutypa lata by biological and chemical methods. S Afr J Enol Vitic 31:125–132
Henderson B, Sosnowski MR, McCarthy MG, Scott ES (2021) Influence of pruning method on the incidence and severity of Eutypa dieback in Shiraz grapevines. Aust J Grape Wine Res 27:87–93. https://doi.org/10.1111/ajgw.12465
John S, Scott ES, Wicks TJ, Hunt JS (2005) Protection of grapevine pruning wounds from infection by Eutypa lata using Trichoderma harzianum and Fusarium lateritium. Australas Plant Pathol 34:569–575
John S, Wicks TJ, Hunt JS, Scott ES (2008) Colonisation of grapevine wood by Trichoderma harzianum and Eutypa lata. Aust J Grape Wine Res 14:18–24
Kraus C, Voegele RT, Fischer M (2019) Temporal development of the culturable, endophytic fungal community in healthy grapevine branches and occurrence of GTD-associated fungi. Microb Ecol 77:866–876
Moller WJ, Carter MV (1965) Production and dispersal of ascospores in Eutypa armeniacae. Aust J Biol Sci 18:67–80
Moller WJ, Kasimatis AN (1978) Dieback of grapevines caused by Eutypa armeniacae. Plant Dis Rep 62:254–258
Munkvold GP, Marois JJ (1993) Efficacy of natural epiphytes and colonizers of grapevine pruning wounds for biological control of eutypa dieback. Phytopathology 83:624–629
Munkvold GP, Marois JJ (1995) Factors associated with variation in susceptibility of pruning wounds to infection by Eutypa lata. Phytopathology 85:249–256
Pancher M, Ceol M, Corneo PE, Longa CMO, Yousaf S, Pertot I, Campisano A (2012) Fungal endophytic communities in grapevines (Vitis vinifera L.) respond to crop management. Appl Environ Microbiol 78:4308–4317
Petzoldt CH, Moller WJ, Sall MA (1981) Eutypa dieback of grapevine. Seasonal differences in infection and duration of susceptibility of pruning wounds. Phytopathology 71:540–543
Price T (1973) Studies on the microbial colonization of sapwood of pruned apricot trees. Aust J Biol Sci 26:379–388
Rolshausen PE, Gubler WD (2005) Use of Boron for the control of Eutypa dieback of grapevines. Plant Dis 89:734–738
Schoch CL, Seifert KA, Huhndorf S, Robert V, Spouge JL, Levesque CA et al (2012) Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc Natl Acad Sci USA 109:6241–6246
Scholefield P, Loschiavo A, Morison J, Ferris M (2010) The true cost of pest and disease. Aust NZ Grapegr Winemak 557a: 6–10
Siebert JB (2001) Eutypa: the economic toll on vineyards. Wines Vines 4:50–56
Sosnowski MR, Ayres MR, Billones-Baaijens R, Savocchia S, Scott ES (2023) Susceptibility of pruning wounds to grapevine trunk disease pathogens Eutypa lata and Diplodia seriata in three climatic conditions in Australia. Fungal Ecol 64 https://doi.org/10.1016/j.funeco.2023.101260
Sosnowski MR, Lardner R, Wicks TJ, Scott ES (2007) Influence of grapevine cultivar and isolate of Eutypa lata on wood and foliar symptoms. Plant Dis 91:924–931
Sosnowski MR, Loschiavo A, Wicks TJ, Scott ES (2013) Evaluating treatments and spray application for the protection of grapevine pruning wounds from infection by Eutypa lata. Plant Dis 97:1599–1604
Sosnowski M, Ayres M, Billones-Baaijens R, Savocchia S (2022) Grapevine trunk disease management for vineyard longevity in diverse climates of Australia. Final Report to Wine Australia, Project SAR1701–1.1, July 2022.
Sun Q, Rost TL, Matthews MA (2008) Wound-induced vascular occlusions in Vitis vinifera (Vitaceae): Tyloses in summer and gels in winter. Am J Bot 95:1498–1505
Taguiam JD, Evallo E, Balendres MA (2021) Epicoccum species: ubiquitous plant pathogens and effective biological control agents. Eur J Plant Pathol 159:713–725
Tassie E, Freeman BM (1992) Pruning. In: Coombe BG, Dry PR (eds) Viticulture, vol 2. – Practices. Winetitles, Adelaide, South Australia, Australia, pp 66–84
Wicks TJ (1975) The dying arm disorder of vines of South Australia. Agric Rec 2:14–20
Acknowledgements
The University of Adelaide provided funding and infrastructure support and SARDI provided infrastructure support. Michael McCarthy provided supervision during the conduct of the investigation and, with Paul Petrie and Marcos Bonada, access to the SARDI vineyard site at Nuriootpa, South Australia. The authors acknowledge Ricardo dos Santos, University of São Paulo, Brazil for DNA sample preparation and interpretation of sequencing results, and Paul Gooding, Maia Rabinovich and John Stephen, Australian Genome Research Facility, for DNA sequencing and advice. Cathryn Todd, Suzanne McKay, Matthew Ayres and Brittany Oswald (SARDI) provided advice about experimental protocols and the identity of fungal isolates.
Author information
Authors and Affiliations
Contributions
BH, MS and ES contributed to conception and design of the project. BH conducted field and laboratory work and analysed the data. BH, MS and ES contributed to interpretation of the data. BH drafted the manuscript and ES and MS provided critical review and revision. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Henderson, B., Sosnowski, M.R. & Scott, E.S. The influence of rainfall on the mycobiota isolated from grapevine pruning wounds. Australasian Plant Pathol. 52, 271–282 (2023). https://doi.org/10.1007/s13313-023-00917-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13313-023-00917-9