Abstract
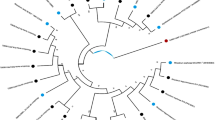
Ironwood (Casuarina equisetifolia subsp. equisetifolia) is a nitrogen-fixing tree of considerable social, economic and environmental importance that commonly occurs in tropical/subtropical zones of Asia, the Pacific, Africa, and Central America. Ironwood decline was first noticed on Guam in 2002 and is now affecting thousands of trees and impacting the ecosystem. In 2012, a survey showed that Ralstonia solanacearum and Klebsiella spp. were associated with wetwood symptoms of declining trees. R. solanacearum strains isolated from diseased ironwood in Guam were similar to R. solanacearum strain GMI1000, having similar BOX-PCR profiles and belonging to phylotype I and biovar 3. Two Klebsiella species (K. variicola and K. oxytoca) were recovered, with K. variicola being the more prevalent species. Pathogenicity tests revealed that R. solanacearum caused wilt in tomato and ironwood seedlings, whereas neither Klebsiella spp. produced symptoms. There were no differences in virulence between Guam R. solanacearum and control strains following inoculation into tomato and ironwood from Hawaii. Additionally, no observable differences in ironwood susceptibility to Ralstonia strains from Guam or Hawaii, were observed, suggesting that the association of Guam R. solanacearum with Guam ironwood is not specific. Co-inoculation studies with both R. solanacearum and Klebsiella variicola and K. oxytoca revealed that Klebsiella sp. did not affect symptoms produced by R. solanacearum alone. In planta studies were feasible only on seedlings and young trees in Hawaii; thus, possible interactions between R. solanacearum and Klebsiealla sp. in adult trees remain to be investigated. A new in-field survey of declining ironwood is needed to better understand the role of Klebsiella and Ralstonia in ironwood tree decline in Guam.





Similar content being viewed by others
References
Ali MIM, Anuratha CS, Sharma JK (1991) Bacterial wilt of Casuarina equisetifolia in India. Eur J Forest Pathol 21(4):234–238
Altschul SF, Gish W, Miller W, Ew M, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Alvarez AM, Berestecky J, Stiles JI, Ferreira SA, Benedict AA Serological and molecular approaches to identification of Pseudomonas solanacearum strains from Heliconia. In ‘ACIAR Proc.’, 1993, Kaohsiung, Taiwan. (Eds GL Hartman and AC Hayward), pp. 62–69
Athens SJ, Ward JV (2004) Holocene vegetation, savanna origins and human settlement of Guam. In A Pacific Odysey: Archaeology and Anthropology in the Western Pacific. Papers in Honour of Jim Specht. Rec Aust Mus 29:15–30
Cook D, Barlow E, Sequeira L (1989) Genetic diversity of Pseudomonas solanacearum: detection of restriction fragment length polymorphisms with DNA probes that specify virulence and the hypersensitive response. Mol Plant Microbe Interact 2(3):113–121
Engelbrecht M (1994) Modification of as semi-selective medium for the isolation and quantification of Pseudomonas solanacearum. In ‘ACIAR Bacterial Wilt Newsletter. Vol. 10’. pp. 3–5)
Fegan M, Prior P (2005) ‘How complex is the Ralstonia solanacearum species complex?’ (American Phytopathological Society Press: St Paul, MN)
Fosberg F, Sachet M-H, Oliver R (1979) A geographical checklist of the Micronesian dicotyledonae. Micronesica 15:41–295
Gabriel DW, Allen C et al (2006) Identification of open reading frames unique to a select agent: Ralstonia solanacearum race 3 biovar 2. Mol Plant Microbe Interact 19(1):69–79
Ghezzi JI, Steck TR (1999) Induction of the viable but non-culturable condition in Xanthomonas campestris pv. campestris in liquid microcosms and sterile soil. FEMS Microbiol Ecol 30(3):203–208
Grey BE, Steck TR (2001) The viable but nonculturable state of Ralstonia solanacearum may be involved in long-term survival and plant infection. Appl Environ Microbiol 67(9):3866–3872
Hartley C, Davidson RW, Crandall BS (1961) Wetwood, bacteria, and increased pH in trees. United States Department of Agriculture Forest Service. In Cooperation with the University of Wisconsin, Madison, WI
Hayward AC (1964) Characteristics of Pseudomonas solanacearum. J Appl Bacteriol 27(2):265–277
He LY, Sequeira L, Kelman A (1983) Characteristics of strains of Pseudomonas solanacearum from China. Plant Dis 67(2):1357–1361
Hugh R, Leifson E (1953) The taxonomic significance of fermentative versus oxidative metabolism of carbohydrates by various gram-negative rods. J Bacteriol 66:24–26
Kogan S, Doherty M, Gitschier J (1987) An improved method for prenatal diagnosis of genetic diseases by analysis of amplified DNA sequences. New Engl J Med 317:985–990
Kubota R, Schell M, Peckham G, Rue J, Alvarez AM, Allen C, Jenkins D (2011) In silico genomic subtraction guides development of highly accurate, DNA-based diagnostics for Ralstonia solanacearum race 3 biovar 2 and blood disease bacterium. J Gen Plant Pathol 77(3):182–193
Kubota R, Vine BG, Alvarez AM, Jenkins DM (2008) Detection of Ralstonia solanacearum by loop-mediated isothermal amplification. Phytopathology 98(9):1045–1051
Mersha Z, Schlub R, Moore A (2009) The state of ironwood (Casuarina equisetifolia subsp. equisetifolia) decline on the Pacific island of Guam. In ‘APS annual proceedings. Vol. 99’. Portland, Oregon )
Mersha Z, Schlub R, Spaine P, Smith J, Nelson S Visual and quantitative characterization of ironwood tree (C.equisetifolia) decline on Guam. In ’APS annual meeting’, 2010, Charlotte, NC,
Morton JF (1980) The Australian pine or beefwood (Casuarina equisetifolia L.), an invasive “weed” tree in Florida. Proc Fla State Hort Soc 93:87–95
Norman D, Alvarez A (1989) A rapid method for presumptive identification of Xanthomonas campestris pv. diffenbachiae and other xanthomonads. Plant Dis 73:654–658
Oliver J (2000) The public health significance of viable but non-culturable bacteria. In ‘Nonculturable microorganisms in the environment.’ (Eds R Colwell and D Grimes) pp. 277–299. (American Society for Microbiology Press: Washington, DC)
Oliver J (2005) The viable but non-culturable state in bacteria. J Microbiol 43:93–100
Opina N, Tavner F et al (1997) A novel method for development of species and strain-specific DNA probes and PCR primers for identifying Burkholderia solanacearum (formerly Pseudomonas solanacearum). Asia Pac J Mol Biol Biotechnol 5:19–30
Orian G (1961) Diseases of Filao (Casuarina equisetifolia) forest in Mauritius. Revue agricole et sucrière de l’île Maurice 40(1):17–45
Podschun R, Ullmann U (1998) Klebsiella spp. as nosocomial pathogens: epidemiology, taxonomy, typing methods, and pathogenicity factors. Clin Microbiol Rev 11(4):589–603
Rosenblueth M, Martínez L, Silva J, Martínez-Romero E (2004) Klebsiella variicola, a novel species with clinical and plant associated isolates. Syst Appl Microbiol 27:27–35
Salanoubat M, Genin S et al (2002) Genome sequence of the plant pathogen Ralstonia solanacearum. Nature 415(6871):497–502
Schaad NW, Jones JB, Chun W, eds (2001) ‘Laboratory Guide for Identification of Plant Pathogenic Bacteria.’ (APS Press: St. Paul, Minnesota, USA)
Schlub KA (2010) Investigating the ironwood tree (Casuarina equisetifolia) decline on Guam using applied multinomial modeling., Louisiana State University,
Schlub RL, Kubota R, Alvarez AM (2013) Casuarina equisetifolia decline in Guam linked to colonization of woody tissues by bacteria. Phytopathology 103:S2.128
Schlub RL, Mersha Z, et al. (2011) Guam Ironwood (Casuarina equisetifolia) Tree Decline Conference and Follow-up. In ‘Improving Smallholder Livelihoods through Improved Casuarina Productivity, Proceedings of the 4th International Casuarina Workshop’, March 21–25, 2010, Haikou, China. (Eds C Zhong, K Pinyopusarerk, A Kalinganire and C Franche), pp. 239–246
Schlub RL, Moore A, Marx BD, Schlub KA, Kennaway L, Quintanilla M, Putnam M, Mersha Z (2011b) Decline of Casuarina equisetifolia (ironwood) trees on Guam: symptomology and explanatory variables. Phytopathology 101:S216
Schneider KL, Marrero G, Alvarez AM, Presting GG (2011) Classification of plant associated bacteria using RIF, a computationally derived DNA marker. PLoS 6(4):e18496
Stone BC (1970) The flora of Guam. Micronesica 6:1–659
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony Methods. Mol Biol Evol 28(10):2731–2739
Xu H-S, Roberts N, Singleton F, Attwell R, Grimes D, Colwell R (1982) Survival and viability of nonculturable Escherichia coli and Vibrio cholerae in the estuarine and marine environment. Microb Ecol 8:313–323
Acknowledgments
This work was supported in part by USDA National Institute for Food and Agriculture, Project HAW00987-H, administered by the College of Tropical Agriculture and Human Resources, University of Hawaii at Mānoa. This work was also supported in part by WPDN-201303063-01 and in part by the Guam Cooperative Extension, University of Guam, Mangilao Guam. The authors thank Zhong Chonglu at the Research Institute of Tropical Forestry, Longdong, Guangzhou, China, for providing the diseased China ironwood sample.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ayin, C.M., Schlub, R.L., Yasuhara-Bell, J. et al. Identification and characterization of bacteria associated with decline of ironwood (Casuarina equisetifolia) in Guam. Australasian Plant Pathol. 44, 225–234 (2015). https://doi.org/10.1007/s13313-014-0341-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13313-014-0341-4