Abstract
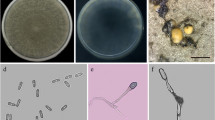
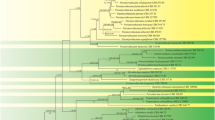
Ray blight disease of pyrethrum (Tanacetum cinerariifolium) is shown to be caused by more than one species of Stagonosporopsis. The Australian pathogen, previously identified as Phoma ligulicola var. inoxydabilis, represents a new species described as Stagonosporopsis tanaceti based on morphological characters and a five-gene phylogeny employing partial sequences of the actin, translation elongation factor 1-alpha, internal transcribed spacers and 5.8S of the nrDNA, 28S large subunit and beta-tubulin 2 gene sequences. Furthermore, the two varieties of Stagonosporopsis ligulicola are elevated to species level as S. chrysanthemi and S. inoxydabilis based on their DNA phylogeny and morphology.





Similar content being viewed by others
References
Abeln ECA, Stax AM, De Gruyter J, Van der Aa HA (2002) Genetic differentiation of Phoma exigua varieties by means of AFLP fingerprints. Mycol Res 106:419–427
Aveskamp MM, De Gruyter J, Crous PW (2008) Biology and recent developments in the systematics of Phoma, a complex genus of major quarantine significance. Fungal Divers 31:1–18
Aveskamp MM, Verkley GJM, De Gruyter J, Murace MA, Perelló A et al (2009a) DNA phylogeny reveals polyphyly of Phoma section Peyronellaea and multiple taxonomic novelties. Mycologia 101:363–382
Aveskamp MM, Woudenberg JHC, De Gruyter J, Turco E, Groenewald JZ, Crous PW (2009b) Development of taxon-specific sequence characterized amplified region (SCAR) markers based on actin sequences and DNA amplification fingerprinting (DAF): a case study in the Phoma exigua species complex. Mol Plant Pathol 10:403–414
Aveskamp MM, De Gruyter J, Woudenberg JHC, Verkley GJM, Crous PW (2010) Highlights of Didymellaceae: a polyphasic approach to characterise Phoma and related Pleosporalean genera. Stud Mycol 65:1–160
Baker KF, Dimock AW, Davis LH (1949) Life history and control of the ascochyta ray blight of chrysanthemum. Phytopathology 39:789–805
Bhat BK, Menary RC (1984) Pyrethrum production in Australia: its past and present potential. J Aust Inst Agric Sci 494:189–192
Boerema GH (1976) Phoma species studied in culture by Dr R.W.G. Dennis. Trans Br Mycol Soc 67:289–319
Boerema GH, Bollen GJ (1975) Conidiogenesis and conidial septation as different criteria between Phoma and Ascochyta. Persoonia 8:111–144
Boerema GH, Howeler LH (1967) Phoma exigua Desm. and its varieties. Persoonia 5:15–28
Boerema GH, Noordeloos ME (1992) Contributions towards a monograph of Phoma (Coelomycetes)—I 1. Section Phoma: taxa with very small conidia in vitro. Persoonia 15:71–92
Boerema GH, Van Kesteren HA (1974) Enkele bijzondere schimmelaantastingen V. (Mycologishe waarnemingen no. 17). Gewasbescherming 5:119–125
Boerema GH, Verhoeven AA (1979) Check-list for scientific names of common parasitic fungi. Series 2c: fungi on field crops: pulse (legumes), and forage crops (herbage legumes). Neth J Plant Pathol 85:151–185
Boerema GH, De Gruyter J, Noordeloos ME (1997) Contributions towards a monograph of Phoma (Coelomycetes)—IV Section Heterospora: taxa with large sized conidial dimorphs, in vivo sometimes as Stagonosporopsis synanamorphs. Persoonia 16:335–371
Boerema GH, De Gruyter J, Noordeloos ME, Hamers MEC (2004) Phoma identification manual: differentiation of specific and intra-specific taxa in culture. CABI Publishing, Wallingford
Carbone I, Kohn LM (1999) A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 91:553–556
Chesters CGC, Blakeman JP (1967) Host range and virulence of Mycosphaerella ligulicola. Ann Appl Biol 60:385–390
Clements FE, Shear CL (1931) The genera of fungi. Wilson, New York
Crous PW, Gams W, Stalpers JA, Robert V, Stegehuis G (2004) MycoBank: an online initiative to launch mycology into the 21st century. Stud Mycol 50:19–22
Crous PW, Verkley GJM, Groenewald JZ, Samson RA (2009) Fungal biodiversity. CBS laboratory manual series1. Centraalbureau voor Schimmelcultures, The Netherlands
Cullen DW, Toth IK, Boonham N, Walsh K, Barker I, Lees AK (2007) Development and validation of conventional and quantitative polymerase chain reaction assays for the detection of storage rot potato pathogens, Phytophthora erythroseptica, Pythium ultimum, Phoma foveata. J Phytopathol 155:309–315
De Gruyter J, Boerema GH, Van der Aa HA (2002) Contributions towards a monograph of Phoma (Coelomycetes)—VI. 2. Section Phyllostictoides: outline of its taxa. Persoonia 18:1–53
De Gruyter J, Aveskamp MM, Woudenberg JHC, Verkley GJM, Groenewald JZ, Crous PW (2009) Molecular phylogeny of Phoma and allied anamorph genera: towards a reclassification of the Phoma complex. Mycol Res 133:508–519
De Gruyter J, Woudenberg JHC, Aveskamp MM, Verkley GJM, Groenewald JZ, Crous PW (2010) Systematic reappraisal of species in Phoma section Paraphoma, Pyrenochaeta and Pleurophoma. Mycologia 102:1066–1081
De Gruyter J, Woudenberg JHC, Aveskamp MM, Verkley GJM, Groenewald JZ, Crous PW (2012) Rediposition of Phoma-like anamorphs in Pleosporales. Stud Mycol 75:1–36
De Hoog GS, Gerrits Van den Ende AHG (1998) Molecular diagnostics of clinical strains of filamentous Basidiomycetes. Mycoses 41:183–189
Diedicke H (1912) Die Abteilung Hyalodidymae der Sphaerioideen. Ann Mycol 10:135–152
Drummond AJ, Ashton B, Buxton S, Cheung M, Cooper A, et al (2011) Geneious v5.4 Available from <www.geneious.com>
EPPO (1980) Data sheets on quarantine organisms List A2. Didymella chrysanthemi (Tassi) Garibaldi & Gullino. Paris. rue Le Notre
EPPO (1982) Datasheets on quarantine organisms Set 5 List A2 (quarantine organisms present in some EPPO countries). EPPO Bull 12:41–46
Farris JS, Kallersjo M, Kluge AG, Bult C (1995) Testing significance of incongruence. Cladistics 10:315–319
Garibaldi A, Gullino G (1971) Brevi notizie sulla presenza in Italia dell’ascochitosi del crisantemo. L’Agricoltura Italiana 71:21–290
Grdiša M, Karovic-Stanko K, Kolak I, Satovic Z (2009) Morphological and biochemical diversity of Dalmatian pyrethrum (Tanacetum cinerariifolium (Trevir.) Sch. Bip.). Agric Conspec Sci 74:73–80
Greene HC (1961) Notes on Wisconsin parasitic fungi. XXVII. Trans Wis Acad Sci Arts Lett 50:141–161
Jones S (2009) Characterisation of cultural, biological and molecular variability of Phoma ligulicola isolates associated with ray blight disease of pyrethrum and chrysanthemum. PhD thesis, University of Tasmania, Australia
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA et al (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948
Lutzoni F, Wagner P, Reeb V, Zoller S (2000) Integrating ambiguously aligned regions of DNA sequences in phylogenetic analyses without violating positional homology. Syst Biol 49:628–651
Müller E, Von Arx JA (1962) Die Gattungen der didymosporen Pyrenomyceten. Beitr Kryptogamenflora Schweiz 11:1–922
Nylander JAA (2004) MrModeltest v.2. Program distributed by the author. Evolutionary Biology Centre. Uppsala University
Oxenham BL (1963) Report of the Plant Pathology Section. Report for the Department of Agriculture. Queensland, Australia
Page RDM (1996) Treeview: an application to display phylogenetic trees on personal computers. Comput Appl Biosci 12:357–358
Peregrine WTH, Watson DRW (1964) Annual report of the plant pathology section. Department of Agriculture, Tanganyika
Pethybridge SJ, Hay F (2001) Influence of Phoma ligulicola on yield, and site factors on disease development, in Tasmanian pyrethrum crops. Australas Plant Pathol 30:17–20
Pethybridge SJ, Wilson CR (1998) Confirmation of ray blight disease of pyrethrum in Australia. Australas Plant Pathol 27:45–48
Pethybridge SJ, Scott JB, Hay FS (2004) Genetic relationships among isolates of Phoma ligulicola from pyrethrum and chrysanthemum based on ITS sequences and its detection by PCR. Australas Plant Pathol 33:173–181
Pethybridge SJ, Esker PD, Dixon P, Hay FS, Groom T et al (2007) Quantifying loss caused by ray blight disease in Tasmanian pyrethrum fields. Plant Dis 91:1116–1121
Pethybridge SJ, Hay FS, Clarkson RA, Groom T, Wilson CR (2008a) Host range of Australian Phoma ligulicola var. inoxydablis isolates from pyrethrum. J Phytopathol 156:506–508
Pethybridge SJ, Hay F, Esker P, Groom T, Wilson C, Nutter FW Jr (2008b) Visual and radiometric assessments for yield losses caused by ray blight in pyrethrum. Crop Sci 48:343–352
Pethybridge SJ, Hay FS, Esker PD, Gent DH, Wilson CR, Nutter FW Jr (2008c) Diseases of pyrethrum in Tasmania: challenges and prospects for management. Plant Dis 92:1260–1272
Punithalingam E (1980) Didymella chrysanthemi. C.M.I. Descriptions of Pathogenic Fungi and Bacteria no. 662:1–3
Quaedvlieg W, Kema GHJ, Groenewald JZ, Verkley GJM, Seifbarghi S et al (2011) Zymoseptoria gen. nov.: a new genus to accommodate Septoria-like species occurring on graminicolous hosts. Persoonia 26:57–69
Rayner RW (1970) A mycological colour chart. Commonwealth Mycological Institute and British Mycological Society, Kew
Rehner SA, Samuels GJ (1994) Taxonomy and phylogeny of Gliocladium analysed from nuclear large subunit ribosomal DNA sequences. Mycol Res 98:625–634
Richardson MJ (1979) An annotated list of seed-borne diseases. C.M.I. Phytopathol Pap 23:1–320
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Simmonds JH (1996) Host index of plant disease in Queensland. Queensland Department of Primary Industries, Brisbane
Stamatakis A (2006) RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22:2688–2690
Stevens FL (1907) The Chrysanthemum ray blight. Bot Gaz 44:241–258
Swofford DL (2003) PAUP Phylogenetic Analysis Using Parsimony (and other methods). Version 4. Sinauer Associates, Sunderland
Tassi F (1900) Novae micromycetum species descriptae et iconibus illustratae. Boll Lab Orto Bot Reale Univ Siena 3:117–132
Van der Aa HA, Noordeloos ME, De Gruyter J (1990) Species concepts in some large genera of the Coelomycetes. Stud Mycol 32:3–19
Vilgalys R, Hester M (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol 172:4238–4246
Voglino P (1902) Sopra una malattia dei crisantemi coltivati. Malpighia 15:329–341
Walker J, Baker KF (1983) The correct binomial for the chrysanthemum ray blight pathogen in relation to its geographical distribution. Trans Br Mycol Soc 80:31–38
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungi ribosomal RNA genes for phylogenetics. In: PCR Protocols. A guide to methods and applications. Academic, San Diego, pp 315–322
Woudenberg JHC, Aveskamp MM, De Gruyter J, Spiers AG, Crous PW (2009) Multiple Didymella teleomorphs are linked to the Phoma clematidina morphotype. Persoonia 22:56–62
Acknowledgments
We would like to thank Dr Frank Hay and Dr Suzanne Jones from the Tasmanian Institute of Agriculture, University of Tasmania, for constructive discussion and information on the isolates. We are very grateful to Drs Johannes Z. Groenewald, Johannes De Gruyter, and Lorenzo Lombard for critical review of this paper. We thank the curators of CBS for providing cultures of the ex-type strains. This project was supported by Botanical Resources Australia—Agricultural Services Pty. Ltd.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vaghefi, N., Pethybridge, S.J., Ford, R. et al. Stagonosporopsis spp. associated with ray blight disease of Asteraceae . Australasian Plant Pathol. 41, 675–686 (2012). https://doi.org/10.1007/s13313-012-0161-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13313-012-0161-3