Abstract
Objective
This network meta-analysis aimed at comparing the efficacy of local control strategies after neoadjuvant chemotherapy in patients with Ewing sarcoma.
Design
Network meta-analysis was used to synthesize direct and indirect evidence in a network of trials that compare multiple interventions and has the potential to rank the competing treatments according to the studied outcome.
Setting
There are three treatment options for local Ewing’s sarcoma after neoadjuvant chemotherapy, namely surgery, radiotherapy and surgery plus radiotherapy (SR).
Participants
Records of 2540 patients from 11 studies were analyzed.
Main outcome measures
Potentially relevant studies were retrieved from PubMed and Embase, and screened according to inclusion and exclusion criteria. Hazard ratios and the associated 95% confidence intervals were used to describe the efficacy of different interventions on 5-year local recurrence rate and 5-year event-free survival rate. Surface under the cumulative ranking curve (SUCRA) was calculated for ranking probabilities of different treatment.
Results
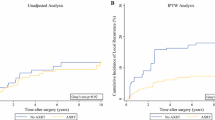
Compared with radiotherapy, surgery had better efficacy [local recurrence, OR (95% CI) 0.48 (0.33–0.87)] and SR had a similar effect as surgery [local recurrence, OR (95% CI) 0.50 (0.29–0.82)]. There were no statistically significant differences between three different local control strategies in 5-year local recurrence rate. SUCRA values suggested that surgery was better than SR for 5-year local recurrence rate (0.79 vs 0.70) and 5-year event free survival rate (0.67 vs 0.50), respectively.
Conclusions
Both surgery and SR were superior to radiotherapy in reducing 5-yer local recurrence of patients with Ewing sarcoma after neoadjuvant chemotherapy. Surgery had higher efficacy than SR on improving the prognosis of patients.
Similar content being viewed by others
References
Werier J, Yao X, Caudrelier JM, Di Primio G, Ghert M, Gupta AA, et al. A systematic review of optimal treatment strategies for localized Ewing’s sarcoma of bone after neoadjuvant chemotherapy. Surg Oncol. 2016;25:16–23
Bolling T, Hardes J, Dirksen U. Management of bone tumours in paediatric oncology. Clin Oncol (R Coll Radiol). 2013;25: 19–26.
Moore DD, Haydon RC. Ewing’s sarcoma of bone. Cancer Treat Res. 2014;162: 93–115.
Falk S, Alpert M. Five-year survival of patients with Ewing’s sarcoma. Surg Gynecol Obstet. 1967;124: 319–24.
Pretz JL, Barysauskas CM, George S, Hornick JL, Raut CP, Chen YE, et al. Localized adult ewing sarcoma: Favorable outcomes with alternating vincristine, doxorubicin, cyclophosphamide, and ifosfamide, etoposide (VDC/IE)-based multimodality therapy. Oncologist. 2017;22: 1265–70.
Wardelmann E, Haas R, Bovée J, Terrier P, Lazar A, Messiou C, et al. Evaluation of Response After Neoadjuvant Treatment in Soft Tissue Sarcomas; the European Organization for Research and Treatment of Cancer-Soft Tissue and Bone Sarcoma Group (EORTC-STBSG) Recommendations for Pathological Examination and Reporting. Eur J Cancer. 2016;53: 84–95.
DuBois SG, Krailo MD, Gebhardt MC, Donaldson SS, Marcus KJ, Dormans J, et al. Comparative evaluation of local control strategies in localized Ewing sarcoma of bone: A report from the Children’s Oncology Group. Cancer. 2015;121: 467–75.
Donati D, Yin J, Di Bella C, Colangeli M, Bacci G, Ferrari S, et al. Local and distant control in non-metastatic pelvic Ewing’s sarcoma patients. J Surg Oncol. 2007;96: 19–25.
Ahmed SK, Randall RL, DuBois SG, Harmsen WS, Krailo M, Marcus KJ, et al. Identification of patients with localized Ewing sarcoma at higher risk for local failure: A report from the Children’s oncology group. Int J Radiat Oncol Biol Phys. 2017;99: 1286–94.
Carrie C. Nonmetastatic Pelvic Ewing Sarcoma: Report of the French Society of Pediatric Oncology. Med Pediatr Oncol. 1999; 33:444–9
De Marco S, Pollera CF, Cognetti F. Nephroblastoma in the adult. Med Pediatr Oncol. 1999;33: 497–9.
Zogopoulos G, Teskey L, Sung L, Dix D, Grant R, Greenberg ML, et al. Ewing sarcoma: Favourable results with combined modality therapy and conservative use of radiotherapy. Pediatr Blood Cancer. 2004;43: 35–9.
Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. British Med J. 2011;343: d5928.
Slim K, Nini E, Forestier D, Kwiatkowski F, Panis Y, Chipponi J. Methodological index for non-randomized studies (minors): Development and validation of a new instrument. ANZ J Surg. 2003;73: 712–6.
Ahmed SK, Robinson SI, Arndt CAS, Petersen IA, Haddock MG, Rose PS, et al. Pelvis Ewing sarcoma: Local control and survival in the modern era. Pediatr Blood Cancer. 2017;64: e26504.
Laitinen M, Parry M, Albergo JI, Jeys L, Sumathi V, Grimer R. Outcome of pelvic bone sarcomas in children. J Pediatr Orthop. 2016;38: 537–42.
Grevener K, Haveman LM, Ranft A, van den Berg H, Jung S, Ladenstein R, et al. Management and outcome of Ewing sarcoma of the head and neck. Pediatr Blood Cancer. 2016;63: 604–10.
Bacci G, Palmerini E, Staals EL, Longhi A, Barbieri E, Alberghini M, et al. Ewing’s sarcoma family tumors of the humerus: Outcome of patients treated with radiotherapy, surgery or surgery and adjuvant radiotherapy. Radiother Oncol. 2009;93: 383–7.
Yock TI, Krailo M, Fryer CJ, Donaldson SS, Miser JS, Chen Z, et al. Local control in pelvic Ewing sarcoma: analysis from INT-0091-A report from the Children’s Oncology Group. J Clin Oncol. 2006;24: 3838–43.
Bacci G, Longhi A, Briccoli A, Bertoni F, Versari M, Picci P. The role of surgical margins in treatment of Ewing’s sarcoma family tumors: Experience of a single institution with 512 patients treated with adjuvant and neoadjuvant chemotherapy. Int J Radiat Oncol Biol Phys. 2006;65: 766–72.
Shankar AG. Local therapy and other factors Infuencing site of relapse in patients with localised Ewing’s sarcoma. Eue J Cancer. 1999;35: 1698–1704.
Sokolov T, Stoyanova A, Mumdjiev I, Mihova A. Comparison of two treatment approaches to localized Ewing’s sarcoma. Ortopediya i Travmatologiya. 2000;36: 509–15.
Shankar AG, Pinkerton CR, Atra A, Ashley S, Lewis I, Spooner D, et al. Local therapy and other factors influencing site of relapse in patients with localised Ewing’s sarcoma. United Kingdom Children’s Cancer Study Group (UKCCSG). Eur J Cancer. 1999;35: 1698–704.
Kawai A, Healey JH, Boland PJ, Lin PP, Huvos AG, Meyers PA. Prognostic factors for patients with sarcomas of the pelvic bones. Cancer. 1998;82: 851–9.
Laitinen M, Parry M, Albergo JI, Jeys L, Sumathi V, Grimer R. Outcome of pelvic bone sarcomas in children. J Pediatr Orthop. 2018;38: 537–42.
O’Sullivan B, Davis AM, Turcotte R, Bell R, Catton C, Chabot P, et al. Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: A randomised trial. Lancet. 2002;359:2235–41
Foulon S, Brennan B, Gaspar N, Dirksen U, Jeys L, Cassoni A, et al. Can postoperative radiotherapy be omitted in localised standard-risk Ewing sarcoma? An observational study of the Euro-EWING group. Eur J Cancer. 2016;61: 128–36.
Bacci G, Forni C, Longhi A, Ferrari S, Donati D, De Paolis M, et al. Long-term outcome for patients with non-metastatic Ewing’s sarcoma treated with adjuvant and neoadjuvant chemotherapies. 402 patients treated at Rizzoli between 1972 and 1992. Eur J Cancer. 2004; 40:73–83.
Author information
Authors and Affiliations
Contributions
HZ,YL,XX: substantial contribution to the conception and design of the work; HZ,SZ,YX: acquisition, analysis, and interpretation of the data; HZ: drafting of the manuscript; TF: revising the manuscript critically. All authors have read and approved the final article.
Corresponding author
Rights and permissions
About this article
Cite this article
Zhu, H., Li, Y., Xie, X. et al. Efficacy of Local Control Strategies for Ewing Sarcoma After Neoadjuvant Chemotherapy: A Network Meta-analysis. Indian Pediatr 57, 527–532 (2020). https://doi.org/10.1007/s13312-020-1850-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13312-020-1850-6