Abstract
Objective
To describe the demographic, clinical, laboratory and bacteriological profile of children with diagnosis of typhoid fever over a six-year period.
Methods
Case record analysis of hospitalized children (≤5 y) with culture positive typhoid fever.
Results
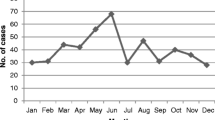
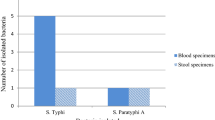
Blood culture was positive in 100 (61%) of 166 suspected cases, with 78 isolates of Salmonella Typhi and 22 Salmonella Paratyphi A. Only 12 children were aged below two years. Hepatomegaly (32), splenomegaly (44), eosinopenia (42), positive widal (15, 21.1%) and positive Typhidot IgM (18, 28.1%) were not consistently observed. High susceptibility to Ampicillin, Chloramphenicol, Cotrimoxazole (87, 89, and 94, isolates, respectively), 100% susceptibility to third generation cephalosporins and Azithromycin, and high resistance to Nalidixic Acid [(S. Typhi 48 (61.5%)], S. Paratyphi A 16 (72.7%)) were observed.
Conclusion
We observed a high isolation rate of salmonella in blood culture, despite prior use of antibiotics. Most salmonella isolates were susceptible in vitro to standard drugs, except nalidixic acid.
Similar content being viewed by others
References
Mogasale V, Maskery B, Ochiai RL, Lee JS, Mogasale VV, Ramani E, et al. Burden of typhoid fever in low-income and middle-income countries: A systematic literature-based update with risk-factor adjustment. Lancet Glob Health. 2014;2:e570–80.
World Health Organization. Typhoid Vaccines: WHO Position Paper — March 2018. Weekly Epidemiological Record. 2018;13:153–72.
Meiring JE, Gibani M, Basnyat B, Bentsi-Enchill AD, Clemens J, Darton TC, et al. The Typhoid Vaccine Acceleration Consortium (TyVAC): Vaccine Effectiveness Study Designs: Accelerating the introduction of typhoid conjugate vaccines and reducing the global burden of enteric fever. Report from a Meeting held on 26–27 October 2016, Oxford, UK. Vaccine. 2017;35:5081–8.
Ochiai RL, Acosta CJ, Danovaro-Holliday M, Baiqing D, Bhattacharya SK, Agtini MD, et al. A study of typhoid fever in five Asian countries: Disease burden and implications for controls. Bull World Health Organ. 2008;86:260–8.
Feasey NA, Gaskell K, Wong V, Msefula C, Selemani G, Kumwenda S, et al. Rapid emergence of multidrug resistant, H58-lineage Salmonella typhi in Blantyre, Malawi. PLoS Negl Trop Dis. 2015;9:e0003748.
Parry CM, Wijedoru L, Arjyal A, Baker S. The utility of diagnostic tests for enteric fever in endemic locations. Expert Rev Anti Infect Ther. 2011;9:711–25.
Sanjeev H, Nayak S, Pai AKB, Rai R, Karnaker V, Ganesh HR. A systematic evaluation of Rapid Dot-EIA, blood culture and Widal test in the diagnosis of typhoid fever. Nitte University J Health Science. 2013;3:21–4.
Upadhyay R, Nadkar MY, Muruganathan A, Tiwaskar M, Amarapurkar D, Banka NH, et al. API Recommendations for the Management of Typhoid Fever. J Assoc Physicians India. 2015;63:77–96.
Ray P, Sharma J, Marak RSK, Garg RK. Predictive efficacy of nalidixic acid resistance as a marker of fluoroquinolone resistance in Salmonella enterica var Typhi. Indian J Med Res. 2006;124:105–8.
Nair S, Ashton P, Doumith M, Connell S, Painset A, Mwaigwisya S, et al. WGS for surveillance of antimicrobial resistance: A pilot study to detect the prevalence and mechanism of resistance to azithromycin in a UK population of non-typhoidal Salmonella. J Antimicrob Chemotherap. 2016;71:3400–8.
Ganesh R, Janakiraman L, Vasanthi T, Sathiyasekeran, M. Profile of typhoid fever in children from a tertiary care hospital in Chennai-South India. Indian J Pediatr. 2010;77:1089–92.
Wong VK, Baker S, Pickard DJ, Parkhill J, Page AJ, Feasey NA, et al. Phylogeographical analysis of the dominant multidrug-resistant H58 clade of Salmonella Typhi identifies inter-and intracontinental transmission events. Nature Genet. 2015;47:632.
Kuijpers LM, Phe T, Veng CH, Lim K, Ieng S, Kham C, et al. The clinical and microbiological characteristics of enteric fever in Cambodia, 2008–2015. PLoS Negl Trop Dis. 2017;11:e0005964.
Hendriksen RS, Leekitcharoenphon P, Lukjancenko O, Lukwesa-Musyani C, Tambatamba B, Mwaba J, et al. Genomic signature of multidrug-resistant Salmonella enterica serovar Typhi isolates related to a massive outbreak in Zambia between 2010 and 2012. J Clin Microbiol. 2015;53:262–72.
Klemm EJ, Shakoor S, Page AJ, Qamar FN, Judge K, Saeed DK, et al. Emergence of an extensively drug-resistant Salmonella enterica serovar Typhi clone harboring a promiscuous plasmid encoding resistance to fluoroquinolones and third generation cephalosporins. MBio. 2018; doi: https://doi.org/10.1128/mBio.00105-18 2018;9:e00105-18.
Funding
Funding: None
Author information
Authors and Affiliations
Contributions
Contributors: AJC: conceived this study; AJC, PSN, MS: contributed to the design of study; SC,MG: helped with data collection and management; AJC,SC: contributed to analysis and manuscript writing. All authors approved the manuscript before submission
Corresponding author
Ethics declarations
Competing interest: None stated.
Rights and permissions
About this article
Cite this article
Chitkara, A.J., Chitkara, S., Narang, P.S. et al. Clinico-Bacteriological Profile of Typhoid Fever in a Private Sector Hospital in New Delhi. Indian Pediatr 56, 1033–1036 (2019). https://doi.org/10.1007/s13312-019-1686-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13312-019-1686-0