Abstract
Objective
Vasoactive-inotropic Score (VIS) was developed to quantify the amount of inotropic support provided in the postoperative period. We investigated the predictive value of (VIS) for mortality in neonates with congenital heart disease (CHD).
Study design
Prospective cohort.
Patients
119 newborns who underwent cardiac surgery.
Setting
Tertiary NICU-CHD center of Ankara from November 2016 to January 2019.
Intervention/Measurement
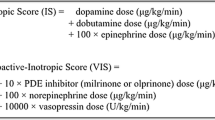
VIS values were calculated by a standard formula for the first 72 postoperative hours, and the maximum score was recorded.
Primary outcomes
Duration of mechanical ventilation, NICU length of stay, and mortality.
Results
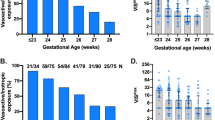
At surgery, the median (IQR) age was 15 d (9–31). The patients were divided into two groups according to mortality; Group 1 (Non-survivors) (n=36) and Group 2 (Survivors) (n=83). Higher VIS score was correlated to longer duration of mechanical ventilation (P=0.009, r=0.33), and was higher among patients who died (P=0.003). Area under the curve (AUC) was 0,83 (P<.001, CI: 95% 0.7–0.9) for VIS to identify mortality. At a cut-off value of 15.5, sensitivity and negative predictive values of VIS for mortality were 73.6% and 85.3%, respectively. The higher VIS (>15.5) was independently associated with increased odds for mortality (OR: 8.1, 95% CI: 1.8–35.7, P=0.005).
Conclusions
In newborns with CHD, a higher VIS within 72 hours after cardiac surgery is associated with increased duration of mechanical ventilation, and mortality. VIS may be useful for prediction of mortality at early postoperative period.
Similar content being viewed by others
References
Cho SY, Oh JH, Lee JH, Lee JY, Lee SJ, Han JW, et al. Recent incidence of congenital heart disease in neonatal care unit of secondary medical center: A single center study. Korean J Pediatr. 2012; 55:232–7.
Båpinar O, Karaaslan S, Oran B, Baysal T, Elmaci AM, Yorulmaz A. Prevalence and distribution of children with congenital heart diseases in the central Anatolian region, Turkey. Turk J Pediatr. 2006: 48:237–43.
Gilboa SM, Salemi JL, Nembhard WN, Fixler DE, Correa A. Mortality resulting from congenital heart disease among children and adults in the United States, 1999 to 2006. Circulation. 2010;122:2254–63.
Saxena A. Congenital heart disease in India: A status report. Indian Pediatr. 2018;55:1075–82.
Tchervenkov CI, Jacobs JP, Bernier PL, Stellin G. The improvement of care for paediatric and congenital cardiac disease across the World: A challenge for the World Society for Pediatric and Congenital Heart Surgery. Cardiol Young. 2008;18:63–9.
Dorfman AT, Marino BS, Wernovsky G, Tabbutt S, Ravishankar C, Godinez RI, et al. Critical heart disease in the neonate: Presentation and outcome at a tertiary care center. Pediatr Crit Care Med. 2008;9:193–202.
Jenkins KJ, Gauvreau K, Newburger JW, Spray TL, Moller JH, Iezzoni LI. Consensus-based method for risk adjustment for surgery for congenital heart disease. J Thorac Cardiovasc Surg. 2002;123:110–8.
Seear MD, Scarfe JC, LeBlanc JG. Predicting major adverse events after cardiac surgery in children. Pediatr Crit Care Med. 2008; 9:606–11.
Hoffman TM, Wernovsky G, Atz AM, Kulik TJ, Nelson DP, Chang AC, et al. Efficacy and safety of milrinone in preventing low cardiac output syndrome in infants and children after corrective surgery for congenital heart disease. Circulation. 2003;107:996–1002.
Richardson DK, Corcoran JD, Escobar GJ, Lee M SK. SNAP-II and SNAPPE-II: Simplified newborn illness severity and mortality risk scores. J Pediatr. 2001;138:92–100.
Lacour-Gayet F, Clarke D, Jacobs J, Comas J, Daebritz S, Daenen W, et al. The Aristotle score: a complexity-adjusted method to evaluate surgical results. Eur J Cardiothorac Surg. 2004;25:911–24.
Joshi SS, Anthony G, Manasa D, Ashwini T, Jagadeesh AM, Borde DP, et al. Predicting mortality after congenital heart surgeries: Evaluation of the aristotle and risk adjustement in congenital heart surgery-1 risk prediction scoring systems: A retrospective single center analysis of 1150 patients. Ann Card Anaesth. 2014;17:266–70.
Wernovsky G, Wypij D, Jonas RA, Mayer JEJ, Hanley FL, Hickey PR, et al. Postoperative course and hemodynamic profile after the arterial switch operation in neonates and infants. A comparison of low-flow cardiopulmonary bypass and circulatory arrest. Circulation. 1995; 92:2226–35.
Gaies MG, Gurney JG, Yen AH, Napoli ML, Gajarski RJ, Ohye RG, et al. Vasoactive-inotropic score as a predictor of morbidity and mortality in infants after cardiopulmonary bypass. Pediatr Crit Care Med. 2010;11:234–8.
Butts RJ, Scheurer MA, Atz AM, Zyblewski SC, Hulsey TC, Bradley SM, et al. Comparison of maximum vasoactive inotropic score and low cardiac output syndrome as markers of early postoperative outcomes after neonatal cardiac surgery. Pediatr Cardiol. 2012;33:633–8.
Davidson J, Tong S, Hancock H, Hauck A, da Cruz E, Kaufman J. Prospective validation of the vasoactive-inotropic score and correlation to short-term outcomes in neonates and infants after cardiothoracic surgery. Intensive Care Med. 2012;38:1184–90.
Reid S, Bajuk B, Lui K, Sullivan EA; NSW and ACT Neonatal Intensive Care Units Audit Group, PSN. Comparing CRIB-II and SNAPPE-II as mortality predictors for very preterm infants. J Paediatr Child Health. 2015;51:524–8.
Jain S, Bansal A. SNAPPE II score for predicting mortality in a level II neonatal intensive care Unit. Dicle Med J. 2009;36:333–5.
O’Brien SM, Jacobs JP, Clarke DR, Maruszewski B, Jacobs ML, Walters HL 3rd, et al. Accuracy of the aristotle basic complexity score for classifying the mortality and morbidity potential of congenital heart surgery operations. Ann Thorac Surg. 2007;84:2027–37.
Erek E, Yýlmaz B, Kaya M, Onan ÝS, Þen O, Öz K. et al. Analysis of results according to the Aristotle scoring system in congenital heart surgery. Turkish J Thoracic Cardiovascular Surgery. 2014;22:509–16.
Kumar M, Sharma R, Sethi SK, Bazaz S, Sharma P, Bhan A, et al. Vasoactive inotrope score as a tool for clinical care in children post cardiac surgery. Indian J Crit Care Med. 2014;18:653–8.
Sanil Y, Aggarwal S. Vasoactive-inotropic score after pediatric heart transplant: A marker of adverse outcome. Pediatr Transplant. 2013;17:567–72.
Yamazaki Y, Oba K, Matsui Y, Morimoto Y. Vasoactive - inotropic score as a predictor of morbidity and mortality in adults after cardiac surgery with cardiopulmonary bypass. J Anesth. 2018;32:167–73.
Cashen K, Costello JM, Grimaldi LM, Narayana Gowda KM, Moser EAS, Piggott KD, et al. Multicenter validation of the vasoactiveventilation-renal score as a predictor of prolonged mechanical ventilation after neonatal cardiac surgery. Pediatr Crit Care Med. 2018;19:1015–23.
Jacobs JP, Mayer JE Jr, Pasquali SK, Hill KD, Overman DM, St Louis JD, et al. The Society of Thoracic Surgeons Congenital Heart Surgery Database: 2018 Update on Outcomes and Quality. Ann Thorac Surg. 2018;105:680–9.
Funding
Funding: None.
Author information
Authors and Affiliations
Contributions
Contributors: DD, IT: concept and designed the study, analyzed data and drafted the manuscript; UAO, MM, Rç, SA, HA, ST: collected the data and helped in data analysis; IT, DD: supervised final manuscript.
Corresponding author
Additional information
Competing Interest: None stated.
Rights and permissions
About this article
Cite this article
Dilli, D., Akduman, H., Orun, U.A. et al. Predictive Value of Vasoactive-inotropic Score for Mortality in Newborns Undergoing Cardiac Surgery. Indian Pediatr 56, 735–740 (2019). https://doi.org/10.1007/s13312-019-1639-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13312-019-1639-7