Abstract
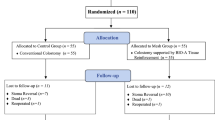
Loop ileostomy (LI) is a widely used temporary stoma technique. Reversal of LI is generally considered a minor and safe procedure, with very low short-term postoperative mortality and morbidity rates. Complications include incisional hernia (IH), carrying a high probability of surgical repair. Clinical measures to reduce the IH rate warrant consideration. Recent researches suggest the use of a prophylactic non-absorbable mesh to reduce IH rate; however, surgeons are reluctant to implant a permanent foreign material in contaminated operative fields, because of a higher risk of mesh-related complications, infection, seroma, and pain. The aim of the present study is to assess feasibility, potential benefits, and safety of a prophylactic biosynthetic mesh placed during LI reversal. From January 2016 to December 2018, 26 consecutive patients underwent LI reversal positioning a resorbable biosynthetic mesh in an on-lay position [mesh group (MG)]. The mesh used was a GORE BIO-A tissue reinforcement, a biosynthetic mesh composed of a bioabsorbable polyglycolide—trimethylene carbonate copolymer. The MG was matched with 58 patients [control group (CG)], undergoing LI reversal without mesh placement from January 2013 to December 2018. To detect IH, abdominal wall was studied according to clinical and ultrasonographic criteria. Primary endpoint was IH rate on LI site, at 6 and 12 months after stomal reversal. Secondary endpoints included incidence of wound events. Thirty-day morbidity was classified according to Clavien–Dindo score; mortality and length of hospital stay were also collected. Mean follow-up was 15.4 ± 2.3 months (range 12.4–22.0) for MG vs 37.2 ± 26.9 (range 24.9–49.7) for. CG. At 1 year of follow-up, IH rate was lower in MG (n = 1/26 [3.8%]) vs CG (n = 19/58 [32.7%]; P < 0.05). A clinically evident IH was less frequent in MG (n = 0 [0%]) vs CG (n = 13 [68%]; P < 0.05). A radiologic IH was less frequent in MG (n = 1 [3%]) vs CG (n = 6 [31%]; P < 0.05). Stoma site hernia was repaired in 9/19 patients (47%) in CG; no patient of MG has hernia repaired. Incarcerated IH was observed in one patient of CG. No postoperative mortality was reported. Overall postoperative morbidity showed no difference comparing MG and MG (n = 5 [17%] vs n = 15 [19%], respectively; P > 0.05). Surgical site infections (SSI) were treated with antibiotic therapy, no debridement was necessary. Seroma occurred in two patients, one for each group. No statistically significant difference for surgical outcomes was found between the two groups at 30 days. Early results of the present study suggest that an on-lay prophylactic placement of GORE BIO-A tissue reinforcement might lower IH rate at LI site. The procedure seems to be safe and effective, even long-term results and further studies are needed.
Similar content being viewed by others
References
Bhangu A, Nepogodiev D, Futaba K (2012) Systematic review and meta-analysis of the incidence of incisional hernia at the site of stoma closure. World J Surg 36:973.e83
Pakkastie TE, Ovaska JT, Pekkala ES, Luukkonen PE, Jarvinen HJ (1997) A randomised study of colostomies in low colorectal anastomoses. Eur J Surg 163:929–933
Fielding LP, Stewart-Brown S, Hittinger R, Blesovsky L (1984) Covering stoma for elective anterior resection of the rectum: an outmoded operation? Am J Surg 147:524–530
Edwards DP, Leppington-Clarke A, Sexton R et al (2001) Stomarelated complications are more frequent after transverse colostomy than loop ileostomy: a prospective randomized clinical trial. Br J Surg 88:360–363
Kaidar-Person O, Person B, Wexner SD (2005) Complications of construction and closure of temporary loop ileostomy. J Am Coll Surg 201:759–773
Bakx R, Busch OR, Bemelman WA et al (2004) Morbidity of temporary loop ileostomies. Dig Surg 21:277–281
Chow A, Tilney HS, Paraskeva P, Jeyarajah S, Zacharakis E, Purkayastha S (2009) The morbidity surrounding reversal of defunctioning ileostomies: a systematic review of 48 studies including 6107 cases. Int J Colorectal Dis 24:711–723
Tilney HS, Sains PS, Lovegrove RE et al (2007) Comparison of outcomes following ileostomy versus colostomy for defunctioning colorectal anastomoses. World J Surg 31:1142–1151
Rondelli F, Reboldi P, Rulli A et al (2009) Loop ileostomy versusloop colostomy for fecal diversion after colorectal or coloanal anastomosis: a meta-analysis. Int J Colorectal Dis 24:479–488
Guenaga KF, Lustosa SA, Saad SS et al (2007) Ileostomy or colostomy for temporary decompression of colorectal anastomosis. Cochrane Database Syst Rev 1:CD004647
van Ramshorst GH, Nieuwenhuizen J, Hop WC, Arends P, Boom J, Jeekel J et al (2010) Abdominal wound dehiscence in adults: development and validation of a risk model. World J Surg 34:20–27
Arumugam PJ, Bevan L, Macdonald L, Watkins AJ, Morgan AR, Beynon J et al (2003) A prospective audit of stomas—analysis of risk factors and complications and their management. Colorectal Dis 5:49–52
Bell C, Asolati M, Hamilton E et al (2005) A comparison of complications associated with colostomy reversal versus ileostomy reversal. Am J Surg 190(5):717–720
Berry DP, Scholefield JH (1997) Closure of loop ileostomy. Br J Surg 84(4):524
Muysoms FE, Detry O, Vierendeels T et al (2016) Prevention of incisional hernias by prophylactic mesh-augmented reinforcement of midline laparotomies for abdominal aortic aneurysm treatment: a randomized controlled trial. Ann Surg 263:638–645
Borab ZM, Shakir S, Lanni MA et al (2017) Does prophylactic mesh placement in elective, midline laparotomy reduce the incidence of incisional hernia? A systematic review and metaanalysis. Surgery 161:1149–1163
Loganathan A, Ainslie WG, Wedgwood KR (2010) Initial evaluation of Permacol bioprosthesis for the repair of complex incisional and parastomal hernias. Surgeon 8:202–205
Slater NJ, Hansson BM, Buyne OR, Hendriks T, Bleichrodt RP (2011) Repair of parastomal hernias with biologic grafts: a systematic review. J Gastrointest Surg 15:1252–1258
Brosi P et al (2018) Prophylactic intraperitoneal onlay mesh reinforcement reduces the risk of incisional hernia, 2-year results of a randomized clinical trial. World J Surg 42(6):1687–1694
Todd B, New CDC (2017) Guideline for the prevention of surgical site infection. Am J Nurs 117(8):17
van Riet M, De Vos-Van-Steenwijk PJ, Bonjer HJ et al (2004) Incisional hernia after repair of wound dehiscence: incidence andrisk factors. Am Surg 70:281–286
Itatsu K, Yokoyama Y, Sugawara G et al (2014) Incidence of and risk factors for incisional hernia after abdominal surgery. Br J Surg 101:1439–1447
Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, Schulick RD, de Santibañes E, Pekolj J, Slankamenac K, Bassi C, Graf R, Vonlanthen R, Padbury R, Cameron JL, Makuuchi M (2009) The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg 250(2):187–196
Timmermans L, de Goede B, Eker HH et al (2013) Meta-analysis of primary mesh augmentation as prophylactic measure to prevent incisional hernia. Dig Surg 30:401–409
Wang X-C, Zhang D, Yang Z-X et al (2017) Mesh reinforcement for the prevention of incisional hernia formation: a systematic review and meta-analysis of randomized controlled trials. J Surg Res 209:17–29
Jairam AP et al (2017) Prevention of incisional hernia with prophylactic onlay and sublay mesh reinforcement versus primary suture only in midline laparotomies (PRIMA): 2-year follow-up of a multicentre, double-blind, randomised controlled trial. Lancet 390(10094):567–576
Carbonell AM (2013) Safety of prosthetic mesh hernia repair in contaminated fields. Surg Clin N Am 93(5):1227–1239
Choi JJ, Palaniappa NC, Dallas KB, Rudich TB, Colon MJ, Divino CM (2012) Use of mesh during ventral hernia repair inclean-contaminated and dontaminated cases. Ann Surg 255:176–180
Smart NJ, Bloor S (2012) Durability of biologic implants for use in hernia repair: a review. Surg Innov 19:221
King KS, Albino FP, Bhanot P (2014) Biologic mesh for abdominal wall reconstruction. Chronic Wound Care Manag Res 1:57–65
Smart NJ, Bryan N, Hunt JA, Daniels IR (2014) Porcine dermis implants in soft-tissue reconstruction: current status. Biologics 8:83–90
Montgomery A (2013) The battle between biological and synthetic meshes in ventral hernia repair. Hernia 17:3–11
Shankaran V, Weber DJ, Reed L, Luchette FA (2011) A review of available prosthetics for ventral hernia repair. Ann Surg 253:16
Atema JJ, de Vries FE (2016) Boermeester MA systematic review and meta-analysis of the repair of potentially contaminated and contaminated abdominal wall defects. Am J Surg. 212(5):982–995.e1
Rosen MJ, Bauer JJ, Harmaty M, Carbonell AM, Cobb WS, Matthews B, Goldblatt MI, Selzer DJ, Poulose BK, Hansson BM, Rosman C, Chao JJ, Jacobsen GR (2017) Multicenter, prospective, longitudinal study of the recurrence, surgical site infection, and quality of life after contaminated ventral hernia repair using biosynthetic absorbable mesh: the COBRA study. Ann Surg. 265(1):205–211
Borab ZM, Shakir S, Lanni MA, Tecce MG, MacDonald J, Hope HW (2017) Does prophylactic mesh placement in elective, midline laparotomy reduce the incidence of incisional hernia? A systematic review and meta-analysis Surgery Surgery 161(4):1149–1163
Guzman-Valdivia G (2008) Incisional hernia at the site of a stoma. Hernia 12:471–474
Pokorny H, Herkner H, Jakesz R et al (2005) Mortality and complications after stoma closure. Arch Surg 140:956–960
van de Pavoordt HD, Fazio VW, Jagelman DG et al (1987) The outcome of loop ileostomy closure in 293 cases. Int J Colorectal Dis 2:214–217
Llaguna OH, Avgerinos DV, Nagda P et al (2011) Does prophylactic biologic mesh placement protect against the development of incisional hernia in high-risk patients? World J Surg 35:1651–1655
Öberg S, Andresen K, Klausen TW (2018) Rosenberg J Chronic pain after mesh versus nonmesh repair of inguinal hernias: a systematic review and a network meta-analysis of randomized controlled trials. Surgery. 163(5):1151–1159
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Francesco Pizza, Dario D’Antonio, Michele Arcopinto, Chiara Dell’Isola, and Alberto Marvaso declare that they have no conflict of interest.
Ethical approval
Research involving human participants registered in Mendeley Data https://www.doi.org/10.17632/w3ybn7bh22.2.
Informed consent
Informed consent was given to all patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pizza, F., D’Antonio, D., Arcopinto, M. et al. Safety and efficacy of prophylactic resorbable biosynthetic mesh in loop-ileostomy reversal: a case–control study. Updates Surg 72, 103–108 (2020). https://doi.org/10.1007/s13304-020-00702-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13304-020-00702-z