Abstract
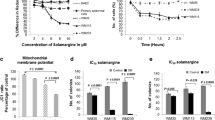
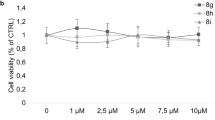
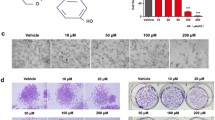
Recent evidence suggests that dimethylfumarate (DMF), known as a highly potent anti-psoriatic agent, might have anti-tumorigenic properties in melanoma. It has recently been demonstrated that DMF inhibits melanoma proliferation by apoptosis and cell cycle inhibition and therefore inhibits melanoma metastasis. Nonetheless, the underlying mechanisms remain to be evaluated. To elucidate the effects of DMF on melanoma cell lines (A375, SK-Mel), we first performed cytotoxicity assays. No significant lactatedehydogenase (LDH) release could be found. In further analysis, we showed that DMF suppresses melanoma cell proliferation in a concentration-dependent manner. To examine whether these effects are conveyed by apoptotic mechanisms, we studied the amount of apoptotic nucleosomes and caspase 3/7 activity using ELISA analysis. Significant apoptosis was induced by DMF in both cell lines, and this could be paralleled with bcl-2 downregulation and PARP-1 cleavage. We also performed cell cycle analysis and found that DMF induced concentration-dependent arrests of G0/G1 as well as G2/M. To examine the underlying mechanisms of cell cycle arrest, we analyzed the expression profiles of important cell cycle regulator proteins such as p53, p21, cyclins A, B1, and D1, and CDKs 3, 4, and 6. Interestingly, DMF induced p53 and p21 yet inhibited cyclin B1 expression in a concentration-dependent manner. Other cell cycle regulators were not influenced by DMF. The knockdown of DMF induced p53 via siRNA led to significantly reduced apoptosis but had no influence on cell cycle arrest. We examined the adhesion of melanoma cells on lymphendothelial cells during DMF treatment and found a significant reduction in interaction. These data provide evidence that DMF inhibits melanoma proliferation by reinduction of important cell cycle inhibitors leading to a concentration-dependent G0/G1 or G2/M cell cycle arrest and induction of apoptosis via downregulation of bcl-2 and induction of p53 and PARP-1 cleavage. Hence, DMF might be an interesting agent in the treatment of melanoma and is worth further investigation in vivo.





Similar content being viewed by others
Abbreviations
- DMF:
-
Dimethylfumarate
- BrdU:
-
Bromodesoxyuridine
- LDH:
-
Lactatedehydrogenase
- DMSO:
-
Dimethylsulfoxide
- DLEC:
-
Dermal lymphatic endothelial cells
References
Schweckendiek W. Treatment of psoriasis vulgaris. Med Monatsschr. 1959;13(2):103–4.
Meissner M, Valesky EM, Kippenberger S, Kaufmann R. Dimethyl fumarate—only an anti-psoriatic medication? J Dtsch Dermatol Ges. 2012;10(11):793–801.
Arbiser JL. Fumarate esters as angiogenesis inhibitors: key to action in psoriasis? J Invest Dermatol. 2011;131(6):1189–91.
Loewe R, Valero T, Kremling S, Pratscher B, Kunstfeld R, Pehamberger H, et al. Dimethylfumarate impairs melanoma growth and metastasis. Cancer Res. 2006;66(24):11888–96.
Yamazoe Y, Tsubaki M, Matsuoka H, Satou T, Itoh T, Kusunoki T, et al. Dimethylfumarate inhibits tumor cell invasion and metastasis by suppressing the expression and activities of matrix metalloproteinases in melanoma cells. Cell Biol Int. 2009;33(10):1087–94.
Valero T, Steele S, Neumüller K, Bracher A, Niederleithner H, Pehamberger H, et al. Combination of dacarbazine and dimethylfumarate efficiently reduces melanoma lymph node metastasis. J Invest Dermatol. 2010;130(4):1087–94.
Meissner M, Doll M, Hrgovic I, et al. Suppression of VEGFR2 expression in human endothelial cells by dimethylfumarate treatment: evidence for anti-angiogenic action. J Invest Dermatol. 2011;131(6):1356–64.
CJ O, Park S, Kim JY, Kim HJ, Jeoung NH, Choi YK, et al. Dimethylfumarate attenuates restenosis after acute vascular injury by cell-specific and Nrf2-dependent mechanisms. Redox Biol. 2014;2:855–64.
Valesky EM, Hrgovic I, Doll M, Wang XF, Pinter A, Kleemann J, et al. Dimethylfumarate effectively inhibits lymphangiogenesis via p21 induction and G1 cell cycle arrest. Exp Dermatol. 2015. doi:10.1111/exd.12907.
Kang HJ, Seo HA, Go Y, CJ O, Jeoung NH, Park KG, et al. Dimethylfumarate suppresses adipogenic differentiation in 3T3-L1 preadipocytes through inhibition of STAT3 activity. PLoS One. 2013;8(4):e61411.
Odom RY, Dansby MY, Rollins-Hairston AM, Jackson KM, Kirlin WG. Phytochemical induction of cell cycle arrest by glutathione oxidation and reversal by N-acetylcysteine in human colon carcinoma cells. Nutr Cancer. 2009;61(3):332–9.
Kedinger V, Meulle A, Zounib O, Bonnet ME, Gossart JB, Benoit E, et al. Sticky siRNAs targeting survivin and cyclin B1 exert an antitumoral effect on melanoma subcutaneous xenografts and lung metastases. BMC Cancer. 2013;13:338.
Lee SY, Debnath T, Kim SK, Lim BO. Anti-cancer effect and apoptosis induction of cordycepin through DR3 pathway in the human colonic cancer cell HT-29. Food Chem Toxicol. 2013;60:439–47.
Miyanaga S, Ninomiya I, Tsukada T, Okamoto K, Harada S, Nakanuma S, et al. Concentration-dependent radiosensitizing effect of docetaxel in esophageal squamous cell carcinoma cells. Int J Oncol. 2016;48(2):517–24.
Chipuk JE. BCL-2 proteins: melanoma lives on the edge. Oncoscience. 2015;2(9):729–30.
Tsubaki M, Komai M, Fujimoto S, Itoh T, Imano M, Sakamoto K, et al. Activation of NF-κB by the RANKL/RANK system up-regulates snail and twist expressions and induces epithelial-to-mesenchymal transition in mammary tumor cell lines. J Exp Clin Cancer Res. 2013;32:62.
Treumer F, Zhu K, Gläser R, Mrowietz U. Dimethylfumarate is a potent inducer of apoptosis in human T cells. J Invest Dermatol. 2003;121(6):1383–8.
Anwar M, Aslam HM, Anwar S. PARP inhibitors. Hered Cancer Clin Pract. 2015;13(1):4.
Chevanne M, Zampieri M, Caldini R, Rizzo A, Ciccarone F, Catizone A, et al. Inhibition of PARP activity by PJ-34 leads to growth impairment and cell death associated with aberrant mitotic pattern and nucleolar actin accumulation in M14 melanoma cell line. J Cell Physiol. 2010;222(2):401–10.
Staibano S, Pepe S, Lo Muzio L, Somma P, Mascolo M, Argenziano G, et al. Poly(adenosine diphosphate-ribose) polymerase 1 expression in malignant melanomas from photoexposed areas of the head and neck region. Hum Pathol. 2005;36(7):724–31.
van der Noll R, Marchetti S, Steeghs N, Beijnen JH, Mergui-Roelvink MW, Harms E, et al. Long-term safety and anti-tumour activity of olaparib monotherapy after combination with carboplatin and paclitaxel in patients with advanced breast, ovarian or fallopian tube cancer. Br J Cancer. 2015;113(3):396–402.
Oza AM, Cibula D, Benzaquen AO, Poole C, Mathijssen RH, Sonke GS, et al. Olaparib combined with chemotherapy for recurrent platinum-sensitive ovarian cancer: a randomised phase 2 trial. Lancet Oncol. 2015;16(1):87–97.
Yu J, Zhang L. The transcriptional targets of p53 in apoptosis control. Biochem Biophys Res Commun. 2005;331(3):851–8.
Acknowledgments
The work was sponsored by the Brigitte und Dr. Konstanze Wegener-Stiftung (M.M.); Dr. Kurt & Eva Herrmann Stipendium, 2013 (I.K.); Nachlässe Maria Christine Held und Erika Hecker (E.V.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Additional information
Irina Kaluzki and Igor Hrgovic contributed equally.
Electronic supplementary material
ESM 1
(PDF 113 kb)
Rights and permissions
About this article
Cite this article
Kaluzki, I., Hrgovic, I., Hailemariam-Jahn, T. et al. Dimethylfumarate inhibits melanoma cell proliferation via p21 and p53 induction and bcl-2 and cyclin B1 downregulation. Tumor Biol. 37, 13627–13635 (2016). https://doi.org/10.1007/s13277-016-5285-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-016-5285-6