Abstract
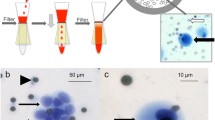
Pancreatic ductal adenocarcinoma (PDAC) is most often diagnosed in a metastatic stage. Circulating tumor cells (CTC) in the blood are hypothesized as the means of systemic dissemination. We aimed to isolate and characterize CTC to evaluate their significance as prognostic markers in PDAC. Blood obtained from healthy donors and patients with PDAC before therapy was filtered with ScreenCell® filtration devices for size-based CTC isolation. Captured cells were analyzed by immunofluorescence for an epithelial to mesenchymal transition (EMT) marker (zinc finger E-box binding homebox 1 (ZEB1)) and an epithelial antigen (cytokeratin (CK)). Molecular analysis of parallel specimens evaluated the KRAS mutation status of the CTC. The survival of each patient after study was recorded. As demonstrated by either cytology or finding of a KRAS mutation, CTC were detected in 18 of 21 patients (86 %) with proven PDAC: 8 out of 10 patients (80 %) with early stage (UICC IIA/IIB) and 10 out of 11 (91 %) with late stage (UICC III/IV) disease. CTC were not found in any of the 10 control patients (p < 0.001). The presence of CTC did not adversely affect median survival: 16 months in CTC-positive (n = 18) vs. 10 months in CTC-negative (n = 3) patients. Neither ZEB1 nor cytological characteristics correlated with overall survival, although ZEB1 was found almost exclusively in CTC of patients with established metastases. Patients with a CTC KRAS mutation (CTC-KRAS mut) had a substantially better survival, 19.4 vs. 7.4 months than patients with wild type KRAS (p = 0.015). With ScreenCell filtration, CTC are commonly found in PDAC (86 %). Molecular and genetic characterization, including mutations such as KRAS, may prove useful for prognosis.

Similar content being viewed by others
References
Jemal A, Siegel R, Xu J, Ward E. Cancer statistics 2010. CA Cancer J Clin. 2010;60:277–300.
Conroy T, Desseigne F, Ychou M, Bouché O, Guimbaud R, Bécouarn Y, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011;364:1817–25.
Valsangkar NP, Bush DM, Michaelson JS, Ferrone CR, Wargo JA, Lillemoe KD, et al. N0/N1, PNL, or LNR? The effect of lymph node number on accurate survival prediction in pancreatic ductal adenocarcinoma. J Gastrointest Surg. 2013;17:257–66.
Khoja L, Backen A, Sloane R, Menasce L, Ryder D, Krebs M, et al. A pilot study to explore circulating tumour cells in pancreatic cancer as a novel biomarker. Br J Cancer. 2012;106:508–16.
Wellner U, Schubert J, Burk UC, Schmalhofer O, Zhu F, Sonntag A, et al. The EMT-activator ZEB1 promotes tumorigenicity by repressing stemness-inhibiting microRNAs. Nat Cell Biol. 2009;11:1487–95.
Rhim AD, Mirek ET, Aiello NM, Maitra A, Bailey JM, McAllister F, et al. EMT and dissemination precede pancreatic tumor formation. Cell. 2012;148:349–61.
Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Matera J, Miller MC, et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med. 2004;351:781–91.
Cohen SJ, Punt CJA, Iannotti N, Saidman BH, Sabbath KD, Gabrail NY, et al. Prognostic significance of circulating tumor cells in patients with metastatic colorectal cancer. Ann Oncol. 2009;20:1223–9.
Scher HI, Jia X, de Bono JS, Fleisher M, Pienta KJ, Raghavan D, et al. Circulating tumour cells as prognostic markers in progressive, castration-resistant prostate cancer: a reanalysis of IMMC38 trial data. Lancet Oncol. 2009;10:233–9.
Panteleakou Z, Lembessis P, Sourla A, Pissimissis N, Polyzos A, Deliveliotis C, et al. Detection of circulating tumor cells in prostate cancer patients: methodological pitfalls and clinical relevance. Mol Med. 2009;15:101–14.
Kaifi JT, Kunkel M, Das A, Harouaka RA, Dicker DT, Li G, et al. Circulating tumor cell isolation during resection of colorectal cancer lung and liver metastases: a prospective trial with different detection techniques. Cancer Biol Ther. 2015;16(5):699–708.
Kulemann B, Pitman MB, Liss AS, Valsangkar N, Fernández-Del Castillo C, Lillemoe KD, et al. Circulating tumor cells found in patients with localized and advanced pancreatic cancer. Pancreas. 2015;44(4):547–50.
Steinert G, Schölch S, Niemietz T, Iwata N, García SA, Behrens B, et al. Immune escape and survival mechanisms in circulating tumor cells of colorectal cancer. Cancer Res. 2014;74:1694–704.
Nagrath S, Sequist LV, Maheswaran S, Bell DW, Irimia D, Ulkus L, et al. Isolation of rare circulating tumour cells in cancer patients by microchip technology. Nature. 2007;450:1235–9.
Bidard FC, Huguet F, Louvet C, Mineur L, Bouché O, Chibaudel B, et al. Circulating tumor cells in locally advanced pancreatic adenocarcinoma: the ancillary CirCe 07 study to the LAP 07 trial. Ann Oncol. 2013;24(8):2057–61.
de Albuquerque A, Kubisch I, Breier G, Stamminger G, Fersis N, Eichler A, et al. Multimarker gene analysis of circulating tumor cells in pancreatic cancer patients: a feasibility study. Oncology. 2012;82(1):3–10.
Negin BP, Meropol NJ, Alpaugh RK, Ruth K, McAleer C, Halbherr T, et al. Characterization and prognostic significance of circulating tumor cells in the peripheral blood of patients with metastatic pancreatic cancer. ASCO Meet Abstr. 2010;28:4127.
Kurihara T, Itoi T, Sofuni A, Itokawa F, Tsuchiya T, Tsuji S, et al. Detection of circulating tumor cells in patients with pancreatic cancer: a preliminary result. J Hepatobiliary Pancreat Surg. 2008;15:189–95.
Bobek V, Gurlich R, Eliasova P, Kolostova K. Circulating tumor cells in pancreatic cancer patients: enrichment and cultivation. World J Gastroenterol. 2014;20(45):17163–70.
Almoguera C, Shibata D, Forrester K, Martin J, Arnheim N, Perucho M. Most human carcinomas of the exocrine pancreas contain mutant c-K-ras genes. Cell. 1988;53:549–54.
Pitman MB. In: Bibbo, Wilbur, editors. Pancreas in comprehensive cytopathology. 4th ed. London: Elsevier; 2014. p. 759.
Valsangkar NP, Ingkakul T, Correa-Gallego C, Mino-Kenudson M, Masia R, Lillemoe KD, et al. Survival in ampullary cancer: potential role of different KRAS mutations. Surgery. 2015;157(2):260–8.
Yu M, Bardia A, Wittner BS, Stott SL, Smas ME, Ting DT, et al. Circulating breast tumor cells exhibit dynamic changes in epithelial and mesenchymal composition. Science. 2013;339:580–4.
Gorges TM, Tinhofer I, Drosch M, Röse L, Zollner TM, Krahn T, et al. Circulating tumour cells escape from EpCAM-based detection due to epithelial-to-mesenchymal transition. BMC Cancer. 2012;12:178.
Bronsert P, Kohler I, Timme S, Kiefer S, Werner M, Schilling O, et al. Prognostic significance of Zinc finger E-box binding homeobox 1 (ZEB1) expression in cancer cells and cancer-associated fibroblasts in pancreatic head cancer. Surgery. 2014;156(1):97–108.
Kwon MJ, Jeon JY, Park HR, Nam ES, Cho SJ, Shin HS, et al. Low frequency of KRAS mutation in pancreatic ductal adenocarcinomas in Korean patients and its prognostic value. Pancreas. 2015;44(3):484–92.
Kim ST, do Lim H, Jang KT, Lim T, Lee J, Choi YL, et al. Impact of KRAS mutations on clinical outcomes in pancreatic cancer patients treated with first-line gemcitabine-based chemotherapy. Mol Cancer Ther. 2011;10(10):1993–9.
Franko J, Krasinskas AM, Nikiforova MN, Zarnescu NO, Lee KK, Hughes SJ, et al. Loss of heterozygosity predicts poor survival after resection of pancreatic adenocarcinoma. J Gastrointest Surg. 2008;12(10):1664–72.
Lee J, Jang KT, Ki CS, Lim T, Park YS, Lim HY, et al. Impact of epidermal growth factor receptor (EGFR) kinase mutations, EGFR gene amplifications, and KRAS mutations on survival of pancreatic adenocarcinoma. Cancer. 2007;109(8):1561–9.
da Cunha Santos G, Dhani N, Tu D, Chin K, Ludkovski O, Kamel-Reid S, et al. Molecular predictors of outcome in a phase 3 study of gemcitabine and erlotinib therapy in patients with advanced pancreatic cancer: National Cancer Institute of Canada Clinical Trials Group Study PA.3. Cancer. 2010;116(24):5599–607.
Kullmann F, Hartmann A, Stöhr R, Messmann H, Dollinger MM, Trojan J, et al. KRAS mutation in metastatic pancreatic ductal adenocarcinoma: results of a multicenter phase II study evaluating efficacy of cetuximab plus gemcitabine/oxaliplatin (GEMOXCET) in first-line therapy. Oncology. 2011;81(1):3–8.
Oliveira-Cunha M, Hadfield KD, Siriwardena AK, Newman W. EGFR and KRAS mutational analysis and their correlation to survival in pancreatic and periampullary cancer. Pancreas. 2012;41(3):428–34.
Bournet B, Muscari F, Guimbaud R, Cordelier P, Buscail L. KRAS mutations and their correlation with survival of patients with advanced pancreatic cancer. Pancreas. 2013;42(3):543–4.
Acknowledgments
We thank Silke Hempel for technical assistance in the KRAS-PNA assay.
Author’s contributions
BK designed the study; BK, ASL, JH, and ALW drafted the manuscript; SS performed the experiments and conducted parts of the analysis, PB and MBP performed the histological analysis, TG performed the statistical analysis. All authors critically reviewed the manuscript and approved the final form.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Additional information
Martha B. Pitman and Jens Hoeppner contributed equally to this work.
Rights and permissions
About this article
Cite this article
Kulemann, B., Liss, A.S., Warshaw, A.L. et al. KRAS mutations in pancreatic circulating tumor cells: a pilot study. Tumor Biol. 37, 7547–7554 (2016). https://doi.org/10.1007/s13277-015-4589-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-4589-2