Abstract
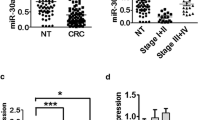
Deregulated microRNA (miRNA) expression has been shown to be involved in the pathogenesis of several types of cancers including colorectal cancer (CRC). Thus, determining miRNA targets of genes that play critical role in the malignant transformation is very important. Here, expression levels of tumor suppressor microtubule-associated tumor suppressor 1 (MTUS1) and its regulatory miRNAs were reported. Predicted and validated targets of MTUS1 gene was determined by a computational approach. Expressions of MTUS1 and miRNAs were determined by using 96.96 Dynamic Array™ integrated fluidic circuit (Fluidigm). As a result, MTUS1 levels were found to be diminished in formalin-fixed, paraffin-embedded (FFPE) tissue samples of CRC patients compared to controls. Also, several of MTUS1 targeting miRNAs were found to be upregulated in CRC samples (miR-373-3p, 183-5p, 142-5p, 200c-3p, 19a-3p, -20a-5p, -181a-5p, -184, -181d-5p, -372-3p, 27b-3p, 98-5p, -let-7i-5p, -let-7d-5p, -let-7g-5p, -let-7b-5p, and -let-7c-5p). Of these miRNAs, miR-135b-5p, -373-3p, 183-5p, 142-5p, 200c-3p, 19a-3p showed marked expression levels. In contrast, expression levels of let-7a-5p, 7e-5p, 7f-5p, hsa-miR-125a-5p, and 125b-5p were found to be downregulated in CRC tissues. Accordingly, some of the overexpressed miRNAs especially the miR-135b-5p, -373-3p, 183-5p, 142-5p, 200c-3p, and 19a-3p may play key roles in CRC pathophysiology through MTUS1. In contrast, let-7a-5p, 7e-5p, 7f-5p, miR-125a-5p, and 125b-5p may play important roles in CRC carcinogenesis independent from the MTUS1. In conclusion, MTUS1 targeting miRNAs may play key roles in the development of CRC by downregulating tumor suppressor MTUS1.



Similar content being viewed by others
References
Geyik E, Igci YZ, Pala E, Suner A, Borazan E, Bozgeyik I, Bayraktar E, Bayraktar R, Ergun S, Cakmak EA, Gokalp A, Arslan A: Investigation of the association between ATP2b4 and ATP5b genes with colorectal cancer. Gene. 2014;540:178-182
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA: Cancer J Clin. 2012;62:10–29.
Centelles JJ: General aspects of colorectal cancer. ISRN oncology 2012;2012
Di Benedetto M, Bieche I, Deshayes F, Vacher S, Nouet S, Collura V, et al. Structural organization and expression of human mtus1, a candidate 8p22 tumor suppressor gene encoding a family of angiotensin ii at2 receptor-interacting proteins, atip. Gene. 2006;380:127–36.
Ding X, Zhang N, Cai Y, Li S, Zheng C, Jin Y, et al. Down-regulation of tumor suppressor mtus1/atip is associated with enhanced proliferation, poor differentiation and poor prognosis in oral tongue squamous cell carcinoma. Mol Oncol. 2012;6:73–80.
Frank B, Bermejo JL, Hemminki K, Sutter C, Wappenschmidt B, Meindl A, et al. Copy number variant in the candidate tumor suppressor gene mtus1 and familial breast cancer risk. Carcinogenesis. 2007;28:1442–5.
Pils D, Horak P, Gleiss A, Sax C, Fabjani G, Moebus VJ, et al. Five genes from chromosomal band 8p22 are significantly down-regulated in ovarian carcinoma. Cancer. 2005;104:2417–29.
Seibold S, Rudroff C, Weber M, Galle J, Wanner C, Marx M. Identification of a new tumor suppressor gene located at chromosome 8p21. 3-22. FASEB J. 2003;17:1180–2.
Ye H, Pungpravat N, Huang B-L, Muzio LL, Mariggio MA, Chen Z, et al. Genomic assessments of the frequent loss of heterozygosity region on 8p21. 3-p22 in head and neck squamous cell carcinoma. Cancer Genet Cytogenet. 2007;176:100–6.
Schetter AJ, Okayama H, Harris CC. The role of micrornas in colorectal cancer. Cancer J (Sudbury, Mass). 2012;18:244.
Chen J, Wang W, Zhang Y, Hu T, Chen Y. The roles of mir-200c in colon cancer and associated molecular mechanisms. Tumor Biol. 2014;35:6475–83.
Hur K, Toiyama Y, Takahashi M, Balaguer F, Nagasaka T, Koike J, et al. Microrna-200c modulates epithelial-to-mesenchymal transition (emt) in human colorectal cancer metastasis. Gut. 2013;62:1315–26.
Song C, Liu LZ, Pei XQ, Liu X, Yang L, Ye F, Xie X, Chen J, Tang H: Mir-200c inhibits breast cancer proliferation by targeting kras. Oncotarget 2015
Gattolliat C-H, Uguen A, Pesson M, Trillet K, Simon B, Doucet L, et al. MicroRNA and targeted mRNA expression profiling analysis in human colorectal adenomas and adenocarcinomas. Eur J Cancer. 2015;51:409–20.
Paraskevopoulou MD, Georgakilas G, Kostoulas N, Vlachos IS, Vergoulis T, Reczko M, et al. Diana-microt web server v5. 0: Service integration into mirna functional analysis workflows. Nucleic Acids Res. 2013;41:W169–73.
Reczko M, Maragkakis M, Alexiou P, Grosse I, Hatzigeorgiou AG. Functional microRNA targets in protein coding sequences. Bioinformatics. 2012;28:771–6.
Lewis BP, Burge CB, Bartel DP. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell. 2005;120:15–20.
Li J-H, Liu S, Zhou H, Qu L-H, Yang J-H: Starbase v2. 0: Decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA interaction networks from large-scale clip-seq data. Nucleic acids research 2013:gkt1248.
Yang J-H, Li J-H, Shao P, Zhou H, Chen Y-Q, Qu L-H. Starbase: a database for exploring microRNA-mRNA interaction maps from argonaute clip-seq and degradome-seq data. Nucleic Acids Res. 2011;39:D202–9.
Betel D, Wilson M, Gabow A, Marks DS, Sander C. The microRNA.org resource: targets and expression. Nucleic Acids Res. 2008;36:D149–53.
Wang X. Mirdb: a microRNA target prediction and functional annotation database with a wiki interface. RNA. 2008;14:1012–7.
Wang X, El Naqa IM. Prediction of both conserved and nonconserved microRNA targets in animals. Bioinformatics. 2008;24:325–32.
Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–97.
Kara M, Yumrutas O, Ozcan O, Celik OI, Bozgeyik E, Bozgeyik I, Tasdemir S: Differential expressions of cancer-associated genes and their regulatory miRNAs in colorectal carcinoma. Gene 2015
Rehmsmeier M, Steffen P, Höchsmann M, Giegerich R. Fast and effective prediction of microRNA/target duplexes. RNA. 2004;10:1507–17.
Chan KL, Man-Fong Lee J, Guan XY, Fan ST, Oi-Lin Ng I. High-density allelotyping of chromosome 8p in hepatocellular carcinoma and clinicopathologic correlation. Cancer. 2002;94:3179–85.
Zuern C, Heimrich J, Kaufmann R, Richter KK, Settmacher U, Wanner C, et al. Down-regulation of mtus1 in human colon tumors. Oncol Rep. 2010;23:183–9.
Nouet S, Amzallag N, Li J-M, Louis S, Seitz I, Cui T-X, et al. Trans-inactivation of receptor tyrosine kinases by novel angiotensin ii at2 receptor-interacting protein, atip. J Biol Chem. 2004;279:28989–97.
Calin GA, Dumitru CD, Shimizu M, Bichi R, Zupo S, Noch E, et al. Frequent deletions and down-regulation of micro-RNA genes mir15 and mir16 at 13q14 in chronic lymphocytic leukemia. Proc Natl Acad Sci. 2002;99:15524–9.
Bandres E, Cubedo E, Agirre X, Malumbres R, Zarate R, Ramirez N, et al. Identification by real-time pcr of 13 mature microRNAs differentially expressed in colorectal cancer and non-tumoral tissues. Mol Cancer. 2006;5:29.
Nagel R, le Sage C, Diosdado B, van der Waal M, Vrielink JAFO, Bolijn A, et al. Regulation of the adenomatous polyposis coli gene by the mir-135 family in colorectal cancer. Cancer Res. 2008;68:5795–802.
Syring I, Bartels J, Holdenrieder S, Kristiansen G, Müller SC, Ellinger J. Circulating serum miRNA (mir-367-3p, mir-371a-3p, mir-372-3p and mir-373-3p) as biomarkers in patients with testicular germ cell cancer. J Urol. 2015;193:331–7.
Wu A, Li J, Wu K, Mo Y, Luo Y, Ye H, et al. Mir-373-3p promotes invasion and metastasis of lung adenocarcinoma cells. Zhongguo fei ai za zhi=. Chinese J lung Cancer. 2014;18:427–35.
Sarver AL, Li L, Subramanian S. MicroRNA mir-183 functions as an oncogene by targeting the transcription factor egr1 and promoting tumor cell migration. Cancer Res. 2010;70:9570–80.
Tang J-F, Yu Z-H, Liu T, Lin Z-Y, Wang Y-H, Yang L-W, et al. Five miRNAs as novel diagnostic biomarker candidates for primary nasopharyngeal carcinoma. Asian Pacific J Cancer Prevention: APJCP. 2013;15:7575–81.
Chang C-W, Wu H-C, Terry MB, Santella RM. MicroRNA expression in prospectively collected blood as a potential biomarker of breast cancer risk in the BCFR. Anticancer Res. 2015;35:3969–77.
Wang L, Zhu M-J, Ren A-M, Wu H-F, Han W-M, Tan R-Y, Tu R-Q: A ten-microRNA signature identified from a genome-wide microRNA expression profiling in human epithelial ovarian cancer. 2014
Bandras E, Cubedo E, Agirre X, Malumbres R, Zarate R, Ramirez N, et al. Identification by real-time PCR of 13 mature microRNAs differentially expressed in colorectal cancer and non-tumoral tissues. Mol Cancer. 2006;5:29.
Zhang X, Yan Z, Zhang J, Gong L, Li W, Cui J, Liu Y, Gao Z, Li J, Shen L: Combination of hsa-mir-375 and hsa-mir-142-5p as a predictor for recurrence risk in gastric cancer patients following surgical resection. Annals of oncology 2011:mdq758.
Jo DH, Kim JH, Park W-Y, Kim K-W, Yu YS, Kim JH. Differential profiles of microRNAs in retinoblastoma cell lines of different proliferation and adherence patterns. J Pediatr Hematol Oncol. 2011;33:529–33.
Saito Y, Suzuki H, Tsugawa H, Imaeda H, Matsuzaki J, Hirata K, Hosoe N, Nakamura M, Mukai M, Saito H: Overexpression of mir-142-5p and mir-155 in gastric mucosa-associated lymphoid tissue (malt) lymphoma resistant to helicobacter pylori eradication. 2012
Balci S, Ayaz L, Gorur A, Yildirim Yaroglu H, Akbayir S, Dogruer Unal N, Bulut B, Tursen U, Tamer L: MicroRNA profiling for early detection of nonmelanoma skin cancer. Clinical and experimental dermatology 2015
Ibarrola-Villava M, Llorca-Cardenosa MJ, Tarazona N, Mongort C, Fleitas T, Perez-Fidalgo JA, Rosello S, Navarro S, Ribas G, Cervantes A: Deregulation of arid1a, cdh1, cmet and pik3ca and target-related microRNA expression in gastric cancer. Oncotarget 2015
Zhi F, Shao N, Wang R, Deng D, Xue L, Wang Q, Zhang Y, Shi Y, Xia X, Wang S: Identification of 9 serum microRNAs as potential noninvasive biomarkers of human astrocytoma. Neuro-oncology 2014:nou169.
Wen Y, Han J, Chen J, Dong J, Xia Y, Liu J, Jiang Y, Dai J, Lu J, Jin G: Plasma mirnas as early biomarkers for detecting hepatocellular carcinoma. International journal of cancer 2015
Ye S-B, Li Z-L, Luo D-h, Huang B-j, Chen Y-S, Zhang X-S, et al. Tumor-derived exosomes promote tumor progression and t-cell dysfunction through the regulation of enriched exosomal microRNAs in human nasopharyngeal carcinoma. Oncotarget. 2014;5:5439.
Calvano Filho CMC, Calvano-Mendes DC, Carvalho KC, Maciel GA, Ricci MD, Torres AP, et al. Triple-negative and luminal a breast tumors: differential expression of mir-18a-5p, mir-17-5p, and mir-20a-5p. Tumor Biol. 2014;35:7733–41.
Chen X, Shi K, Wang Y, Song M, Zhou W, Tu H, Lin Z: Clinical value of integrated-signature miRNAs in colorectal cancer: miRNA expression profiling analysis and experimental validation. Oncotarget 2015
Li Y, Kuscu C, Banach A, Zhang Q, Pulkoski-Gross A, Kim D, Liu J, Roth E, Li E, Shroyer KR: MicroRNA-181a-5p inhibits cancer cell migration and angiogenesis via downregulation of matrix metalloproteinase-14. Cancer research 2015:canres. 2875.2014.
Ma Z, Qiu X, Wang D, Li Y, Zhang B, Yuan T, et al. Mir-181a-5p inhibits cell proliferation and migration by targeting kras in non-small cell lung cancer a549 cells. Acta Biochim Biophys Sin. 2015;47:630–8.
He S, Zeng S, Zhou Z-W, He Z-X, Zhou S-F. Hsa-microRNA-181a is a regulator of a number of cancer genes and a biomarker for endometrial carcinoma in patients: a bioinformatic and clinical study and the therapeutic implication. Drug Des Devel and Ther. 2015;9:1103.
Acknowledgments
This study was funded by a project from the Scientific Research Projects Management Unit of Mugla Sitki Kocman University (grant number 13/152).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Rights and permissions
About this article
Cite this article
Ozcan, O., Kara, M., Yumrutas, O. et al. MTUS1 and its targeting miRNAs in colorectal carcinoma: significant associations. Tumor Biol. 37, 6637–6645 (2016). https://doi.org/10.1007/s13277-015-4550-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-4550-4