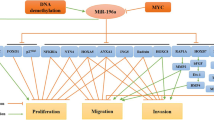
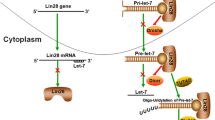
Abstract
TARBP2 is a RNA-binding protein (RBP) involved in miRNA processing and maturation. TARBP2 plays significant roles in many biological and pathological conditions, including viral expression of HIV-1, microsatellite instability, cancer stem cell properties, and tumor progression. Overexpression of TARBP2 was observed in many cancers such as prostate cancer, cutaneous malignant melanoma, and adrenocortical carcinoma. In addition, TARBP2 was also found to be downregulated in some cancers including colorectal cancer, gastric cancer, Ewing sarcoma, and upper urinary tract urothelial carcinoma. Therefore, whether TARBP2 functions as the tumor suppressor or tumor promoter is conflicting. In the present review, we provide an overview of current knowledge concerning the role of TARBP2 in tumor development and progression.
Similar content being viewed by others
References
Garre P, Perez-Segura P, Diaz-Rubio E, Caldes T, de la Hoya M. Reassessing the TARBP2 mutation rate in hereditary nonpolyposis colorectal cancer. Nat Genet. 2010;42:817–8. author reply 818.
Zimmermann J, Latta L, Beck A, Leidinger P, Fecher-Trost C, Schlenstedt G, et al. Trans-activation response (TAR) RNA-binding protein 2 is a novel modulator of transient receptor potential canonical 4 (TRPC4) protein. J Biol Chem. 2014;289:9766–80.
Bannwarth S, Talakoub L, Letourneur F, Duarte M, Purcell DF, Hiscott J, et al. Organization of the human tarbp2 gene reveals two promoters that are repressed in an astrocytic cell line. J Biol Chem. 2001;276:48803–13.
Melo SA, Ropero S, Moutinho C, Aaltonen LA, Yamamoto H, Calin GA, et al. A TARBP2 mutation in human cancer impairs microRNA processing and DICER1 function. Nat Genet. 2009;41:365–70.
Sand M, Skrygan M, Georgas D, Arenz C, Gambichler T, Sand D, et al. Expression levels of the microRNA maturing microprocessor complex component DGCR8 and the RNA-induced silencing complex (RISC) components argonaute-1, argonaute-2, PACT, TARBP1, and TARBP2 in epithelial skin cancer. Mol Carcinog. 2012;51:916–22.
de Sousa GR, Ribeiro TC, Faria AM, Mariani BM, Lerario AM, Zerbini MC, Soares IC, Wakamatsu A, Alves VA, Mendonca BB, Fragoso MC, Latronico AC, Almeida MQ. Low DICER1 expression is associated with poor clinical outcome in adrenocortical carcinoma. Oncotarget. 2015.
Kozak CA, Gatignol A, Graham K, Jeang KT, McBride OW. Genetic mapping in human and mouse of the locus encoding TRBP, a protein that binds the TAR region of the human immunodeficiency virus (HIV-1). Genomics. 1995;25:66–72.
Caramuta S, Lee L, Ozata DM, Akcakaya P, Xie H, Hoog A, et al. Clinical and functional impact of TARBP2 over-expression in adrenocortical carcinoma. Endocr Relat Cancer. 2013;20:551–64.
Bai S, Nunez AL, Wei S, Ziober A, Yao Y, Tomaszewski JE, et al. Microsatellite instability and TARBP2 mutation study in upper urinary tract urothelial carcinoma. Am J Clin Pathol. 2013;139:765–70.
De Vito C, Riggi N, Cornaz S, Suva ML, Baumer K, Provero P, et al. A TARBP2-dependent miRNA expression profile underlies cancer stem cell properties and provides candidate therapeutic reagents in Ewing sarcoma. Cancer Cell. 2012;21:807–21.
Sousa E, Graca I, Baptista T, Vieira FQ, Palmeira C, Henrique R, et al. Enoxacin inhibits growth of prostate cancer cells and effectively restores microRNA processing. Epigenetics. 2013;8:548–58.
Sand M, Skrygan M, Georgas D, Sand D, Gambichler T, Altmeyer P, et al. The miRNA machinery in primary cutaneous malignant melanoma, cutaneous malignant melanoma metastases and benign melanocytic nevi. Cell Tissue Res. 2012;350:119–26.
Kim MS, Oh JE, Kim YR, Park SW, Kang MR, Kim SS, et al. Somatic mutations and losses of expression of microRNA regulation-related genes AGO2 and TNRC6A in gastric and colorectal cancers. J Pathol. 2010;221:139–46.
Zhong J, Peters AH, Lee K, Braun RE. A double-stranded RNA binding protein required for activation of repressed messages in mammalian germ cells. Nat Genet. 1999;22:171–4.
Goodarzi H, Zhang S, Buss CG, Fish L, Tavazoie S, Tavazoie SF. Metastasis-suppressor transcript destabilization through TARBP2 binding of mRNA hairpins. Nature. 2014;513:256–60.
Lin X, Wu M, Liu P, Wei F, Li L, Tang H, et al. Up-regulation and worse prognostic marker of cytoplasmic TARBP2 expression in obstinate breast cancer. Med Oncol. 2014;31:868.
Caramuta S, Lee L, Ozata DM, Akcakaya P, Georgii-Hemming P, Xie H, et al. Role of microRNAs and microRNA machinery in the pathogenesis of diffuse large B-cell lymphoma. Blood Cancer J. 2013;3:e152.
Yamamoto H, Imai K. Microsatellite instability: an update. Arch Toxicol. 2015;89:899–921.
Audenet F, Yates DR, Cussenot O, Roupret M. The role of chemotherapy in the treatment of urothelial cell carcinoma of the upper urinary tract (UUT-UCC). Urol Oncol. 2013;31:407–13.
Cornaz-Buros S, Riggi N, DeVito C, Sarre A, Letovanec I, Provero P, et al. Targeting cancer stem-like cells as an approach to defeating cellular heterogeneity in Ewing sarcoma. Cancer Res. 2014;74:6610–22.
Torrezan GT, Ferreira EN, Nakahata AM, Barros BD, Castro MT, Correa BR, et al. Recurrent somatic mutation in DROSHA induces microRNA profile changes in Wilms tumour. Nat Commun. 2014;5:4039.
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (NSFC) (Grant Number: 81401847).
Author information
Authors and Affiliations
Corresponding author
Additional information
Xin Yu and Zheng Li contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yu, X., Li, Z. The role of TARBP2 in the development and progression of cancers. Tumor Biol. 37, 57–60 (2016). https://doi.org/10.1007/s13277-015-4273-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-4273-6