Abstract
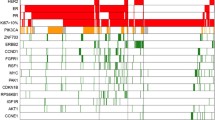
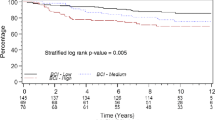
The gene profile of primary tumors, as well as the identification of circulating tumor cells (CTCs), can provide important prognostic and predictive information. In this study, our objective was to perform tumor gene profiling (TGP) in combination with CTC characterization in women with nonmetastatic breast cancer. Biological samples (from peripheral blood and tumors) from 167 patients diagnosed with stage I, II, and III mammary carcinoma, who were also referred for adjuvant/neoadjuvant chemotherapy, were assessed for the following parameters: (a) the presence of CTCs identified by the expression of CK-19 and c-erbB-2 in the peripheral blood mononuclear cell (PBMC) fraction by quantitative reverse transcription PCR (RT-PCR) and (b) the TGP, which was determined by analyzing the expression of 21 genes in paraffin-embedded tissue samples by quantitative multiplex RT-PCR with the Plexor® system. We observed a statistically significant correlation between the progression-free interval (PFI) and the clinical stage (p = 0.000701), the TGP score (p = 0.006538), and the presence of hormone receptors in the tumor (p = 0.0432). We observed no correlation between the PFI and the presence or absence of CK-19 or HER2 expression in the PBMC fraction prior to the start of treatment or in the two following readouts. Multivariate analysis revealed that only the TGP score significantly correlated with the PFI (p = 0.029247). The TGP is an important prognostic variable for patients with locoregional breast cancer. The presence of CTCs adds no prognostic value to the information already provided by the TGP.

Similar content being viewed by others
References
INCA (2012) Atlas de mortalidade por cancer. http://mortalidade.inca.gov.br/Mortalidade/.
Pusztai L. Current status of prognostic profiling in breast cancer. Oncologist. 2008;13:350–60.
Wang Y, Klijn JG, Zhang Y, Sieuwerts AM, Look MP, Yang F, et al. Gene-expression profiles to predict distant metastasis of lymph-node-negative primary breast cancer. Lancet. 2005;365:671–9.
Paik S, Shak S, Tang G, Kim C, Baker J, Cronin M, et al. A multigene assay to predict recurrence of tamoxifen-treated, node-negative breast cancer. N Engl J Med. 2004;351:2817–26.
van de Vijver MJ, He YD, van’t Veer LJ, Dai H, Hart AA, Voskuil DW, et al. A gene-expression signature as a predictor of survival in breast cancer. N Engl J Med. 2002;347:1999–2009.
Fisher B, Jeong JH, Bryant J, Anderson S, Dignam J, Fisher ER, et al. Treatment of lymph-node-negative, estrogen-receptor-positive breast cancer: long-term findings from national surgical adjuvant breast and bowel project randomised clinical trials. Lancet. 2004;364:858–68.
Gianni L, Zambetti M, Clark K, Baker J, Cronin M, Wu J, et al. Gene expression profiles in paraffin-embedded core biopsy tissue predict response to chemotherapy in women with locally advanced breast cancer. J Clin Oncol. 2005;23:7265–727.
Habel LA, Quesenberry CP, Jacobs M, Greenberg D, Fehrenbacher L, Alexander C, et al. Gene expression and breast cancer mortality in Northern California Kaiser Permanente patients: a large population-based case control study. J Clin Oncol. 2005;23(Suppl 18S):603.
Mamounas EP, Tang G, Tang G, Paik S, Baehner FL, Liu Q, et al. Prognostic impact of 21-gene recurrence score (RS) on disease-free and overall survival of node-positive, ER-positive breast cancer patients (pts.) treated with adjuvant chemotherapy: results from NSABP B-28 [abstract]. J Clin Oncol (2012), 30(Suppl 1).
Mikulová V, Kološtová K, Zima T. Methods for detection of circulating tumour cells and their clinical value in cancer patients. Folia Biol (Praha). 2011;57:151–61.
Ring A, Smith IE, Dowsett M. Circulating tumour cells in breast cancer. Lancet Oncol. 2004;5:79–88.
Cristofanilli M, Budd GT, Ellis MJ, Stopeck A, Matera J, Miller MC, et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med. 2004;351:781–91.
Strati A, Markou A, Parisi C, Politaki E, Mavroudis D, Georgoulias V, et al. Gene expression profile of circulating tumor cells in breast cancer by RT-qPCR. BMC Cancer. 2011;11:422.
Camara O, Rengsberger M, Egbe A, Koch A, Gajda M, Hammer U, et al. The relevance of circulating epithelial tumor cells (CETC) for therapy during neoadjuvant (primary systemic) chemotherapy in breast cancer. Ann Oncol. 2007;18:1484–92.
Lobodasch K, Frohlich F, Rengsberger M, Schubet R, Dengler R, Pachmann U, et al. Quantification of circulating tumor cells for the monitoring of therapy adjuvant therapy in breast cancer: an increase in cell number at completion of therapy is a predictor of early relapse. Breast. 2007;16:211–8.
Pachmann K, Camara O, Kavallaris A, Krauspe S, Malarski N, Gajda M, et al. Monitoring the response of circulating epithelial tumor cells to adjuvant chemotherapy in breast cancer allows detection of patients at risk of early relapse. J Clin Oncol. 2008;26:1208–15.
Novaes M, Bendit I, Garicochea B, del Giglio A. Reverse transcriptase-polymerase chain reaction analysis of cytokeratin 19 expression in the peripheral blood mononuclear cells of normal female blood donors. Mol Pathol. 1997;50:209–11.
Manhani AR, Manhani R, Soares HP, Bendit I, Lopes F, Nicoletti AG, et al. CK-19 expression by RT-PCR in the peripheral blood of breast cancer patients correlates with response to chemotherapy. Breast Cancer Res Treat. 2001;66:249–54.
Fonseca FL, Soares HP, Manhani AR, Bendit I, Novaes M, Zatta SM, et al. Peripheral blood c-erbB-2 expression by reverse transcriptase-polymerase chain reaction in breast cancer patients receiving chemotherapy. Clin Breast Cancer. 2002;3:201–5.
Apostolaki S, Perraki M, Pallis A, Bozionelou V, Agelaki S, Kanellou P, et al. Circulating HER2 mRNA-positive cells in the peripheral blood of patients with stage I and II breast cancer after the administration of adjuvant chemotherapy: evaluation of their clinical relevance. Ann Oncol. 2007;18:851–8. Erratum in: Ann Oncol 2007;18:1916.
Ignatiadis M, Perraki M, Apostolaki S, Politaki E, Xenidis N, Kafousi M, et al. Molecular detection and prognostic value of circulating cytokeratin-19 messenger RNA-positive and HER2 messenger RNA-positive cells in the peripheral blood of women with early-stage breast cancer. Clin Breast Cancer. 2007;7:883–9.
Toussaint J, Sieuwerts AM, Haibe-Kains B, Desmedt C, Rouas G, Harris AL, et al. Improvement of the clinical applicability of the Genomic Grade Index through a qRT-PCR test performed on frozen and formalin-fixed paraffin-embedded tissues. BMC Genomics. 2009;10:424.
Savino M, Garrubba M, Parrella P, Baorda F, Copetti M, Murgo R, et al. Development of real-time quantitative reverse transcription-PCR for Her2 detection in peripheral blood from patients with breast cancer. Clin Chim Acta. 2007;38:52–6.
Čabiňaková M, Tesařová P. Disseminated and circulating tumour cells and their role in breast cancer. Folia Biol (Praha). 2012;58:87–97.
You F, Roberts LA, Kang SP, Nunes RA, Dias C, Iglehart JD, et al. Low-level expression of HER2 and CK19 in normal peripheral blood mononuclear cells: relevance for detection of circulating tumor cells. J Hematol Oncol. 2008;1:2.
Daskalaki A, Agelaki S, Perraki M, Apostolaki S, Xenidis N, Stathopoulos E, et al. Detection of cytokeratin-19 mRNA-positive cells in the peripheral blood and bone marrow of patients with operable breast cancer. Br J Cancer. 2009;101:589–97.
Filipits M, Rudas M, Jakesz R, Dubsky P, Fitzal F, Singer CF, et al. A new molecular predictor of distant recurrence in ER-positive, HER2-negative breast cancer adds independent information to conventional clinical risk factors. Clin Cancer Res. 2011;17:6012–20.
Acknowledgments
We thank Fundação de Amparo a Pesquisa do Estado de São Paulo (Fapesp) for supporting this research (Proc. no. 09/50361-4).
Conflicts of interest
None
Author contributions
RKK, FSG, and BCAA carried out the validation of genes and Plexor system. VVB, AEC, and FLAF carried out the lab analysis from blood samples. NS and JN contributed to the acquisition of data. BCAA and FLAF contributed to the analysis and interpretation of data. ADG performed the statistical analysis. ADG conceived the study and participated in its design and coordination and helped to draft the manuscript. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kuniyoshi, R.K., de Sousa Gehrke, F., Alves, B.C.A. et al. Gene profiling and circulating tumor cells as biomarker to prognostic of patients with locoregional breast cancer. Tumor Biol. 36, 8075–8083 (2015). https://doi.org/10.1007/s13277-015-3529-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3529-5