Abstract
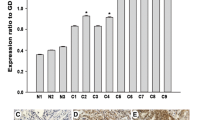
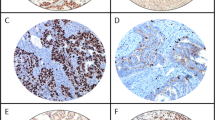
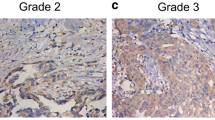
Overexpression of cyclin-dependent kinase 1 (CDK1) has been noted to correlation with several human cancers. However, the effects of CDK1 on ovarian cancer development remain unclear. The aim of this study was to examine the effect of CDK1 and related mechanism in the proliferation and resistance to chemotherapeutic drugs of epithelial ovarian cancer (EOC). Immunohistochemical analysis was performed in 119 human ovarian cancer samples, and the data were correlated with clinicopathologic features. Furthermore, Western blot analysis was performed for CDK1 in EOC samples and cell lines to evaluate their protein levels and molecular interaction. Kaplan-Meier survival analysis showed that strong expression of CDK1 exhibited a significant correlation with poor prognosis in human EOC (P = 0.02). Meanwhile, we found that knockdown CDK1 by shCDK1 promoted the apoptosis rate and increased the sensitivity to chemotherapy drugs. Thus, CDK1 might serve as a prognostic marker, and it might be of great value for experimental therapies in EOC.






Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin. 2012;62(1):10–29. doi:10.3322/caac.20138.
Lambert HE, Gregory WM, Nelstrop AE, Rustin GJ. Long-term survival in 463 women treated with platinum analogs for advanced epithelial carcinoma of the ovary: life expectancy compared to women of an age-matched normal population. Int J Gynecol Cancer Off J Int Gynecol Cancer Soc. 2004;14(5):772–8. doi:10.1111/j.1048-891X.2004.014507.x.
Leitao Jr MM, Hummer A, Dizon DS, Aghajanian C, Hensley M, Sabbatini P, et al. Platinum retreatment of platinum-resistant ovarian cancer after nonplatinum therapy. Gynecol Oncol. 2003;91(1):123–9.
Luvero D, Milani A, Ledermann JA. Treatment options in recurrent ovarian cancer: latest evidence and clinical potential. Ther Adv Med Oncol. 2014;6(5):229–39. doi:10.1177/1758834014544121.
Malumbres M. Physiological relevance of cell cycle kinases. Physiol Rev. 2011;91(3):973–1007. doi:10.1152/physrev.00025.2010.
Goranov AI, Amon A. Growth and division—not a one-way road. Curr Opin Cell Biol. 2010;22(6):795–800. doi:10.1016/j.ceb.2010.06.004.
Malumbres M, Barbacid M. Cell cycle, CDKs and cancer: a changing paradigm. Nat Rev Cancer. 2009;9(3):153–66. doi:10.1038/nrc2602.
Li JM, Brooks G. Cell cycle regulatory molecules (cyclins, cyclin-dependent kinases and cyclin-dependent kinase inhibitors) and the cardiovascular system; potential targets for therapy? Eur Heart J. 1999;20(6):406–20.
Kang J, Sergio CM, Sutherland RL, Musgrove EA. Targeting cyclin-dependent kinase 1 (CDK1) but not CDK4/6 or CDK2 is selectively lethal to MYC-dependent human breast cancer cells. BMC Cancer. 2014;14:32. doi:10.1186/1471-2407-14-32.
Kim SJ, Nakayama S, Shimazu K, Tamaki Y, Akazawa K, Tsukamoto F, et al. Recurrence risk score based on the specific activity of CDK1 and CDK2 predicts response to neoadjuvant paclitaxel followed by 5-fluorouracil, epirubicin and cyclophosphamide in breast cancers. Ann Oncol Off J Eur Soc Med Oncol / ESMO. 2012;23(4):891–7. doi:10.1093/annonc/mdr340.
Zeestraten EC, Maak M, Shibayama M, Schuster T, Nitsche U, Matsushima T, et al. Specific activity of cyclin-dependent kinase I is a new potential predictor of tumour recurrence in stage II colon cancer. Br J Cancer. 2012;106(1):133–40. doi:10.1038/bjc.2011.504.
Chang CC, Hung CM, Yang YR, Lee MJ, Hsu YC. Sulforaphane induced cell cycle arrest in the G2/M phase via the blockade of cyclin B1/CDC2 in human ovarian cancer cells. J Ovarian Res. 2013;6(1):41. doi:10.1186/1757-2215-6-41.
Itamochi H, Yoshida T, Walker CL, Bartholomeusz C, Aoki D, Ishihara H, et al. Novel mechanism of reduced proliferation in ovarian clear cell carcinoma cells: cytoplasmic sequestration of CDK2 by p27. Gynecol Oncol. 2011;122(3):641–7. doi:10.1016/j.ygyno.2011.05.003.
Yang Y, Ma B, Li L, Jin Y, Ben W, Zhang D, et al. CDK2 and CDK4 play important roles in promoting the proliferation of SKOV3 ovarian carcinoma cells induced by tumor-associated macrophages. Oncol Rep. 2014;31(6):2759–68. doi:10.3892/or.2014.3127.
Zhu X, Li Y, Xie C, Yin X, Liu Y, Cao Y, et al. miR-145 sensitizes ovarian cancer cells to paclitaxel by targeting Sp1 and Cdk6. Int J Cancer. 2014;135(6):1286–96. doi:10.1002/ijc.28774.
Joshi PM, Sutor SL, Huntoon CJ, Karnitz LM. Ovarian cancer-associated mutations disable catalytic activity of CDK12, a kinase that promotes homologous recombination repair and resistance to cisplatin and poly(ADP-ribose) polymerase inhibitors. J Biol Chem. 2014;289(13):9247–53. doi:10.1074/jbc.M114.551143.
Wu W, Ye H, Wan L, Han X, Wang G, Hu J, et al. Millepachine, a novel chalcone, induces G2/M arrest by inhibiting CDK1 activity and causing apoptosis via ROS-mitochondrial apoptotic pathway in human hepatocarcinoma cells in vitro and in vivo. Carcinogenesis. 2013;34(7):1636–43. doi:10.1093/carcin/bgt087.
Chen X, Zhang FH, Chen QE, Wang YY, Wang YL, He JC et al. The clinical significance of CDK1 expression in oral squamous cell carcinoma. Medicina oral, patologia oral y cirugia bucal. 2014.
Zhang C, Elkahloun AG, Robertson M, Gills JJ, Tsurutani J, Shih JH, et al. Loss of cytoplasmic CDK1 predicts poor survival in human lung cancer and confers chemotherapeutic resistance. PLoS ONE. 2011;6(8):e23849. doi:10.1371/journal.pone.0023849.
Hedblom A, Laursen KB, Miftakhova R, Sarwar M, Anagnostaki L, Bredberg A, et al. CDK1 interacts with RARgamma and plays an important role in treatment response of acute myeloid leukemia. Cell Cycle. 2013;12(8):1251–66. doi:10.4161/cc.24313.
Acknowledgments
A graduate project of the Science and Technology Innovation in Nantong University (YKS14023), contract grant number 13025202.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Qinghua Xi and Menghui Huang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Xi, Q., Huang, M., Wang, Y. et al. The expression of CDK1 is associated with proliferation and can be a prognostic factor in epithelial ovarian cancer. Tumor Biol. 36, 4939–4948 (2015). https://doi.org/10.1007/s13277-015-3141-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3141-8