Abstract
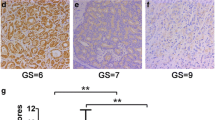
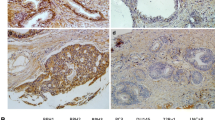
This study aimed to analyze the expression, clinical significance of f epithelial membrane protejn-1 (EMP-1) in prostate carcinoma, and the biological effect in its cell line by EMP1 overexpression. Immunohistochemistry and Western blot were used to analyze EMP1 protein expression in 76 cases of prostate cancer and 34 cases of normal tissues to study the relationship between EMP1 expression and clinical factors. EMP1 lentiviral vector and empty vector were respectively transfected into prostate cancer PC-3 cell line. Quantitative real-time reverse transcription polymerase chain reaction and Western blot were used to detect the mRNA level and protein of EMP1. 3-[4,5-Dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide assay, migration, and invasion assays were also conducted as to the influence of the upregulated expression of EMP1 that might be found on PC-3 cell biological effect. Immunohistochemistry: The level of EMP1 protein expression was found to be significantly lower in prostate cancer tissue than normal tissues (P < 0.05). Western blot: The relative amount of EMP1 protein in prostate cancer tissue was found to be significantly lower than in normal tissues (P < 0.05). The level of EMP1 protein expression was not correlated with age and prostate-specific antigen (PSA) concentration (P > 0.05), but it was correlated with T stages, lymph node metastasis, clinic stage, and Gleason score (P < 0.05). The result of biological function shown that PC-3 cell transfected EMP1 had a lower survival fraction, higher cell apoptosis, significant decrease in migration and invasion, higher caspase-9, and lower VEGFC protein expression compared with PC-3 cell untransfected EMP1 (P < 0.05). EMP1 expression decreased in prostate cancer and correlated significantly T stages, lymph node metastasis, clinic stage, and Gleason score, suggesting that EMP1 may play important roles as a negative regulator to prostate cancer PC-3 cell by regulating the expression of regulation of caspase-9 and VEGFC protein.







Similar content being viewed by others
Reference
Greenlee RT, Murray T, Bolden S, Wingo PA, Cancer statistics. CA CancerJ Clin. 2000;50:7–33.
Dong JT, Isaacs WB, Isaacs JT. Molecular advances in prostate cancer. Curr opin oncol. 1997;9:101–7.
Lai S, Wang G, Cao X, Li Z, Hu J, Wang J. EMP-1 promotes tumorigenesis of NSCLC through PI3K/AKT pathway. J Huazhong Univ Sci Technolog Med Sci. 2012;32:834–8.
Taylor V, Welcher AA, Program AE, Suter U. Epithelial membrane protein-1, peripheral myelin protein 22, and lens membrane protein 20 define a novel gene family. J Biol Chem. 1995;270:28824–33.
Lobsiger CS, Magyar JP, Taylor V, Wulf P, Welcher AA, Program AE, et al. Identification and characterization of a cDNA and the structural gene encoding the mouse epithelial membrane protein-1. Genomics. 1996;36:379–87.
Wulf P, Suter U. Embryonic expression of epithelial membrane protein 1 in early neurons. Brain Res Dev Brain Res. 1999;116:169–80.
Zoidl G, Blass-Kampmann S, D'Urso D, Schmalenbach C, Müller HW. Retroviral-mediated gene transfer of the peripheral myelin protein PMP22 in Schwann cells: modulation of cell growth. EMBO J. 1995;14:1122–8.
Jetten AM, Suter U. The peripheral myelin protein 22 and epithelial membrane protein family. Prog Nucleic Acid Res Mol Biol. 2000;64:97–129.
Lee HS, Sherley JL, Chen JJ, Chiu CC, Chiou LL, Liang JD, et al. EMP1 is a junctional protein in a liver stem cell line and in the liver. Biochem Biophys res commun. 2005;334:996–1003.
Gnirke AU, Weidle UH. Investigation of prevalence and regulation of expression of progression associated protein (PAP). Anticancer Res. 1998;18:4363–9.
Wang HT, Kong JP, Ding F, Wang XQ, Wang MR, Liu LX, et al. Analysis of gene expression profile induced by EMP-1 inesophageal cancer cells using cDNA microarray. World J Gastroenterol. 2003;9:392–8.
Zhang J, Cao W, Xu Q, Chen WT. The expression of EMP1 is downregulated in oral squamous cell carcinoma and possibly associated with tumour metastasis. J Clin Pathol. 2011;64:25–9.
Fu L, Chen W, Guo W, Wang J, Tian Y, Shi D, et al. Berberine targets AP-2/hTERT, NF-κB/COX-2, HIF-1α/VEGF and cytochrome-c/caspase signaling to suppress human cancer cell growth. PLoS One. 2013;8:e69240.
Sen S, Kawahara B, Chaudhuri G. Mitochondrial-associated nitric oxide synthase activity inhibits cytochrome c oxidase: implications for breast Cancer. Free Radic Biol Med. 2013;57:210–20.
Zhang JM, Wang HC, Wang HX, Ruan LH, Zhang YM, Li JT, et al. Oxidative stress and activities of caspase-8,-9,and −3 are involved in cryopreservation- induced apoptosis ingranulosa cells. Eur J Obstet Gynecol Reprod Biol. 2013;166:52–5.
Peng J, Shao N, Peng H, Chen LQ. Prognostic significance of vascular endothelial growth factor expression in esophageal carcinoma: a meta-analysis. J Buon. 2013;18:398–406.
Xie LX, Zhai TT, Yang LP, Yang E, Zhang XH, Chen JY, et al. Lymphangiogenesis and prognostic significance of vascular endothelial growth factor C in gastro-oesophageal junction adenocarcinoma. Int J Exp Pathol. 2013;94:39–46.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Sun, G.G., Wang, Y.D., Cui, D.W. et al. EMP1 regulates caspase-9 and VEGFC expression and suppresses prostate cancer cell proliferation and invasion. Tumor Biol. 35, 3455–3462 (2014). https://doi.org/10.1007/s13277-013-1456-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-013-1456-x