Abstract
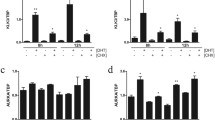
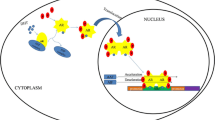
Recent data suggested that tissue human kallikrein 2 (KLK2) might be involved in the carcinogenesis and tumor metastasis of prostate cancer (PCa). However, the detailed pathophysiological roles of KLK2 in PCa remain unclear. We report here that KLK2 may be treated as a potential therapeutic target in castration-resistant PCa (CRPC). Histologic analyses show that the increased KLK2 expression is correlated with higher cell proliferation rate and lower cell apoptosis index in CRPC specimens. Adding functional KLK2 cDNA into high passage LNCaP cells led to increased cell growth, and knockdown of KLK2 expression with KLK2-siRNA in LNCaP cells resulted in increased cell apoptosis with cell growth arrest at the G1 phase. Results from in vitro colony formation assay and in vivo xenografted PCa tissues also demonstrated that targeting KLK2 led to suppressed growth of PCa in the castration-resistant stage. Further mechanism dissection shows that KLK2 may cooperate with the AR coregulator, ARA70, to enhance AR transactivation that may result in alteration of PCa formation. Together, these results suggested KLK2 might become a new therapeutic target to battle the CRPC and KLK2-siRNA may be developed as an alternative approach to suppress PCa growth.





Similar content being viewed by others
References
Borgoño CA, Michael IP, Diamandis EP. Human tissue kallikreins. Physiologic roles and applications in cancer. Mol Cancer Res. 2004;2:257–80.
Lövgren J, Valtonen-André C, Marsal K, Lilja H, Lundwall A. Measurement of prostate-specific antigen and human glandular kallikrein 2 in different body fluids. J Androl. 1999;20:348–55.
Kumar A, Mikolajczyk SD, Goel AS, Millar LS, Saedi MS. Expression of pro form of prostate-specific antigen by mammalian cells and its conversion to mature, active form by kallikrein 2. Cancer Res. 1997;57:3111–4.
Murtha P, Tindall DJ, Young CY. Androgen induction of a human prostate-specific kallikrein, hKLK2: characterization of an androgen response element in the 5′ promoter region of the gene. Biochemistry. 1993;32:6459–64.
Diamandis EP, Yousef GM, Luo LY, Magklara A, Obiezu CV. The new human kallikrein gene family: implications in carcinogenesis. Trend Endocrinol Metab. 2000;11:54–60.
Solovyeva NI, Balayevskaya TO, Dilakyan EA, Topol LZ, Kisseliov FL. Plasminogen activators, kallikrein-like proteinase and type I and type IV collagenases at various stages of oncogenic transformation. Immunopharmacology. 1996;32:131–4.
Williams SA, Xu Y, De Marzo AM, Isaacs JT, Denmeade SR. Prostate-specific antigen (PSA) is activated by KLK2 in prostate cancer ex vivo models and in prostate-targeted PSA/KLK2 double transgenic mice. Prostate. 2010;70(7):788–96.
Yeh S, Chang C. Cloning and characterization of a specific coactivator, ARA70, for the androgen receptor in human prostate cells. Proc Nat Acad Sci. 1996;93:5517–21.
Heinlein CA, Ting HJ, Yeh S, Chang C. Identification of ARA70 as a ligand-enhanced coactivator for the peroxisome proliferator-activated receptor-gamma. J Biol Chem. 1999;274:16147–52.
Lin HK, Hu YC, Yang L, Altuwaijri S, Chen YT, Kang HY, et al. Suppression versus induction of androgen receptor functions by the phosphatidylinositol 3-kinase/Akt pathway in prostate cancer LNCaP cells with different passage numbers. J Biol Chem. 2003;278:50902–7.
Yeh S, Miyamoto H, Chang C. Hydroxyflutamide may not always be a pure antiandrogen. Lancet. 1997;349:852–3.
Miyamoto H, Yeh S, Wilding G, Chang C. Promotion of agonist activity of antiandrogens by the androgen receptor co-activator, ARA70, in human prostate DU145 cells. Proc Natl Acad Sci U S A. 1998;95:7379–84.
Diamandis EP. Prostate-specific antigen: its usefulness in clinical medicine. Trends Endocrinol Metab. 1988;9:310–6.
Clements JA, Willemsen NM, Myers SA, Dong Y. The tissue kallikrein family of serine proteases: functional roles in human disease and potential as clinical biomarkers. Crit Rev Clin Lab Sci. 2004;41:265–312.
Bindukumar B, Schwartz SA, Nair MP, Aalinkeel R, Kawinski E, Chadha KC. Prostate-specific antigen modulates the expression of genes involved in prostate tumor growth. Neoplasia. 2005;7:241–52.
Takayama TK, Fujikawa K, Davie EW. Characterization of the precursor of prostate-specific antigen. Activation by trypsin and by human glandular kallikrein. J Biol Chem. 1997;272:21582–8.
Lilja H, Oldbring J, Rannevik G, Laurell CB. Seminal vesicle-secreted proteins and their reactions during gelation and liquefication of human semen. J Clin Invest. 1987;80:281–5.
Koeneman KS, Yeung F, Chung LW. Osteomimetic properties of prostate cancer cells: a hypothesis supporting the predilection of prostate cancer metastasis and growth in the bone environment. Prostate. 1999;39(4):246–61.
Heidtmann HH, Nettelbeck DM, Mingels A, Jäger R, Welker HG, Kontermann RE. Generation of angiostatin-like fragments from plasminogen by prostate-specific antigen. Br J Cancer. 1999;811:269–1273.
Réhault S, Monget P, Mazerbourg S, Tremblay R, Gutman N, Gauthier F, et al. Insulin-like growth factor binding proteins (IGFBPs) as potential physiological substrates for human kallikreins KLK2 and hK3. Eur J Biochem. 2001;268:2960–8.
Mize GJ, Wang W, Takayama TK. Prostate-specific kallikreins-2 and −4 enhance the proliferation of DU-145 prostate cancer cells through protease-activated receptors-1 and −2. Mol Cancer Res. 2008;6(6):1043–51.
Rahman MM, Miyamoto H, Takatera H, Yeh S, Altuwaijri S, Chang C. Reducing the agonist activity of antiandrogens by a dominant-negative androgen receptor coregulator ARA70 in prostate cancer cells. J Biol Chem. 2003;278:19619–26.
Gregory CW, Hamil KG, Kim D, Hall SH, Pretlow TG, Mohler JL, et al. Androgen receptor expression in androgen-independent prostate cancer is associated with increased expression of androgen-regulated genes. Cancer Res. 1998;58:5718–24.
Hu YC, Yeh S, Yeh SD, Sampson ER, Huang J, Li P, et al. Functional domain and motif analyses of androgen receptor coregulator ARA70 and its differential expression in prostate cancer. J Biol Chem. 2004;279:33438–46.
Niu Y, Yeh S, Miyamoto H, Li G, Altuwaijri S, Yuan J, et al. Tissue prostate-specific antigen facilitates refractory prostate tumor progression via enhancing ARA70-regulated androgen receptor transactivation. Cancer Res. 2008;68(17):7110–9.
Koistinen H, Närvänen A, Pakkala M, Hekim C, Mattsson JM, Zhu L, et al. Development of peptides specifically modulating the activity of KLK2 and KLK3. Biol Chem. 2008;389(6):633–42.
Hekim C, Leinonen J, Närvänen A, Koistinen H, Zhu L, Koivunen E, et al. Novel Peptide inhibitors of human kallikrein 2. J Biol Chem. 2006;281(18):12555–60.
Acknowledgments
This work was supported by National Basic Research Program of China (grant no. 2012CB518304); National Institute of Health grants (grant no. CA155477, CA156700); TSTC grant 2012DFG32220; Nature Science Foundation of Tianjin (grant no. 11JCYBJC28400).
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding authors
Additional information
Zhiqun Shang and Yuanjie Niu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Shang, Z., Niu, Y., Cai, Q. et al. Human kallikrein 2 (KLK2) promotes prostate cancer cell growth via function as a modulator to promote the ARA70-enhanced androgen receptor transactivation. Tumor Biol. 35, 1881–1890 (2014). https://doi.org/10.1007/s13277-013-1253-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-013-1253-6