Abstract
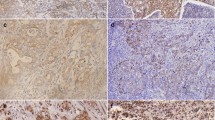
Abnormal activation of the hedgehog (Hh) signaling pathway has been found to be involved in the occurrence, invasion, and metastasis of cancers. Epithelial–mesenchymal transition (EMT) also plays an important role in the invasion and metastasis of cancers. However, the significance of the Hh signaling pathway and EMT in the invasion and metastasis of gastric cancer is still unclear. This study aimed to investigate the significance and prognostic value of the Hh signaling pathway and EMT in progressive gastric cancer. Immunohistochemistry was performed to detect the expression of the Hh-induced transcriptional factor Gli-1 and the EMT-related molecules Snail and E-cadherin in 121 patients with progressive gastric cancer. Histological type, depth of invasion, lymph node metastasis, and pTNM stage were also recorded. In progressive gastric cancer, Gli-1 expression increased markedly, and was closely associated with increased Snail expression and decreased E-cadherin expression. Diffuse type cancer, lymph node metastasis, and abnormal expression of E-cadherin were independent factors influencing the prognosis of patients with progressive gastric cancer. These findings suggest that abnormal activation of the Hh signaling pathway is closely related to the presence of EMT and is an important factor influencing the prognosis of patients with diffuse progressive gastric cancer.


Similar content being viewed by others
References
Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55:74–108.
Lin Y, Ueda J, Kikuchi S, Totsuka Y, Wei W, Qiao Y, et al. Comparative epidemiology of gastric cancer between Japan and China. World J Gastroenterol. 2011;17:4421–8.
Tsugane S, Sasazuki S. Diet and the risk of gastric cancer: review of epidemiological evidence. Gastric Cancer. 2007;10:75–83.
Jemal A, Tiwari RC, Murray T, Ghafoor A, Samuels A, Ward E, et al. Cancer statistics, 2004. CA Cancer J Clin. 2004;54:8–29.
Beachy PA, Karhadkar SS, Berman DM. Tissue repair and stem cell renewal in carcinogenesis. Nature. 2004;432(7015):324–31.
Wicking C, Smyt HI, Bale A. The hedgehog signaling pathway in tumorigenesis and development. Oncogene. 1999;18(55):7844–51.
Yoo YA, Kang MH, Kim JS, Oh C. Sonic hedgehog signaling promotes motility and invasiveness of gastric cancer cells through TGF-beta-mediated activation of the ALK5-Smad 3 pathway. Carcinogenesis. 2008;29(3):480–90.
Thiery JP. Epithelial−mesenchymal transitions in tumour progression. Nat Rev Cancer. 2002;2:442–54.
Hugo H, Ackland ML, Blick T, Lawrence MG, Clements JA, Williams ED, et al. Epithelial–mesenchymal and mesenchymal–epithelial transitions in carcinoma progression. J Cell Physiol. 2007;213(2):374–83.
Chaffer CL, Brennan JP, Slavin JL, Blick T, Thompson EW, Williams ED. Mesenchymal-to-epithelial transition facilitates bladder cancer metastasis: role of fibroblast growth factor receptor-2. Cancer Res. 2006;66:11271–8.
Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A, editors. AJCC cancer staging manual. 7th ed. Chicago: Springer; 2010. p. 117–26.
Ohno T, Aihara R, Kamiyama Y, Mochiki E, Asao T, Kuwano H. Prognostic significance of combined expression of MUC1 and adhesion molecules in advanced gastric cancer. Eur J Cancer. 2006;42(2):256–63.
Yoshikawa R, Nakano Y, Tao L, Koishi K, Matsumoto T, Sasako M, et al. Hedgehog signal activation in oesophageal cancer patients undergoing neoadjuvant chemoradiotherapy. Br J Cancer. 2008;98:1670–4.
Rosivatz E, Becker I, Specht K, Fricke E, Luber B, Busch R, et al. Differential expression of the epithelial−mesenchymal transition regulators snail, SIP-1, and twist in gastric cancer. Am J Pathol. 2002;161(5):1881–91.
Dessaud E, McMahon AP, Briscoe J. Pattern formation in the vertebrate neural tube: a sonic hedgehog morphogen-regulated transcriptional network. Development. 2008;135(15):2489–503.
Gupta S, Takebe N, LoRusso P. Targeting the hedgehog pathway in cancer. Ther Adv Med Oncol. 2010;2:237–50.
Katoh Y, Katoh M. Hedgehog signaling pathway and gastric cancer. Cancer Biol Ther. 2005;4:1050–4.
Ma X, Chen K, Huang S, Zhang X, Adegboyega PA, Evers BM, et al. Frequent activation of the hedgehog pathway in advanced gastric adenocardinomas. Carcinogenesis. 2005;26:1698–705.
Kim JY, Ko GH, Lee YJ, Ha WS, Choi SK, Jung EJ, et al. Prognostic value of hedgehog protein expression in gastric cancer. Jpn J Clin Oncol. 2012;42:1054–9.
Kamikihara T, Ishigami S, Arigami T, Matsumoto M, Okumura H, Uchikado Y, et al. Clinical implications of N-cadherin expression in gastric cancer. Pathol Int. 2012;62:161–6.
Ohta H, Aoyagi K, Fukaya M, Danjoh I, Ohta A, Isohata N, et al. Cross talk between hedgehog and epithelial−mesenchymal transition pathways in gastric pit cells and in diffuse-type gastric cancers. Br J Cancer. 2009;100:389–98.
Yoo YA, Kang MH, Lee HJ, Kim BH, Park JK, Kim HK, et al. Sonic hedgehog pathway promotes metastasis and lymphangiogenesis via activation of Akt, EMT, and MM-9 pathway in gastric cancer. Cancer Res. 2011;71:7061–70.
Kim MA, Lee HS, Lee HE, Kim JH, Yang HK, Kim WH. Prognostic importance of epithelial−mesenchymal transition-related protein expression in gastric carcinoma. Histopathology. 2009;54(4):442–51.
Katoh Y, Katoh M. Hedgehog signaling, epithelial-to-mesenchymal transition and miRNA (review). Int J Mol Med. 2008;22(3):271–5. Review.
Katoh M. Epithelial−mesenchymal transition in gastric cancer. Int J Oncol. 2005;27:1677–83.
Matsumura T, Makino R, Mitamura K. Frequent down-regulation of E-cadherin by genetic and epigenetic changes in the malignant progress of hepatocellular carcinomas. Clin Cancer Res. 2001;7(3):594–9.
Saze Z, Terashima M, Kogure M, Ohsuka F, Suzuki H, Gotoh M. Activation of the sonic hedgehog pathway and its prognostic impact in patients with gastric cancer. Dig Surg. 2012;29(2):115–23.
Wei L, Xu Z. Cross-signaling among phosphoinositide 3-kinase, mitogen-activated protein kinase and sonic hedgehog pathways in esophageal cancer. Int J Cancer. 2011;129:275–84.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Additional information
Zhan-shan Wang and Yang Shen contributed equally to this study.
Rights and permissions
About this article
Cite this article
Wang, Zs., Shen, Y., Li, X. et al. Significance and prognostic value of Gli-1 and Snail/E-cadherin expression in progressive gastric cancer. Tumor Biol. 35, 1357–1363 (2014). https://doi.org/10.1007/s13277-013-1185-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-013-1185-1