Abstract
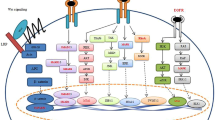
The objective of this study was to explore the expression and clinical significance of Notch signaling genes in colorectal cancer. Colorectal cancer samples were prospectively collected from patients post-surgery at the 3rd Affiliated Hospital, Nanjing University of Traditional Chinese Medicine. Immunohistochemistry of tissue arrays was used to analyze the samples and genes involved in the Notch signaling pathway. Microsatellite instability (MSI) was detected by fluorescence multiplex polymerase chain reaction. A total of 146 colorectal cancer samples were collected, including 84 men (57.7%) and 62 women (42.5%). The average age of the study population was 60.8 ± 10.5 years. Notch1 and Notch2 gene expression correlated with tumor pathology type and degree of differentiation. In addition, Jagged 1 (JAG1) and hairy enhancer of split 1 gene expression correlated with the degree of tumor differentiation. Delta-like 1 gene expression varied significantly with tumor location. There was a significant difference between gene expression and MSI. Of the 138 patients, 134 (91.8%) participated in on-site visits, and the average follow-up time was 42.3 ± 13.3 months. During this period, 86 patients (71.6%) were tumor-free. At 1 year post-surgery, 93% of patients were alive, 74% of patients lived for 3 years, and 67% of patients lived for 5 years. The log-rank test was used to perform univariate analysis, and the COX proportional hazards model was used for the multivariate analysis. Based on these analyses, tumor prognosis correlated with the TNM stage, pathological type, microsatellite status as well as Notch2 and JAG1 gene expression. Patients expressing high levels of Notch2 and JAG1 presented with a significantly better prognosis compared to patients expressing negative or weak levels of Notch2 and JAG1. The expression levels of genes associated with the Notch signaling pathway correlated with tumor pathology and the degree of differentiation. In addition, Notch2 and JAG1 expression levels correlated with survival; however, the underlying mechanism for these correlations remains unclear.



Similar content being viewed by others
References
Jemal A, Thomas A, Murray T, Thun M. Cancer statistics. CA Cancer J Clin. 2002;52:23–47.
Radtke F, Raj K. The role of NOTCH in tumorigenesis: oncogene or tumor suppressor? Nature Rev. 2003;3:756–67.
Artavanis-Tsakonas S, Rand MD, Lake RJ. Notch signaling: cell fate control and signal integration in development. Science. 1999;284:770–6.
Qiao L, Wong B. Role of Notch signaling in colorectal cancer. Carcinogenesis. 2009;30:1979–86.
Zhang Y, Li B, Ji ZZ, Zheng PS. Notch1 regulates the growth of human colon cancers. Cancer. 2010;116:5207–18.
Radtke F, Clevers H. Self-renewal and cancer of the gut: two sides of a coin. Science. 2005;307:1904–9.
Sikandar SS, Pate KT, Anderson S, et al. Activation of Notch signaling in human colon adenocarcinoma. Int J Oncol. 2008;33:1223–9.
Jin HY, Liu X, Li VK, Ding Y, Yang B, Geng J, Lai R, Ding S, Ni M, Zhao R. Detection of mismatch repair gene germline mutation carrier among Chinese population with colorectal cancer. BMC Cancer. 2008;8:44.
Wang J. Current and future situation of colorectal carcinoma screening. J Dig Surg. 2006;5:303–5.
Subramaniam D, Ramalingam S, Houchen CW, Anant S. Cancer stem cells: a novel paradigm for cancer prevention and treatment. Mini Rev Med Chem. 2010;10:359–71.
Koch U, Radtke F. Notch and cancer: a double-edged sword. Cell Mol Life Sci. 2007;64:2746–62.
Sang L, Coller HA, Roberts JM. Control of the reversibility of cellular quiescence by the transcriptional repressor HES1. Science. 2008;321:1095–100.
Katoh M, Katoh M. Integrative genomic analyses on HES/HEY family: Notch-independent HES1, HES3 transcription in undifferentiated ES cells, and Notch-dependent HES1, HES5, HEY1, HEY2, HEYL transcription in fetal tissues, adult tissues, or cancer. Int J Oncol. 2007;31:461–6.
Indraccolo S, Minuzzo S, Masiero M, Pusceddu I, Persano L, Moserle L, Reboldi A, Favaro E, Mecarozzi M, Di Mario G, Screpanti I, Ponzoni M, Doglioni C, Amadori A. Cross-talk between tumor and endothelial cells involving the Notch3-Dll4 interaction marks escape from tumor dormancy. Cancer Res. 2009;69:1314–23. 15.
Rodilla V, Villanueva A, Obrador-Hevia A, Robert-Moreno A, Fernández-Majada V, Grilli A, López-Bigas N, Bellora N, Albà MM, Torres F, Duñach M, Sanjuan X, Gonzalez S, Gridley T, Capella G, Bigas A, Espinosa L. Jagged1 is the pathological link between Wnt and Notch pathways in colorectal cancer. Proc Natl Acad Sci U S A. 2009;106:6315–20.
Peignon G, Durand A, Cacheux W, Ayrault O, Terris B, Laurent-Puig P, Shroyer N, Van Seuningen I, Honjo T, Perret C, Romagnolo B. Complex interplay between b-catenin signaling and Notch effectors in intestinal tumorigenesis. Gut. 2011;60:166–76.
Chu D, Zhang Z, Zhou Y, Wang W, Li Y, Zhang H, Dong G, Zhao Q, Ji G. Notch1 and Notch2 have opposite prognostic effects on patients with colorectal cancer. Ann Oncol. 2011;22:2440–7.
Ungerbäck J, Elander N, Grünberg J, Sigvardsson M, Söderkvist P. The Notch-2 gene is regulated by Wnt signaling in cultured colorectal cancer cells. PLoS One. 2011;6(3):e17957. 18.
Schreck KC, Taylor P, Marchionni L, Gopalakrishnan V, Bar EE, Gaiano N, Eberhart CG. The Notch target Hes1 directly modulates Gli1 expression and Hedgehog signaling: a potential mechanism of therapeutic resistance. Clin Cancer Res. 2010;16:6060–70.
Acknowledgments
This study was funded by the National Nature Science Foundation of China (nos. 30572447 and 30973837).
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Additional information
Hei-Ying Jin and Hong-Ying Zhang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Jin, HY., Zhang, HY., Wang, X. et al. Expression and clinical significance of Notch signaling genes in colorectal cancer. Tumor Biol. 33, 817–824 (2012). https://doi.org/10.1007/s13277-011-0301-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-011-0301-3