Abstract
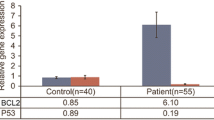
Programmed cell death plays a vital role in a wide variety of physiological processes. Defects in apoptotic cell death contribute to neoplastic diseases by preventing or delaying normal cell death. BCL2 (Bcl-2) is an anti-apoptotic gene with marked up-regulation in various malignancies, such as breast cancer, in which expression of the BCL2 protein has been proposed as a prognostic tumor biomarker. The purpose of the current study was to investigate mRNA expression of the BCL2 gene in nasopharyngeal carcinoma (NPC) biopsies and assess its prognostic value. For this purpose, total RNA was isolated from 89 malignant and hyperplastic nasopharyngeal biopsies from Tunisian patients. After testing the quality of the extracted RNA, cDNA was prepared by reverse transcription. A highly sensitive real-time PCR methodology for BCL2 mRNA quantification was developed using SYBR® Green chemistry. GAPDH served as an endogenous control gene. Relative quantification analysis was performed using the comparative C T (2−∆∆CT) method. High BCL2 mRNA levels were detected in advanced-stage nasopharyngeal tumors (p = 0.030). Furthermore, BCL2 mRNA expression was strongly associated with lymph node involvement (p = 0.009) and presence of distal metastases (p = 0.013). Survival analysis demonstrated that patients with BCL2-positive nasopharyngeal tumors have significantly shorter disease-free and overall survival (p = 0.011 and p = 0.028, respectively). The major contribution of the current study is the quantification and evaluation, for the first time, of the prognostic significance of the BCL2 mRNA expression in nasopharyngeal carcinoma (NPC) patients. Our results suggest that mRNA expression levels of BCL2 may represent a novel unfavorable and independent tumor biomarker for nasopharyngeal carcinoma.


Similar content being viewed by others
References
Kerr JF, Wyllie AH, Currie AR. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer. 1972;26:239–57.
Thompson CB. Apoptosis in the pathogenesis and treatment of disease. Science. 1995;267:1456–62.
Thomadaki H, Scorilas A. Bcl2 family of apoptosis-related genes: functions and clinical implications in cancer. Crit Rev Clin Lab Sci. 2006;43:1–67.
Evan GI, Vousden KH. Proliferation, cell cycle and apoptosis in cancer. Nature. 2001;411:342–8.
Reed JC. Dysregulation of apoptosis in cancer. J Clin Oncol. 1999;17:2941–53.
Adams JM, Cory S. The bcl-2 protein family: arbiters of cell survival. Science. 1998;281:1322–6.
Youle RJ, Strasser A. The bcl-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol. 2008;9:47–59.
Petros AM, Olejniczak ET, Fesik SW. Structural biology of the bcl-2 family of proteins. Biochim Biophys Acta. 2004;1644:83–94.
Korsmeyer SJ. Bcl-2 gene family and the regulation of programmed cell death. Cancer Res. 1999;59:1693s–700.
de Jong D, Prins FA, Mason DY, Reed JC, van Ommen GB, Kluin PM. Subcellular localization of the bcl-2 protein in malignant and normal lymphoid cells. Cancer Res. 1994;54:256–60.
Krajewski S, Tanaka S, Takayama S, Schibler MJ, Fenton W, Reed JC. Investigation of the subcellular distribution of the bcl-2 oncoprotein: residence in the nuclear envelope, endoplasmic reticulum, and outer mitochondrial membranes. Cancer Res. 1993;53:4701–14.
Tsujimoto Y, Cossman J, Jaffe E, Croce CM. Involvement of the bcl-2 gene in human follicular lymphoma. Science. 1985;228:1440–3.
Cleary ML, Smith SD, Sklar J. Cloning and structural analysis of cdnas for bcl-2 and a hybrid bcl-2/immunoglobulin transcript resulting from the t(14;18) translocation. Cell. 1986;47:19–28.
Katsumata M, Siegel RM, Louie DC, Miyashita T, Tsujimoto Y, Nowell PC, et al. Differential effects of bcl-2 on t and b cells in transgenic mice. Proc Natl Acad Sci U S A. 1992;89:11376–80.
Reed JC. Apoptosis-targeted therapies for cancer. Cancer Cell. 2003;3:17–22.
Lee KH, Im SA, Oh DY, Lee SH, Chie EK, Han W, et al. Prognostic significance of bcl-2 expression in stage iii breast cancer patients who had received doxorubicin and cyclophosphamide followed by paclitaxel as adjuvant chemotherapy. BMC Cancer. 2007;7:63.
Martin B, Paesmans M, Berghmans T, Branle F, Ghisdal L, Mascaux C, et al. Role of bcl-2 as a prognostic factor for survival in lung cancer: a systematic review of the literature with meta-analysis. Br J Cancer. 2003;89:55–64.
Sun SY, Yue P, Zhou JY, Wang Y, Choi Kim HR, Lotan R, et al. Overexpression of bcl2 blocks tnf-related apoptosis-inducing ligand (trail)-induced apoptosis in human lung cancer cells. Biochem Biophys Res Commun. 2001;280:788–97.
Krajewski S, Blomqvist C, Franssila K, Krajewska M, Wasenius VM, Niskanen E, et al. Reduced expression of proapoptotic gene bax is associated with poor response rates to combination chemotherapy and shorter survival in women with metastatic breast adenocarcinoma. Cancer Res. 1995;55:4471–8.
Gee JM, Robertson JF, Ellis IO, Willsher P, McClelland RA, Hoyle HB, et al. Immunocytochemical localization of bcl-2 protein in human breast cancers and its relationship to a series of prognostic markers and response to endocrine therapy. Int J Cancer. 1994;59:619–28.
Lipponen P, Pietilainen T, Kosma VM, Aaltomaa S, Eskelinen M, Syrjanen K. Apoptosis suppressing protein bcl-2 is expressed in well-differentiated breast carcinomas with favourable prognosis. J Pathol. 1995;177:49–55.
Schorr K, Li M, Krajewski S, Reed JC, Furth PA. Bcl-2 gene family and related proteins in mammary gland involution and breast cancer. J Mammary Gland Biol Neoplasia. 1999;4:153–64.
Campos L, Rouault JP, Sabido O, Oriol P, Roubi N, Vasselon C, et al. High expression of bcl-2 protein in acute myeloid leukemia cells is associated with poor response to chemotherapy. Blood. 1993;81:3091–6.
Karakas T, Maurer U, Weidmann E, Miething CC, Hoelzer D, Bergmann L. High expression of bcl-2 mrna as a determinant of poor prognosis in acute myeloid leukemia. Ann Oncol. 1998;9:159–65.
Bairey O, Zimra Y, Shaklai M, Okon E, Rabizadeh E. Bcl-2, bcl-x, bax, and bak expression in short- and long-lived patients with diffuse large b-cell lymphomas. Clin Cancer Res. 1999;5:2860–6.
Nicholls JM, Agathanggelou A, Fung K, Zeng X, Niedobitek G. The association of squamous cell carcinomas of the nasopharynx with epstein-barr virus shows geographical variation reminiscent of Burkitt's lymphoma. J Pathol. 1997;183:164–8.
Busson P, Keryer C, Ooka T, Corbex M. Ebv-associated nasopharyngeal carcinomas: from epidemiology to virus-targeting strategies. Trends Microbiol. 2004;12:356–60.
Khabir A, Sellami A, Sakka M, Ghorbel AM, Daoud J, Frikha M, et al. Contrasted frequencies of p53 accumulation in the two age groups of North African nasopharyngeal carcinomas. Clin Cancer Res. 2000;6:3932–6.
Daoud J, Toumi N, Bouaziz M, Ghorbel A, Jlidi R, Drira MM, et al. Nasopharyngeal carcinoma in childhood and adolescence: analysis of a series of 32 patients treated with combined chemotherapy and radiotherapy. Eur J Cancer. 2003;39:2349–54.
Chang ET, Adami HO. The enigmatic epidemiology of nasopharyngeal carcinoma. Cancer Epidemiol Biomark Prev. 2006;15:1765–77.
Feng BJ, Jalbout M, Ayoub WB, Khyatti M, Dahmoul S, Ayad M, et al. Dietary risk factors for nasopharyngeal carcinoma in Maghrebian countries. Int J Cancer. 2007;121:1550–5.
Lo KW, To KF, Huang DP. Focus on nasopharyngeal carcinoma. Cancer Cell. 2004;5:423–8.
Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100:57–70.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-delta delta c(t)) method. Methods. 2001;25:402–8.
Giulietti A, Overbergh L, Valckx D, Decallonne B, Bouillon R, Mathieu C. An overview of real-time quantitative PCR: applications to quantify cytokine gene expression. Methods. 2001;25:386–401.
Camp RL, Dolled-Filhart M, Rimm DL. X-tile: a new bio-informatics tool for biomarker assessment and outcome-based cut-point optimization. Clin Cancer Res. 2004;10:7252–9.
Cammoun M, Hoerner V, Mourali N. Tumors of the nasopharynx in Tunisia. An anatomic and clinical study based on 143 cases. Cancer. 1974;33:184–92.
Van Houten N, Blake SF, Li EJ, Hallam TA, Chilton DG, Gourley WK, et al. Elevated expression of bcl-2 and bcl-x by intestinal intraepithelial lymphocytes: resistance to apoptosis by glucocorticoids and irradiation. Int Immunol. 1997;9:945–53.
Friedman M, Grey P, Venkatesan TK, Bloch I, Chawla P, Caldarelli DD, et al. Prognostic significance of bcl-2 expression in localized squamous cell carcinoma of the head and neck. Ann Otol Rhinol Laryngol. 1997;106:445–50.
Gallo O, Bianchi S, Porfirio B. Bcl-2 overexpression and smoking history in head and neck cancer. J Natl Cancer Inst. 1995;87:1024–5.
Michaud WA, Nichols AC, Mroz EA, Faquin WC, Clark JR, Begum S, et al. Bcl-2 blocks cisplatin-induced apoptosis and predicts poor outcome following chemoradiation treatment in advanced oropharyngeal squamous cell carcinoma. Clin Cancer Res. 2009;15:1645–54.
Vera-Sempere FJ, Burgos JS, Botella MS, Morera C. Immunohistochemical expression of bcl-2 oncoprotein in EBV-associated nasopharyngeal carcinoma correlated to histological type and survival. Histol Histopathol. 1997;12:9–18.
Fontanini G, Vignati S, Bigini D, Mussi A, Lucchi M, Angeletti CA, et al. Bcl-2 protein: a prognostic factor inversely correlated to p53 in non-small-cell lung cancer. Br J Cancer. 1995;71:1003–7.
Laudanski J, Chyczewski L, Niklinska WE, Kretowska M, Furman M, Sawicki B, et al. Expression of bcl-2 protein in non-small cell lung cancer: correlation with clinicopathology and patient survival. Neoplasma. 1999;46:25–30.
Binder C, Marx D, Overhoff R, Binder L, Schauer A, Hiddemann W. Bcl-2 protein expression in breast cancer in relation to established prognostic factors and other clinicopathological variables. Ann Oncol. 1995;6:1005–10.
Sarac S, Akyol MU, Kanbur B, Poyraz A, Akyol G, Yilmaz T, et al. Bcl-2 and lmp1 expression in nasopharyngeal carcinomas. Am J Otolaryngol. 2001;22:377–82.
Yu Y, Dong W, Li X, Yu E, Zhou X, Li S. Significance of c-myc and bcl-2 protein expression in nasopharyngeal carcinoma. Arch Otolaryngol Head Neck Surg. 2003;129:1322–6.
Acknowledgments
This work was supported by a Greek–Tunisian joint research and technology grant (EPAN.M.4.3.6.1 No 125-E), co-funded by the European Regional Development Funds (70% from the European Union), National Resources–Ministry of Development–General Secretariat for Research and Technology of Greece (30% from the governmental funds), and by the Biopaths Co.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fendri, A., Kontos, C.K., Khabir, A. et al. Quantitative analysis of BCL2 mRNA expression in nasopharyngeal carcinoma: an unfavorable and independent prognostic factor. Tumor Biol. 31, 391–399 (2010). https://doi.org/10.1007/s13277-010-0047-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-010-0047-3