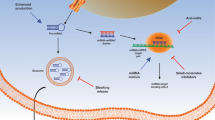
Abstract
Background
Genistein is an isoflavone and phytoestrogen originated from soybean and soy products. Due to the close structural and functional proximity towards 17β-estradiol, genistein has been suggested to influence endocrine and reproductive systems. Previous studies showed that genistein could affect hypothalamic–pituitary–gonadal (HPG) axis and impact gonadotropin-releasing hormone (GnRH) secretion in hypothalamic GT1-7 neurons. However, the underlying mechanism remains mostly unknown.
Objectives
Comparative transcriptomic analyses of mRNAs, long non-coding RNAs (lncRNAs) and microRNAs (miRNAs) were performed in genistein-treated GT1-7 cells by high-throughput RNA sequencing. Competing endogenous RNA (ceRNA) networks were constructed based on potential interactions in lncRNAs, miRNAs and mRNAs.
Results
Compared to the control, 1134, 1126 and 30 differentially expressed mRNA, lncRNA and miRNAs were identified. The most significantly upregulated mRNA was growth-regulating estrogen receptor binding 1 (Greb1), possibly related to the increased levels of estrogen receptors (Esr1 and Esr2). Gene Ontology and Kyoto Encyclopedia of Genes and Genomes enrichment analyses demonstrated that genistein interfered with cell cycle, metabolic processes, as well as GnRH and mitogen-activated protein kinase signaling pathways in GT1-7 cells. CeRNA networks predicted that prostatic cancer-related miRNA mmu-miR-212-5p and its targeted genes Phf2 and Aldh3b1 might be associated with the regulation of genistein-induced GnRH secretion in GT1-7 cells, and 27 lncRNAs could completely interact with mmu-miR-212-5p and downregulate the transcription of target genes.
Conclusion
Results from the study could provide potential targets of both mRNA and non-coding RNAs for further studies to explore the endocrine-interfering effects of genistein.



Similar content being viewed by others
References
Al-Kawlani B et al (2020) Doxorubicin induces cytotoxicity and miR-132 expression in granulosa cells. Reprod Toxicol 96:95–101
Arispe SA, Adams B, Adams TE (2013) Effect of phytoestrogens on basal and GnRH-induced gonadotropin secretion. J Endocrinol 219:243–250
Bateman HL, Patisaul HB (2008) Disrupted female reproductive physiology following neonatal exposure to phytoestrogens or estrogen specific ligands is associated with decreased GnRH activation and kisspeptin fiber density in the hypothalamus. Neurotoxicology 29:988–997
Bliss SP, Navratil AM, Xie J, Roberson MS (2010) GnRH signaling, the gonadotrope and endocrine control of fertility. Front Neuroendocrinol 31(3):322–340. https://doi.org/10.1016/j.yfrne.2010.04.002
Cao C et al (2018) Reproductive role of miRNA in the hypothalamic-pituitary axis. Mol Cell Neurosci 88:130–137
Chen H et al (2019) Identification and functional characterization of microRNAs in rat Leydig cells during development from the progenitor to the adult stage. Mol Cell Endocrinol 493:110453
Cheng W, Li XW, Xiao YQ, Duan SB (2019) Non-coding RNA-associated ceRNA networks in a new contrast-induced acute kidney injury rat model. Mol Ther Nucleic Acids 17:102–112
Day FR et al (2017) Genomic analyses identify hundreds of variants associated with age at menarche and support a role for puberty timing in cancer risk. Nat Genet 49:834–841
Dey BK, Mueller AC, Dutta A (2014) Long non-coding RNAs as emerging regulators of differentiation, development, and disease. Transcription 5:e944014
D’Occhio MJ, Baruselli PS, Campanile G (2019) Influence of nutrition, body condition, and metabolic status on reproduction in female beef cattle: a review. Theriogenology 125:277–284
Fan YX et al (2018) Effect of dietary energy restriction and subsequent compensatory feeding on testicular transcriptome in developing rams. Theriogenology 119:198–207
Fuqua JS (2013) Treatment and outcomes of precocious puberty: an update. J Clin Endocrinol Metab 98:2198–2207
Guzmán A, Hernández-Coronado CG, Rosales-Torres AM, Hernández-Medrano JH (2019) Leptin regulates neuropeptides associated with food intake and GnRH secretion. Ann Endocrinol (paris) 80:38–46
Hayder H et al (2021) Overexpression of miR-210-3p impairs extravillous trophoblast functions associated with uterine spiral artery remodeling. Int J Mol Sci 22:3961
Jaiswal N, Akhtar J, Singh SP, Badruddeen AF (2019) An overview on genistein and its various formulations. Drug Res (stuttg) 69:305–313
Kanasaki H, Oride A, Mijiddorj T, Sukhbaatar U, Kyo S (2017) How is GnRH regulated in GnRH-producing neurons? Studies using GT1-7 cells as a GnRH-producing cell model. Gen Comp Endocrinol 247:138–142
Kim KH et al (2020) Human placenta-derived mesenchymal stem cells stimulate ovarian function via miR-145 and bone morphogenetic protein signaling in aged rats. Stem Cell Res Ther 11:472
Kuiper GG et al (1998) Interaction of estrogenic chemicals and phytoestrogens with estrogen receptor beta. Endocrinology 139:4252–4263
Li Y, Kong D, Ahmad A, Bao B, Sarkar FH (2012) Targeting bone remodeling by isoflavone and 3,3’-diindolylmethane in the context of prostate cancer bone metastasis. PLoS ONE 7:e33011
Li Y et al (2013) Epigenetic reactivation of estrogen receptor-α (ERα) by genistein enhances hormonal therapy sensitivity in ERα-negative breast cancer. Mol Cancer 12:9
Li R, Zhao F, Diao H, Xiao S, Ye X (2014) Postweaning dietary genistein exposure advances puberty without significantly affecting early pregnancy in C57BL/6J female mice. Reprod Toxicol 44:85–92
Li D et al (2017) CYP1A1 based on metabolism of xenobiotics by cytochrome P450 regulates chicken male germ cell differentiation. In Vitro Cell Dev Biol Anim 53:293–303
Li X et al (2020) Screening and evaluating of long non-coding RNAs in prenatal and postnatal pituitary gland of sheep. Genomics 112:934–942
Livadas S, Chrousos GP (2019) Molecular and environmental mechanisms regulating puberty initiation: an integrated approach. Front Endocrinol (lausanne) 10:828
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta C(T)) method. Methods 25:402–408
Losa SM et al (2011) Neonatal exposure to genistein adversely impacts the ontogeny of hypothalamic kisspeptin signaling pathways and ovarian development in the peripubertal female rat. Reprod Toxicol 31:280–289
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:550
Machtinger R et al (2017) Extracellular microRNAs in follicular fluid and their potential association with oocyte fertilization and embryo quality: an exploratory study. J Assist Reprod Genet 34:525–533
Mahesh VB, Zamorano P, De Sevilla L, Lewis D, Brann DW (1999) Characterization of ionotropic glutamate receptors in rat hypothalamus, pituitary and immortalized gonadotropin-releasing hormone (GnRH) neurons (GT1-7 cells). Neuroendocrinology 69:397–407
Mao X, Cai T, Olyarchuk JG, Wei L (2005) Automated genome annotation and pathway identification using the KEGG Orthology (KO) as a controlled vocabulary. Bioinformatics 21:3787–2793
Marchitti SA, Brocker C, Orlicky DJ, Vasiliou V (2010) Molecular characterization, expression analysis, and role of ALDH3B1 in the cellular protection against oxidative stress. Free Radic Biol Med 49:1432–1443
McGarvey C et al (2001) Phytoestrogens and gonadotropin-releasing hormone pulse generator activity and pituitary luteinizing hormone release in the rat. Endocrinology 142:1202–1208
Melamed P et al (2012) Gonadotrophin-releasing hormone signalling downstream of calmodulin. J Neuroendocrinol 24:1463–1475
Mercer TR, Mattick JS (2013) Structure and function of long noncoding RNAs in epigenetic regulation. Nat Struct Mol Biol 20:300–207
Messina M (2014) Soy foods, isoflavones, and the health of postmenopausal women. Am J Clin Nutr 100(Suppl 1):423S-430S
Messina A, Langlet F, Prevot V (2017) MicroRNAs: new players in the hypothalamic control of fertility. Med Sci (paris) 33:506–511
Mukund V (2020) Genistein: its role in breast cancer growth and metastasis. Curr Drug Metab 21:6–10
Pappa S, Padilla N, Iacobucci S, Vicioso M, Álvarez de la Campa E, Navarro C, Marcos E, de la Cruz X, Martínez-Balbás MA (2019) PHF2 histone demethylase prevents DNA damage and genome instability by controlling cell cycle progression of neural progenitors. Proc Natl Acad Sci USA 116(39):19464–19473. https://doi.org/10.1073/pnas.1903188116
Park C et al (2019) Induction of G2/M cell cycle arrest and apoptosis by genistein in human bladder cancer T24 cells through inhibition of the ROS-dependent PI3k/Akt signal transduction pathway. Antioxidants (basel) 8:327
Patisaul HB (2017) Endocrine disruption of vasopressin systems and related behaviors. Front Endocrinol (lausanne) 8:134
Patisaul HB, Fortino AE, Polston EK (2007) Differential disruption of nuclear volume and neuronal phenotype in the preoptic area by neonatal exposure to genistein and bisphenol-A. Neurotoxicology 28:1–12
Qi JC et al (2021) CDK13 upregulation-induced formation of the positive feedback loop among circCDK13, miR-212-5p/miR-449a and E2F5 contributes to prostate carcinogenesis. J Exp Clin Cancer Res 40:2
Rae JM et al (2005) GREB 1 is a critical regulator of hormone dependent breast cancer growth. Breast Cancer Res Treat 92:141–149
Shioda T et al (2013) Expressomal approach for comprehensive analysis and visualization of ligand sensitivities of xenoestrogen responsive genes. Proc Natl Acad Sci USA 110:16508–16513
Stamou M et al (2020) A balanced translocation in Kallmann syndrome implicates a long noncoding RNA, RMST, as a GnRH neuronal regulator. J Clin Endocrinol Metab 105:e23-44
Tan S et al (2021) Comprehensive transcriptome analysis of hypothalamus reveals genes associated with disorders of sex development in pigs. J Steroid Biochem Mol Biol 210:105875
Thangavel P, Puga-Olguín A, Rodríguez-Landa JF, Zepeda RC (2019) Genistein as potential therapeutic candidate for menopausal symptoms and other related diseases. Molecules 24:3892
Xiong J et al (2022) Genistein affects gonadotrophin-releasing hormone secretion in GT1-7 cells via modulating kisspeptin receptor and key regulators. Syst Biol Reprod Med 2022:1–13
Xu Z et al (2018) miR-17-3p downregulates mitochondrial antioxidant enzymes and enhances the radiosensitivity of prostate cancer cells. Mol Ther Nucleic Acids 13:64–77
Young MD, Wakefield MJ, Smyth GK, Oshlack A (2010) Gene ontology analysis for RNA-seq: accounting for selection bias. Genome Biol 11:R14
Yuen T, Ruf F, Chu T, Sealfon SC (2009) Microtranscriptome regulation by gonadotropin-releasing hormone. Mol Cell Endocrinol 302:12–17
Zhang X et al (2009) MicroRNA-17-3p is a prostate tumor suppressor in vitro and in vivo, and is decreased in high grade prostate tumors analyzed by laser capture microdissection. Clin Exp Metastasis 26:965–979
Zhang Q, Bao J, Yang J (2019) Genistein-triggered anticancer activity against liver cancer cell line HepG2 involves ROS generation, mitochondrial apoptosis, G2/M cell cycle arrest and inhibition of cell migration. Arch Med Sci 15:1001–1009
Zhou L et al (2010) Integrated profiling of microRNAs and mRNAs: microRNAs located on Xq27.3 associate with clear cell renal cell carcinoma. PLoS ONE 5:e15224
Zhou X et al (2016) The aberrantly expressed miR-193b-3p contributes to preeclampsia through regulating transforming growth factor-β signaling. Sci Rep 6:19910
Acknowledgements
This study was funded by Department of Science and Technology of Sichuan Province (Grant 2021YJ0156), the Study of Diet and Nutrition Assessment and Intervention Technology (No. 2020YFC2006300) from Active Health and Aging Technologic Solutions Major Project of National Key R&D Program, and the National Nature Science Foundation of China (Grant 82173512).
Author information
Authors and Affiliations
Contributions
JX, YT and GC designed the study. JX, YT, GM and AL conducted the experiments. JX and YT analyzed and interpreted the data. JX, SS and GC provided reagents. JX and AL prepared the manuscript. JX, SS and GC reviewed and revised the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
JX states that there is no conflict of interest to disclose. YT states that there is no conflict of interest to disclose. GM states that there is no conflict of interest to disclose. AL states that there is no conflict of interest to disclose. SS states that there is no conflict of interest to disclose. GC states that there is no conflict of interest to disclose.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.





Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xiong, J., Tian, Y., Ma, G. et al. Comparative RNA-seq analysis and ceRNA network of genistein-treated GT1-7 neurons. Mol. Cell. Toxicol. 19, 499–507 (2023). https://doi.org/10.1007/s13273-022-00279-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-022-00279-1