Abstract
Backgrounds
Mast cells play a major role in allergic inflammation by the release of histamine, an important mediator of type I hypersensitivity. Cencerns regarding potential harmful effects of perfluorooctane sulfonate (PFOS) have been raised. Previous studies reported that PFOS causes various adverse effects such as immunotoxicity and neurotoxicity. This report studied whether PFOS affects mast cells-mediated allergic inflammation.
Methods
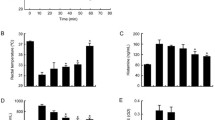
Ovalbumin-induced active systemic anaphylaxis model was used to assess for the type I hypersensitivity. After sensitization, mice were orally administered with PFOS and then allergic symptoms such as hypothermia and increase of serum allergic mediator were measured. In additional, this study investigated whether PFOS deteriorate allergic inflammation in immunoglobulin E-stimulated mast cells.
Results
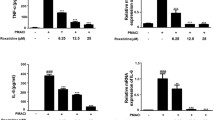
PFOS aggravated the allergic symptoms such as hypothermia, and increase of serum histamine, tumor necrosis factor-α and immunoglobulin (Ig) E/ G1. PFOS increased the release of histamine and β-hexosaminidase through the up-regulation of intracellular calcium in IgE-stimulated mast cells. PFOS also enhanced the gene expression of pro-inflammatory cytokines by activating nuclear factor-κB.
Conclusion
This study demonstrated that PFOS more intensifies the mast cell-mediated allergic inflammation.
Similar content being viewed by others
References
Genuis, S. J., Beesoon, S. & Birkholz, D. Biomonitoring and elimination of perfluorinated compounds and polychlorinated biphenyls through perspiration: blood, urine, and sweat study. ISRN Toxicol 2013, 483832 (2013).
Tian, H., Gao, J., Li, H., Boyd, S. A. & Gu, C. Complete defluorination of perfluorinated compounds by hydrated electrons generated from 3-indole-acetic-acid in organomodified montmorillonite. Sci Rep 6, 32949 (2016).
Olsen, G. W. et al. Half-life of serum elimination of perfluorooctanesulfonate,perfluorohexanesulfonate, and perfluorooctanoate in retired fluorochemical production workers. Environ Health Perspect 115, 1298–1305 (2007).
Harada, K. H. & Koizumi, A. Environmental and biological monitoring of persistent fluorinated compounds in Japan and their toxicities. Environ Health Prev Med 14, 7–19 (2009).
Granum, B. et al. Pre-natal exposure to perfluoroalkyl substances may be associated with altered vaccine antibody levels and immune-related health outcomes in early childhood. J Immunotoxicol 10, 373–379 (2013).
Austin, M. E. et al. Neuroendocrine effects of perfluorooctane sulfonate in rats. Environ Health Perspect 111, 1485–1489 (2003).
Florentin, A., Deblonde, T., Diguio, N., Hautemaniere, A. & Hartemann, P. Impacts of two perfluorinated compounds (PFOS and PFOA) on human hepatoma cells: cytotoxicity but no genotoxicity? Int J Hyg Environ Health 214, 493–499 (2011).
Hainsworth, T. Raising awareness of the rise in allergy-related conditions. Nurs Times 99, 22–23 (2003).
Amin, K. The role of mast cells in allergic inflammation. Respir Med 106, 9–14 (2012).
Castle, J. D., Guo, Z. & Liu, L. Function of the t-SNARE SNAP-23 and secretory carrier membrane proteins (SCAMPs) in exocytosis in mast cells. Mol Immunol 38, 1337–1340 (2002).
Gwack, Y., Feske, S., Srikanth, S., Hogan, P. G. & Rao, A. Signalling to transcription: store-operated Ca2+ entry and NFAT activation in lymphocytes. Cell Calcium 42, 145–156 (2007).
Boyce, J. A. Mast cells: beyond IgE. J Allergy Clin Immunol 111, 24–32 (2003).
Rivera, J. & Gilfillan, A. M. Molecular regulation of mast cell activation. J Allergy Clin Immunol 117, 1214–1225 (2006).
Singh, T. S., Lee, S., Kim, H. H., Choi, J. K. & Kim, S. H. Perfluorooctanoic acid induces mast cell-mediated allergic inflammation by the release of histamine and inflammatory mediators. Toxicol Lett 210, 64–70 (2012).
Yamaki, K. & Yoshino, S. Enhancement of Fcepsilon-RI-mediated degranulation response in the rat basophilic leukemia cell line RBL-2H3 by the fluorosurfactants perfluorooctanoic acid and perfluorooctane sulfonate. Environ Toxicol Pharmacol 29, 183–189 (2010).
Je, I. G. et al. Tyrosol suppresses allergic inflammation by inhibiting the activation of phosphoinositide 3-kinase in mast cells. PLoS One 10, e0129829 (2015).
Je, I. G. et al. SG-HQ2 inhibits mast cell-mediated allergic inflammation through suppression of histamine release and pro-inflammatory cytokines. Exp Biol Med (Maywood) 240, 631–638 (2015).
Bae, Y., Lee, S. & Kim, S. H. Chrysin suppresses mast cell-mediated allergic inflammation: involvement of calcium, caspase-1 and nuclear factor-kappaB. Toxicol Appl Pharmacol 254, 56–64 (2011).
Yoon, S. Y. et al. 1-palmitoyl-2-linoleoyl-3-acetyl-racglycerol (EC-18) modulates Th2 immunity through attenuation of IL-4 expression. Immune Netw 15, 100–109 (2015).
Finkelman, F. D., Rothenberg, M. E., Brandt, E. B., Morris, S. C. & Strait, R. T. Molecular mechanisms of anaphylaxis: lessons from studies with murine models. J Allergy Clin Immunol 115, 449–457 (2005).
Galli, S. J. & Tsai, M. IgE and mast cells in allergic disease. Nat Med 18, 693–704 (2012).
Tak, P. P. & Firestein, G. S. NF-kappaB: a key role in inflammatory diseases. J Clin Invest 107, 7–11 (2001).
Post, G. B., Cohn, P. D. & Cooper, K. R. Perfluorooctanoic acid (PFOA), an emerging drinking water contaminant: a critical review of recent literature. Environ Res 116, 93–117 (2012).
Loccisano, A. E., Longnecker, M. P., Campbell, J. L., Jr., Andersen, M. E. & Clewell, H. J., 3rd. Development of PBPK models for PFOA and PFOS for human pregnancy and lactation life stages. J Toxicol Environ Health A 76, 25–57 (2013).
Tonnelier, A., Coecke, S. & Zaldivar, J. M. Screening of chemicals for human bioaccumulative potential with a physiologically based toxicokinetic model. Arch Toxicol 86, 393–403 (2012).
Kannan, K. et al. Perfluorinated compounds in aquatic organisms at various trophic levels in a Great Lakes food chain. Arch Environ Contam Toxicol 48, 559–566 (2005).
D’Hollander, W., de Voogt, P., De Coen, W. & Bervoets, L. Perfluorinated substances in human food and other sources of human exposure. Rev Environ Contam Toxicol 208, 179–215 (2010).
Fromme, H., Tittlemier, S. A., Volkel, W., Wilhelm, M. & Twardella, D. Perfluorinated compounds-exposure assessment for the general population in Western countries. Int J Hyg Environ Health 212, 239–270 (2009).
Trudel, D. et al. Estimating consumer exposure to PFOS and PFOA. Risk Anal 28, 251–269 (2008).
Kudo, N. & Kawashima, Y. Toxicity and toxicokinetics of perfluorooctanoic acid in humans and animals. J Toxicol Sci 28, 49–57 (2003).
Qazi, M. R. et al. The atrophy and changes in the cellular compositions of the thymus and spleen observed in mice subjected to short-term exposure to perfluorooctanesulfonate are high-dose phenomena mediated in part by peroxisome proliferator-activated receptor-alpha. Toxicology 260, 68–76 (2009).
EFSA. Opinion of the scientific panel on contaminants in the food chain on perfluorooctane sulfonate (PFOS), perfluorooctanoic acid (PFOA) and their salts. The EFSA Journal 653, 1–131 (2008).
ATSDR. in Atlanta: U.S. Department of Health and Human Services (2015).
Haug, L. S., Huber, S., Becher, G. & Thomsen, C. Characterisation of human exposure pathways to perfluorinated compounds-comparing exposure estimates with biomarkers of exposure. Environ Int 37, 687–693 (2011).
Kennedy, G. L., Jr. et al. The toxicology of perfluorooctanoate. Crit Rev Toxicol 34, 351–384 (2004).
Sampson, H. A. et al. Second symposium on the definition and management of anaphylaxis: summary report-second National Institute of Allergy and Infectious Disease/Food Allergy and Anaphylaxis Network symposium. Ann Emerg Med 47, 373–380 (2006).
He, S. H., Zhang, H. Y., Zeng, X. N., Chen, D. & Yang, P. C. Mast cells and basophils are essential for allergies: mechanisms of allergic inflammation and a proposed procedure for diagnosis. Acta Pharmacol Sin 34, 1270–1283 (2013).
Wex, E., Thaler, E., Blum, S. & Lamb, D. A novel model of IgE-mediated passive pulmonary anaphylaxis in rats. PLoS One 9, e116166 (2014).
Osterfeld, H. et al. Differential roles for the IL-9/IL-9 receptor alpha-chain pathway in systemic and oral antigen-induced anaphylaxis. J Allergy Clin Immunol 125, 469–476 e462 (2010).
Ahrens, R. et al. Intestinal mast cell levels control severity of oral antigen-induced anaphylaxis in mice. Am J Pathol 180, 1535–1546 (2012).
Ishikawa, R. et al. IgG-mediated systemic anaphylaxis to protein antigen can be induced even under conditions of limited amounts of antibody and antigen. Biochem Biophys Res Commun 402, 742–746 (2010).
Devey, L., Festing, M. F. & Wigmore, S. J. Effect of temperature control upon a mouse model of partial hepatic ischaemia/reperfusion injury. Lab Anim 42, 12–18 (2008).
Laidlaw, T. M. et al. Characterization of a novel human mast cell line that responds to stem cell factor and expresses functional FcepsilonRI. J Allergy Clin Immunol 127, 815–822 e811-815 (2011).
Nishida, K. et al. Fc{epsilon}RI-mediated mast cell degranulation requires calcium-independent microtubule-dependent translocation of granules to the plasma membrane. J Cell Biol 170, 115–126 (2005).
Suzuki, Y. et al. Fc epsilon RI signaling of mast cells activates intracellular production of hydrogen peroxide: role in the regulation of calcium signals. J Immunol 171, 6119–6127 (2003).
Woolley, D. E. & Tetlow, L. C. Mast cell activation and its relation to proinflammatory cytokine production in the rheumatoid lesion. Arthritis Res 2, 65–74 (2000).
DeWitt, J. C., Peden-Adams, M. M., Keller, J. M. & Germolec, D. R. Immunotoxicity of perfluorinated compounds: recent developments. Toxicol Pathol 40, 300–311 (2012).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Lee, JK., Lee, S., Choi, YA. et al. Perfluorooctane sulfonate exacerbates mast cell-mediated allergic inflammation by the release of histamine. Mol. Cell. Toxicol. 14, 173–181 (2018). https://doi.org/10.1007/s13273-018-0019-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-018-0019-z