Abstract
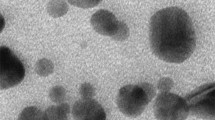
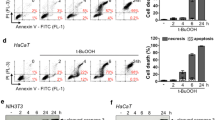
Zinc Oxide nanoparticles (ZnO NPs) in its small size with large reactive surfaces can lead to toxicological injury by generating reactive oxygen species (ROS) and oxidative stress. Recently, ZnO NPs were shown to play a role in acute or chronic toxicities with mammalian cells, where its mechanism of toxicity is not fully characterized yet. In this study, the potential mechanisms of ZnO NPs in inducing and increasing oxidative stress for causing cellular apoptosis were investigated with human keratinocytes. Indeed, ZnO NPs induced significant intracellular ROS and mitochondrial ROS productions. And it seemed that cellular ROS levels induced by ZnO NPs led to the dissipation of the mitochondrial membrane potentials and elicited the cellular apoptosis. The induced ROS production by ZnO NPs was blocked with chelator treatment, which inhibited the ZnO ion. The present study demonstrates that increased levels of ROS by ZnO NPs cause mitochondrial dysfunction and cellular apoptosis.
Similar content being viewed by others
References
Liu, W. T. Nanoparticles and their biological and environmental applications. J Biosci Bioeng 102:1–7 (2006).
Bae, H. C. & Ryu, H. J. et al. Oxidative stress and apoptosis induced by ZnO nanoparticles in HaCaT cells. Mol Cell Toxicol 7:333–337 (2011).
Gojova, A. et al. Induction of inflammation in vascular endothelial cells by metal oxide nanoparticles: effect of particle composition. Environ Health Perspect 115:403–409 (2007).
Yang, H. et al. Comparative study of cytotoxicity, oxidative stress and genotoxicity induced by four typical nanomaterials: the role of particle size, shape and composition. J Appl Toxicol 29:69–78 (2009).
Babior, B. M., Lambeth, J. D. & Nauseef, W. The neutrophil NADPH oxidase. Arch Biochem Biophys 397: 342–344 (2002).
Zamzami, N. et al. Sequential reduction of mitochondrial transmembrane potential and generation of reactive oxygen species in early programmed cell death. J Exp Med 182:367–377 (1995).
Gottlieb, E., Armour, S. M., Harris, M. H. & Thompson, C. B. Mitochondrial membrane potential regulates matrix configuration and cytochrome c release during apoptosis. Cell Death Differ 10:709–717 (2003).
Li, P. F., Dietz, R. & von Harsdorf, R. p53 regulates mitochondrial membrane potential through reactive oxygen species and induces cytochrome c-independent apoptosis blocked by Bcl-2. EMBO J 18:6027–6036 (1999).
Jeong, S. H. et al. ZnO nanoparticles induce TNF-α expression via ROS-ERK-Egr-1 pathway in human keratinocytes. J Dermatol Sci 72:263–273 (2013).
Samberg, M. E., Oldenburg, S. J. & Monteiro-Riviere, N. A. Evaluation of silver nanoparticle toxicity in skin in vivo and keratinocytes in vitro. Environ Health Perspect 118:407–413 (2010).
Manke, A., Wang, L. & Rojanasakul, Y. Mechanisms of nanoparticle-induced oxidative stress and toxicity. Biomed Res Int 2013:942916 (2013).
Sharma, V., Anderson, D. & Dhawan, A. Zinc oxide nanoparticles induce oxidative DNA damage and ROS-triggered mitochondria mediated apoptosis in human liver cells (HepG2). Apoptosis 17:852–870 (2012).
Shen, C. et al. Relating cytotoxicity, zinc ions, and reactive oxygen in ZnO nanoparticle-exposed human immune cells. Toxicol Sci 136:120–130 (2013).
Nohynek, G. J., Dufour, E. K. & Roberts, M. S. Nano-technology, cosmetics and the skin: is there a health risk? Skin Pharmacol Physiol 21:136–149 (2008).
Guo, D. et al. Reactive oxygen species-induced cytotoxic effects of zinc oxide nanoparticles in rat retinal ganglion cells. Toxicol In Vitro 27:731–738 (2013).
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
Rights and permissions
About this article
Cite this article
Ryu, WI., Park, YH., Bae, H.C. et al. ZnO nanoparticle induces apoptosis by ROS triggered mitochondrial pathway in human keratinocytes. Mol. Cell. Toxicol. 10, 387–391 (2014). https://doi.org/10.1007/s13273-014-0043-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-014-0043-6