Abstract
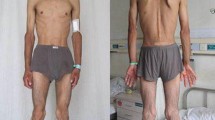
The Mendelian inherited progressive external ophthalmoplegia (PEO) and mitochondrial neurogastrointestinal encephalomyopathy (MNGIE) are genetically heterogeneous mitochondrial diseases caused by nuclear-mitochondrial intergenomic defects. The PEO1 and TYMP nuclear genes are closely related in the machinery of the mitochondrial DNA (mtDNA) replication. Mutations in PEO1 and TYMP genes usually cause autosomal dominant PEO, and autosomal recessive MNGIE. We identified a PEO family of Korean origin with additional phenotype of incomplete MNGIE symptom (Family ID: MT16). The entire mitochondrial genome and all coding exons of PEO1, TYMP, ANT1, POLG1, POLG2, DGUOK, and TK2 nuclear genes were sequenced. Clinical information was obtained through history taking, physical examinations, clinical observations, and electrophysiological investigations. Muscle biopsy of left biceps brachii and shoulder magnetic resonance imaging (MRI) were undertaken. We found two heterozygous mutations, Arg374Gln in PEO1 and Glu106Gln in TYMP from the proband who showed complex phenotypes of a typical PEO and late-onset incomplete MNGIE. The PEO1 Arg374Gln has been reported in several PEO patients, but TYMP Glu106Gln has not been reported. Neither large deletion nor causative point mutations were observed in the mtDNA. We suggest that the heterozygous TYMP mutation might affect complex phenotypes as a secondary genetic cause in the co-presence of PEO1 mutation.
Similar content being viewed by others
References
Baloh RH, Salavaggione E, Milbrandt J, and Pestronk A (2007) Familial parkinsonism and ophthalmoplegia from a mutation in the mitochondrial DNA helicase twinkle. Arch. Neurol. 64: 998–1000.
Bakker JA, Schlesser P, Smeets HJ, Francois B, and Bierau J (2010) Biochemical abnormalities in a patient with thymidine phosphorylase deficiency with fatal outcome. J. Inherit. Metab. Dis. “in press”.
Fratter C, Gorman GS, Stewart JD, Buddles M, Smith C, Evans J, Seller A, Poulton J, Roberts M, Hanna MG, et al. (2010) The clinical, histochemical, and molecular spectrum of PEO1 (Twinkle)-linked adPEO. Neurology 74: 1619–1626.
Gamez J, Ferreiro C, Accarino ML, Guarner L, Tadesse S, Marti RA, Andreu AL, Raguer N, Cervera C, and Hirano M (2002) Phenotypic variability in a Spanish family with MNGIE. Neurology 59: 455–457.
Giordano C, Powell H, Leopizzi M, De Curtis M, Travaglini C, Sebastiani M, Gallo P, Taylor RW, and d’Amati G (2009) Fatal congenital myopathy and gastrointestinal pseudo-obstruction due to POLG1 mutations. Neurology 72: 1103–1105.
Goffart S, Cooper HM, Tyynismaa H, Wanrooij S, Suomalainen A, and Spelbrink JN (2009) Twinkle mutations associated with autosomal dominant progressive external ophthalmoplegia lead to impaired helicase function and in vivo mtDNA replication stalling. Hum. Mol. Genet. 18: 328–340.
Hirano M, Nishigaki Y, and Martí R (2004) Mitochondrial neurogastrointestinal encephalomyopathy (MNGIE): a disease of two genomes. Neurologist 10: 8–17.
Horvath R, Hudson G, Ferrari G, Fütterer N, Ahola S, Lamantea E, Prokisch H, Lochmüller H, McFarland R, Ramesh V, et al. (2006) Phenotypic spectrum associated with mutations of the mitochondrial polymerase gamma gene. Brain 129: 1674–1684.
Ingman M, and Gyllensten U (2006) mtDB: Human Mitochondrial Genome Database, a resource for population genetics and medical sciences. Nucleic. Acids. Res. 34: D749–D751.
Kaukonen J, Juselius JK, Tiranti V, Kyttälä A, Zeviani M, Comi GP, Keränen S, Peltonen L, and Suomalainen A (2000) Role of adenine nucleotide translocator 1 in mtDNA maintenance. Science 289: 782–785.
Korhonen JA, Pande V, Holmlund T, Farge G, Pham XH, Nilsson L, and Falkenberg M (2008) Structure-function defects of the TWINKLE linker region in progressive external ophthalmoplegia. J. Mol. Biol. 377: 691–705.
Lamantea E, Tiranti V, Bordoni A, Toscano A, Bono F, Servidei S, Papadimitriou A, Spelbrink H, Silvestri L, Casari G, et al. (2002) Mutations of mitochondrial DNA polymerase gamma-A are a frequent cause of autosomal dominant or recessive progressive external ophthalmoplegia. Ann. Neurol. 52: 211–219.
López LC, Akman HO, García-Cazorla A, Dorado B, Martí R, Nishino I, Tadesse S, Pizzorno G, Shungu D, Bonilla E, et al. (2009) Unbalanced deoxynucleotide pools cause mitochondrial DNA instability in thymidine phosphorylase-deficient mice. Hum. Mol. Genet. 18: 714–722.
Marti R, Verschuuren JJGM, Buchman A, Hirano I, Tadesse S, van Kuilenburg ABP, van Gennip AH, Poorthuis BJHM, and Hirano M (2005) Late-onset MNGIE due to partial loss of thymidine phosphorylase activity. Ann. Neurol. 58: 649–652.
Massa R, Tessa A, Margollicci M, Micheli V, Romigi A, Tozzi G, Terracciano C, Piemonte F, Bernardi G, and Santorelli FM (2009) Late-onset MNGIE without peripheral neuropathy due to incomplete loss of thymidine phosphorylase activity. Neuromuscul. Disord. 19: 837–840.
Naïmi M, Bannwarth S, Procaccio V, Pouget J, Desnuelle C, Pellissier JF, Rötig A, Munnich A, Calvas P, Richelme C, et al. (2006) Molecular analysis of ANT1, TWINKLE and POLG in patients with multiple deletions or depletion of mitochondrial DNA by a dHPLC-based assay. Eur. J. Hum. Genet. 14: 917–922.
Nishino I, Spinazzola A, and Hirano M (1999) Thymidine phosphorylase gene mutations in MNGIE, a human mitochondrial disorder. Science 283: 689–692.
Ruiz-Pesini E, Lott MT, Procaccio V, Poole J, Brandon MC, Mishmar D, Yi C, Kreuziger J, Baldi P, and Wallace DC (2009) An enhanced MITOMAP with a global mtDNA mutational phylogeny. Nucleic Acids Res. 35: D823–D828.
Sarzi E, Goffart S, Serre V, Chretien D, Slama A, Munnich A, Spelbrink JN, and Rotig A (2007) Twinkle helicase (PEO1) gene mutation causes mitochondrial DNA depletion. Ann. Neurol. 62: 579–587.
Spelbrink JN, Li FY, Tiranti V, Nikali K, Yuan QP, Tariq M, Wanrooij S, Garrido N, Comi G, Morandi L, et al. (2001) Human mitochondrial DNA deletions associated with mutations in the gene encoding Twinkle, a phage T7 gene 4-like protein localized in mitochondria. Nat. Genet. 28: 223–231.
Spinazzola A and Zeviani M (2005) Disorders of nuclear-mitochondrial intergenomic signaling. Gene 354: 162–168.
Suomalainen A, and Isohanni P (2010) Mitochondrial DNA depletion syndromes: many genes, common mechanisms. Neuromusc. Disord. 20: 429–437.
Szigeti K, Wong L-JC, Perng C-L, Saifi GM, Eldin K, Adesina AM, Cass DL, Hirano M, Lupski JR, and Scaglia F (2004) MNGIE with lack of skeletal muscle involvement and a novel TP splice site mutation. J. Med. Genet. 41: 125–129.
Tyynismaa H, Sembongi H, Bokori-Brown M, Granycome C, Ashley N, Poulton J, Jalanko A, Spelbrink JN, Holt IJ, and Suomalainen A (2004) Twinkle helicase is essential for mtDNA maintenance and regulates mtDNA copy number. Hum. Mol. Genet. 13: 3219–3227.
Tyynismaa H, Mjosund KP, Wanrooij S, Lappalainen I, Ylikallio E, Jalanko A, Spelbrink JN, Paetau A, and Suomalainen A (2005) Mutant mitochondrial helicase Twinkle causes multiple mtDNA deletions and a late-onset mitochondrial disease in mice. Proc. Natl. Acad. Sci. USA. 102: 17687–17692.
Tyynismaa H, Ylikallio E, Patel M, Molnar MJ, Haller RG, and Suomalainen A (2009) A heterozygous truncating mutation in RRM2B causes autosomal-dominant progressive external ophthalmoplegia with multiple mtDNA deletions. Am. J. Hum. Genet. 85: 290–295.
Author information
Authors and Affiliations
Corresponding author
Additional information
The first two authors, KN and KWC, contributed equally to this work.
Rights and permissions
About this article
Cite this article
Nakhro, K., Chung, K.W., Kim, SM. et al. Compound mutations of PEO1 and TYMP in a progressive external ophthalmoplegia patient with incomplete mitochondrial neurogastrointestinal encephalomyopathy phenotype. Genes Genom 33, 431–437 (2011). https://doi.org/10.1007/s13258-011-0089-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-011-0089-y