Abstract
Objective
In this study, a mechanical circulatory support simulation tool was used to investigate the application of a unique device with two centrifugal pumps and one motor for the biventricular assist device (BVAD) support application. Several conditions—including a range of combined left and right systolic heart failure severities, aortic and pulmonary valve regurgitation, and combinations of high and low systemic and pulmonary vascular resistances—were considered in the simulation matrix. Relative advantages and limitations of using the device in BVAD applications are discussed.
Methods
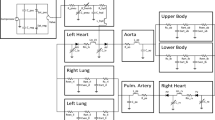
The simulated BVAD pump was based on the Cleveland Clinic pediatric continuous-flow total artificial heart (P-CFTAH), which is currently under development. Different combined disease states (n = 10) were evaluated to model the interaction with the BVAD, considering combinations of normal heart, moderate failure and severe systolic failure of the left and right ventricles, regurgitation of the aortic and pulmonary valves and combinations of vascular resistance. The virtual mock loop simulation tool (MATLAB; MathWorks®, Natick, MA) simulates the hemodynamics at the pump ports using a lumped-parameter model for systemic/pulmonary circulation characteristic inputs (values for impedance, systolic and diastolic ventricular compliance, beat rate, and blood volume), and characteristics of the cardiac chambers and valves.
Results
Simulation results showed that this single-pump BVAD can provide regulated support of up to 5 L/min over a range of combined heart failure states and is suitable for smaller adult and pediatric support. However, good self-regulation of the atrial pressure difference was not maintained with the introduction of aortic valve regurgitation or high systemic vascular resistance when combined with low pulmonary vascular resistance.
Conclusions
This initial in silico study demonstrated that use of the P-CFTAH as a BVAD supports cardiac output and arterial pressure in biventricular heart failure conditions. A similar but larger device would be required for a large adult patient who needs more than 5 L/min of support.









Similar content being viewed by others
References
Dandel, M., T. Krabatsch, and V. Falk. Left ventricular vs. biventricular mechanical support: decision making and strategies for avoidance of right heart failure after left ventricular assist device implantation. Int. J. Cardiol. 198:241–250, 2015.
Fukamachi, K., D. J. Horvath, A. L. Massiello, H. Fumoto, T. Horai, S. Rao, and L. A. Golding. An innovative, sensorless, pulsatile, continuous-flow total artificial heart: device design and initial in vitro study. J. Heart. Lung Transplant 29:13–20, 2010.
Fukamachi, K., J. H. Karimov, D. J. Horvath, G. Sunagawa, N. A. Byram, B. D. Kuban, and N. Moazami. Initial in vitro testing of a paediatric continuous-flow total artificial heart. Interact. Cardiovasc. Thorac. Surg. 26:897–901, 2018.
Horvath, D., N. Byram, J. H. Karimov, B. Kuban, G. Sunagawa, L. A. R. Golding, N. Moazami, and K. Fukamachi. Mechanism of self-regulation and in vivo performance of the Cleveland Clinic continuous-flow total artificial heart. Artif. Organs 41:411–417, 2017.
Horvath, D. J., K. Fukamachi, and J. H. Karimov. Options for modeling and simulation used in mechanical circulatory support development. In: Mechanical Support for Heart Failure, Current Solutions and New Technologies, edited by J. H. Karimov, and et al. New York: Springer, 2020. https://doi.org/10.1007/978-3-030-47809-4.
Horvath, D. J., D. W. Horvath, J. H. Karimov, N. Byram, B. D. Kuban, T. Miyamoto, and K. Fukamachi. Use of a mechanical circulatory support simulation to study pump interactions with the variable hemodynamic environment. Artif. Organs. 42:E420–E427, 2018.
Horvath DJ, Horvath DW, Karimov JH, Kuban BD, Miyamoto T, Fukamachi K. A simulation tool for mechanical circulatory support device interaction with diseased states. J Artif Organs (in press).
Horvath, D. J., J. H. Karimov, N. A. Byram, B. D. Kuban, G. Sunagawa, N. Moazami, and K. Fukamachi. Advantages of integrating pressure-regulating devices into mechanical circulatory support pumps. ASAIO J. 65:e1–e3, 2019.
Karimov, J. H., D. J. Horvath, N. Byram, G. Sunagawa, B. D. Kuban, S. Gao, R. Dessoffy, and K. Fukamachi. Early in vivo experience with the pediatric continuous-flow total artificial heart. J. Heart Lung Transplant. 37:1029–1034, 2018.
Karimov, J. H., N. Moazami, M. Kobayashi, S. Sale, K. Such, N. Byram, et al. First report of 90-day support of 2 calves with a continuous-flow total artificial heart. J. Thorac. Cardiovasc. Surg. 150:e1, 2015.
Miyamoto, T., D. J. Horvath, D. W. Horvath, J. H. Karimov, N. Byram, B. D. Kuban, and K. Fukamachi. Simulated performance of the Cleveland Clinic continuous-flow total artificial heart using the virtual mock loop. ASAIO J. 65:565–572, 2019.
Patent: Horvath DJ GL, Massiello A, Kuban BD. Two-stage rotodynamic blood pump. US9162019B2. In: USPTO, ed. US: Cleveland Clinic Foundation, 2012.
Patent: Horvath DJ KB, Golding LAR. Artificial heart system implementing suction recognition and avoidance methods. US10077777B2. In: USPTO, ed. US: Cleveland Clinic Foundation, 2014.
Shah, P., R. Ha, R. Singh, W. Cotts, E. Adler, M. Kiernan, M. Brambatti, K. Meehan, S. Phillips, S. Kidambi, G. P. Macaluso, D. Banerjee, D. Mooney, D. Pham, and V. D. Pretorius. Multicenter experience with durable biventricular assist devices. J Heart Lung Transplant 37:1093–1101, 2018.
Vierecke, J., B. Gahl, T. de By, H. Antretter, F. Beyersdorf, K. Caliskan, V. Krachak, A. Loforte, E. Potapov, F. Schoenrath, B. Stockman, M. Vanderheyden, B. Meyns, J. Gummert, and P. Mohacsi. Results of primary biventricular support: an analysis of data from the EUROMACS registry. Eur. J. Cardiothorac. Surg. 56:1037–1045, 2019.
Wever-Pinzon, O., S. G. Drakos, A. G. Kfoury, J. N. Nativi, E. M. Gilbert, M. Everitt, R. Alharethi, K. Brunisholz, F. M. Bader, D. Y. Li, C. H. Selzman, and J. Stehlik. Morbidity and mortality in heart transplant candidates supported with mechanical circulatory support: is reappraisal of the current United Network for Organ Sharing thoracic organ allocation policy justified? Circulation 127:452–462, 2013.
Funding
The virtual mock loop software development has been funded by Cleveland Clinic and R1 Engineering.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The simulated device and technology are owned by Cleveland Clinic. David Horvath is a device inventor. All other authors report no conflicts.
Additional information
Associate Editor Scott C. Corbett oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Horvath, D.W., Polakowski, A.R., Flick, C. et al. Modeling of Virtual Mechanical Circulatory Hemodynamics for Biventricular Heart Failure Support. Cardiovasc Eng Tech 11, 699–707 (2020). https://doi.org/10.1007/s13239-020-00501-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-020-00501-y