Abstract
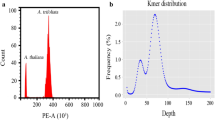
Tinospora cordifolia (Giloy), with immense therapeutic properties has been used as a powerful rasayana and immunomodulator in alternative systems of medicine. In the present study, two distinct morphotypes (arbitrarily labeled 1 and 2) were identified in the wild Indian giloy populations. Flow cytometric analysis for DNA quantification revealed 1.5-fold moreDNA (2C = 4.32 pg) in morphotype 2 than in morphotype 1 (2C = 2.8 pg). The cytogenetic analysis showed the somatic cells of morphotype 2 to possess triploid (2n = 3x = 39; first report in giloy) compared to diploid (2n = 2x = 26) chromosome number in morphotype 1. While the diploid T. cordifolia and the allied diploid T. sinensis cytotypes exhibited normal meiosis, the triploids showed highly irregular meiosis with average (10.80 II) number of bivalents per cell outnumbering trivalents (3.52 III) and univalents (6.68 I). The chloroplast PCR–RFLP analysis in the diploid and triploid T. cordifolia, and T. sinensis involving 224 chloroplast genes/intergenic spacers and 28 restriction enzyme combinations revealed the contribution of diploid T. cordifolia as the maternal progenitor of the triploid cytotype. The present study unveils a new wild triploid giloy cytotype with significantly higher biomass of great commercial advantage and relevance in plant breeding and biotechnology. The cultivation of this novel, and vigorous triploid giloy cytotype is anticipated to bridge the gap between the demand and supply of superior, uniform giloy raw material for the Ayurveda industry and will also help minimize batch-to-batch variations commonly observed in Ayurvedic drugs. We have also characterized the triploid cytotypes to understand their nature and origin to shed light on evolutionary genetic differentiation mechanisms in giloy, a species with relatively obscure genetics.
Graphical abstract







Similar content being viewed by others
Data availability
Most data that supports the findings of the manuscript is included in this MS and available from the authors upon request.
References
Agrawal R, Agrawal N, Tandon R, Raina, SN. Chloroplast genes as genetic markers for inferring patterns of change, maternal ancestry and phylogenetic relationships among Eleusine species. AoB Plants 2014; 6: plt056; doi:https://doi.org/10.1093/aobpla/plt056
Ahmad SM, Hoot SB, Qazi PH, Verma V. Phylogenetic patterns and genetic diversity of Indian Tinospora species based on chloroplast sequence data and cytochrome P450 polymorphisms. Plant Syst Evol. 2009;281:87–96. https://doi.org/10.1007/s00606-009-0189-1.
Ahmed SM, Verma V, Qazi PH, Ganaie MM, Bakshi SK, Qazi GN. Molecular phylogeny in Indian Tinospora species by DNA based molecular markers. Plant Syst Evol. 2006;256:75–87. https://doi.org/10.1007/s00606-004-0293-1.
Ahsan R, Mishra A, Badar B, Owais M, Mishra V. Therapeutic application, phytoactives and pharmacology of tinospora cordifolia: an evocative review. Chin J Integr Med. 2023;29(6):549–55. https://doi.org/10.1007/s11655-023-3733-2.
Bon PV, Harwood CE, Nghiem QC, Thinh HH, Son DH, Chinh NV. Growth of triploid and diploid Acacia clones in three contrasting environments in Viet Nam. Aust For. 2020;83:265–74. .
Chen JZ, Birchler JA. Polyploid and hybrid genomics. Wiley Blackwell, Oxford, UK, 2013.
Cheng S, Huang Z, Li Y, Liao T, Suo Y, Zhang P, Wang J, Kang X. Differential transcriptome analysis between Populus and its synthesized allotriploids driven by second- division restitution. J Integr Plant Biol. 2015;57:1031–45. https://doi.org/10.1111/jipb.12328.
Chi S, She G, Han D, Wang W, Liu Z, Liu B. Genus Tinospora: Ethnopharmacology, Phytochemistry, and Pharmacology. Evidence-Based Complementary and Alternative Medicine, 2016; 1–32. https://doi.org/10.1155/2016/9232593
Doležel J, Greilhuber J, Suda J. Estimation of nuclear DNA content in plants using flow cytometry. Nat Protoc. 2007;2:2233–44. https://doi.org/10.1038/nprot.2007.310.
Erilova A, Brownfield L, Exner V, Rosa M, Twell D, Scheid OM, Hennig L, Köhler C. Imprinting of the polycomb group gene MEDEA serves as a ploidy sensor in Arabidopsis. PLoS Genet. 2009;5: e1000663
Export of herbs and herbal products, Press information bureau of Government of India, Ministry of Commerce and Industry, 2019. https://pib.gov.in/Pressreleaseshare.aspx?PRID=1558955).
Forman LLA. revision of Tinospora (Menispermaceae) in Asia to Australia and the Pacific. Kew Bull. 1981;36(2):375–421. https://doi.org/10.2307/4113613.
Gautam A, Kaur H, Kaur A, Prashar P K, Sood A, Singh S.K, Gulati M, Pandey N. K, and Kumar, B. Tinospora cordifolia: A Successful Story from Botanical background to Pharmaceutical Product. Research J. Pharm. and Tech. 2020; 13(11):5620–5630. https://doi.org/10.5958/0974-360X.2020.00980.4 Available on: https://rjptonline.org/AbstractView.aspx?PID=2020-13-11-95
Herbal medicines: Global Strategic Business Repot, Jan 2023, https://www.researchandmarkets.com/reports/5302363/herbal-medicines-global-strategic-business-report#tag-pos-5
Hoshino Y, Miyashita T, Thomas TD. In vitro culture of endosperm and its application in plant breeding: approaches to polyploidy breeding. Sci Hortic. 2011;130:1–8.
Jain R, Prasad B. Karyotype analysis and chromosome number confirmation in Tinospora cordifolia (Willd) Miers. Ex. Hook. F. and Thomas. A. International Journal of Pharma and Bio Sciences 2014; 5:996–1001
Kaur K, Sidhu MC. Meiotic studies in some medicinal angiosperms from Doaba region of Punjab, India. Int J Phytomed. 2014;6:216–24.
Kaushal P, Paul S, Saxena S, Dwivedi KK, Chakraborti M, Radhakrishna A, Roy AK, Malaviya, DR. Generating higher ploidies (7x and 11x) in guinea grass (Panicum maximum Jacq.) utilizing reproductive diversity and uncoupled apomixis components. Current Sci. 2015; 109;1392–1395.
Kumar M, Hasan M, Sharma A. Tinospora cordifolia (Willd.) Hook.f. & Thomson polysaccharides: A review on extraction, characterization, and bioactivities. Int J Biol Macromol. 2023;229:463–475. doi:https://doi.org/10.1016/j.ijbiomac.2022.12.181
Lavia GI, Ortiz AM, Robledo G, Fernández A, Seijo G. Origin of triploid Arachis pintoi (Leguminosae) by autopolyploidy evidenced by FISH and meiotic behavior. Ann Bot. 2011;108:103–11. https://doi.org/10.1093/aob/mcr108.
Li Y, Yang J, Song L, Qi Q, Du K, Han Q, Kang X. Study of variation in the growth, photosynthesis, and content of secondary metabolites in Eucommia triploids. Trees. 2019;33:817–26. https://doi.org/10.1007/s00468-019-01818-5.
Liao T, Cheng S, Zhu X, Min Y, Kang X. Effects of triploid status on growth, photosynthesis, and leaf area in Populus. Trees. 2016;30:1137–47. https://doi.org/10.1007/s00468-016-1352-2.
Lourkisti R, Froelicher Y, Herbette S, Morillon R, Tomi F, Gibernau M, Giannettini J, Berti L, Santini J. Triploid citrus genotypes have a better tolerance to natural chilling conditions of photosynthetic capacities and specific leaf volatile organic compounds. Front Plant Sci. 2020;11:330. https://doi.org/10.3389/fpls.2020.00330.
Manjrekar PN, Jolly CI, Narayanan S. Comparative studies of the immunomodulatory activity of Tinospora cordifolia and Tinospora sinensis. Fitoterapia. 2000;71:254–7. https://doi.org/10.1016/S0367-326X(99)00167-7.
Mu HZ, Liu ZJ, Lin L, Li HY, Jiang J, Liu GF. Transcriptomic analysis of phenotypic changes in birch (Betula platyphylla) autotetraploids. Int J Mol Sci. 2012;13:13012–29. https://doi.org/10.3390/ijms131013012.
Murshid G, Kundu S, Sohrab M, Mazid M. Pharmacological overview of tinospora cordifolia, an ethnologically important plant of Bangladesh. Pharmacol Pharm. 2022;13:93–106. https://doi.org/10.4236/pp.2022.133007.
Parisod C, Holderegger R, Brochmann C. Evolutionary consequences of autopolyploidy. New Phytol. 2010;186:5–17. https://doi.org/10.1111/j.1469-8137.2009.03142.x.
Pawar RP, Murkute PS, Kathar NP, Sanap GP, More AA, Tapadiya G, Hudekar G, Pimple AP. Morphological, physiochemical and antimicrobial analysis of Tinospora cordifolia extract. Acta Sci Pharmacol. 2022;3(8):47–58.
Prashant, Tomar P, Rajoriya JS, et al. Tinospora cordifolia modulates the seminal parameters, leakage of intracellular enzymes and seminal antioxidants in equilibrated and cryopreserved semen of Sahiwal bulls [published online ahead of print, 2023 Apr 3]. Reprod Domest Anim. 2023; https://doi.org/10.1111/rda.14352. doi:https://doi.org/10.1111/rda.14352
Raina SN, Ogihara Y. Chloroplast DNA diversity in Vicia faba and its close wild relatives: implications for reassessment. Theor Appl Genet. 1994;88:261–6. https://doi.org/10.1007/BF00225907.
Raina SN, Rani V, Kojima T, Ogihara Y, Singh KP, Devarumath RM. RAPD and ISSR fingerprints as useful genetic markers for analysis of genetic diversity, varietal identification, and phylogenetic relationships in peanut (Arachis hypogaea) cultivars and wild species. Genome. 2001;44:763–72. https://doi.org/10.1139/g01-064.
Ramsey J, Schemske DW. Pathways, mechanisms, and rates of polyploid formation in flowering plants. Annu Rev Ecol Syst. 1998;29:467–501. https://doi.org/10.1146/annurev.ecolsys.29.1.467.
Saghai-Maroof MA, Soliman KM, Jorgensen RA, Allard RW. Ribosomal DNA spacer-length polymorphisms in barley: Mendelian inheritance, chromosomal location, and population dynamics. The Proc Nat Acad Sci. 1984;81:8014–8. https://doi.org/10.1073/pnas.81.24.8014.
Sajith KS, Hafiz F. Pharmacological effects of Tinospora cordifolia: (Giloy) in human body. Pharma Innov J. 2022;11(7S):07–10.
Sarkar AK, Datta N, Chatterjee, U. Chromosome number reports LXVII. Taxon 1980; 29: 360–361
Selvam PK, Payyappallimana U, Ravikumar K, Venkatasubramanian P. Can Guduchi (Tinospora cordifolia), a well-known ayurvedic hepato-protectant cause liver damage? J Ayurveda Integr Med. 2023;14(1): 100658. https://doi.org/10.1016/j.jaim.2022.100658.
Sharma H, Rao PS, Singh AK. Fifty years of research on Tinospora cordifolia: From botanical plant to functional ingredient in foods. Trends in Food Science & Technology, Volume 118, Part A, Pages 189–206, 2021. https://doi.org/10.1016/j.tifs.2021.10.003.
Sharma N, Kumar A, Sharma PR, Qayum A, Singh SK, Dutt P, Paul S, Gupta V, Verma MK, Satti NK, Vishwakarma R. A new clerodane furano diterpene glycoside from Tinospora cordifolia triggers autophagy and apoptosis in HCT-116 colon cancer cells. J Ethnopharmacol. 2018;211:295–310. https://doi.org/10.1016/j.jep.2017.09.034.
Shivakumar, Vanishree H, Tegginamani, A, Zain, N. Antimicrobial efficiency of Tinospora cordifolia and Ocimum tenuiflorum against Streptococcus mutans and Candida albicans. Journal of Oral and Maxillofacial Pathology 26(4):p 470–475, Oct–Dec 2022. | DOI: https://doi.org/10.4103/jomfp.jomfp_68_22
Singh SS, Pandey SC, Srivastava S, Gupta VS, Patro B, Ghosh AC. Chemistry and medicinal properties of Tinospora cordifolia (Guduchi). Indian J Pharmacol. 2003;35:83–91.
Sinha K, Mishra NP, Singh J, Khanuja SPS. Tinospora cordifolia (Guduchi), a reservoir plant for therapeutic applications: A Review. Indian J Tradition Knowl. 2004;3:257–70.
Sybenga J. Chromosome pairing affinity and quadrivalent formation in polyploids: do segmental allopolyploids exist? Genome. 1996;39:1176–84.
Technical dossier on Guduchi (Tinospora cordifolia), All India Institute of Ayurveda, Ministry of Ayush, Govt. of India, ISBN: 978–81–958261–0–0 (https://cdn.ayush.gov.in/wp-content/uploads/2022/09/Technical-Dossier-on-GUDUCHI-Web-version.pdf).
Thakur RK, Rajpal VR, Raina SN, Kumar P, Sonkar A, Joshi L. UPLC-DAD assisted phytochemical quantitation reveals a sex, ploidy and ecogeography specificity in the expression levels of selected secondary metabolites in medicinal Tinospora cordifolia: implications for elites’ identification program. Current Topics in Medicinal Chemistry 2020 b; 20: 698–709. https://doi.org/10.2174/1568026620666200124105027
Thakur RK, Rajpal VR, Rao SR, Singh A, Joshi L, Kaushal P, Raina S.N. Induction and evaluation of colchitetraploids of two species of Tinospora Miers, 1851. Comparative Cytogenetics 2020 a; 14: 211–229. https://doi.org/10.3897/Comp. Cytogen.v14i2.33394.
Trucco F, Tatum T, Rayburn AL, Tranel PJ. Fertility, segregation at a herbicide-resistance locus, and genome structure in BC1 hybrids from two important weedy Amaranthus species. Mol Ecol. 2005;14:2717–28. https://doi.org/10.1111/j.1365-294X.2005.02581.x.
Udayan PS, George S, Tushar KV, Balachandran I. Tinospora sinensis (Lour.) Merr. from Sickupara, Kolli Hills Forest, Namakkal District, Tamil Nadu. Zoos' Print Journal 2004; 19: 1622–1623.
Udayan PS, Pradeep AK, Balachandran I. A new species of Tinospora (Menispermaceae) from South India. Edinberg J Botany. 2009;66:77–80. https://doi.org/10.1017/S0960428609005204.
Wang HC, Meng AP, Li JQ, HE, ZC. Chromosome numbers for eight species in five genera of Menispermaceae. J Japanese Botany 2004; 79: 241–246.
Wang X, Cheng ZMM, Zhi S, Xu F. Breeding triploid plants: A review. Czech J Genet Plant Breed. 2016;52:41–54. https://doi.org/10.17221/151/2015-CJGPB.
WHO supports scientifically proven traditional medicine (2020) (https://www.afro.who.int/news/who-supports-scientifically-proven-traditional-medicine).
Xu C, Zhang Y, Huang Z, Yao P, Li Y, Kang X. Impact of the leaf cut callus development stages of Populus on the tetraploid production rate by colchicine treatment. J Plant Growth Regul. 2018;37:635–44. https://doi.org/10.1007/s00344-017-9763-x.
Zhang Y, Wang B, Qi S, Dong M, Wang Z, Li Y, Chen S, Li B, Zhang J. Ploidy and hybridity effects on leaf size, cell size and related genes expression in triploids, diploids and their parents in Populus. Planta. 2019;249:635–46. https://doi.org/10.1007/s00425-018-3029-0.
Acknowledgements
The authors would like to thank National Medicinal Plants Board (NMPB), Ministry of AYUSH, Government of India for financial support. We also thank the anonymous reviewers and the subject editor for comments on the manuscript and helpful suggestions.
Funding
The research was supported by award of a grant no Z.18017/187/CSS/R&D/DL-02/2013–14-NMPB IVA to VRR and SNR by National Medicinal Plants Board (NMPB), Ministry of AYUSH, Government of India.
Author information
Authors and Affiliations
Contributions
VRR, and SNR conceptualized, wrote the grant, supervised research, guided Ph.D and prepared the first draft of the manuscript. RKT designed and conducted experiments, analyzed data, prepared tables, figures, and the 1st draft. MC performed cp RFLP analysis; PK and BKD helped in flow cytometric analysis. AS, SRR and SG contributed to editing of the MS and data analysis. VRR and SNR finalized the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
Authors declare no competing interests. All the authors have read the current manuscript and agreed to its submission in the current form.
Ethical approval
N/A.
Additional information
Corresponding Editor: Manoj Kumar Dhar; Reviewers: Veenu Kaul and Aijaz Ahmad Wani.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Thakur, R.K., Rajpal, V.R., Chaudhary, M. et al. Bringing to light the preponderance and origin of spontaneous triploid cytotypes in medicinal Tinospora cordifolia: implications for genetic improvement. Nucleus 66, 245–260 (2023). https://doi.org/10.1007/s13237-023-00437-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13237-023-00437-3