Abstract
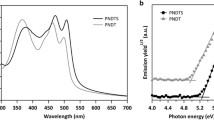
Three types of tail-to-tail (PDCBTTT), head-to-tail (PDCBTHT), and head-to-head (PDCBTHH) regioisomeric polythiophene copolymers were synthesized by modification of the substitution positions of their alkoxycarbonyl side-chains. Chain conformation, and optical, electrochemical, morphological, and charge-transport characteristics of the polymers were significantly influenced by their regiochemistry. PDCBTHH showed an amorphous morphology due to strong steric hindrance between its head-to-head alkoxycarbonyl side-chains, resulting in the poorest performance in organic solar cells and transistors among three types. PDCBTHT had a regiorandom structure with both head-to-tail and tail-to-head linkages in a polymer chain. As determined by field-effect mobility measurements, a highly regioregular and planar PDCBTTT exhibited the highest hole mobility (μ = 0.065 cm2 V-1s-1), which was two orders of magnitude higher than that of the regiorandom PDCBTHT. The highest photovoltaic performance was also measured for PDCBTTT:a fullerene acceptor (PC71BM), by maintaining the packed structures of pristine PDCBTTT polymers in the blend with PC71BM. Altering the topology of alkoxycarbonyl side-chains of the copolymers resulted in significant differences in crystalline morphologies and electrical properties and thus should be carefully considered in the molecular design of organic semiconductors.

Similar content being viewed by others
References
G. Yu, J. Gao, J. C. Hummelen, F. Wudl, and A. J. Heeger, Scienc., 270, 1789 (1995).
T. Kim, J. H. Kim, T. E. Kang, C. Lee, H. Kang, M. Shin, C. Wang, B. W. Ma, U. Jeong, T. S. Kim, and B. J. Kim, Nat. Commun., 6, 8547 (2015).
S. S. Chen, S. Jung, H. J. Cho, N. H. Kim, S. Jung, J. Q. Xu, J. Oh, Y. Cho, H. Kim, B. Lee, Y. An, C. F. Zhang, M. Xiao, H. Ki, Z. G. Zhang, J. Y. Kim, Y. F. Li, H. Park, and C. Yang, Angew. Chem. Int. Ed., 57, 13277 (2018).
F. C. Krebs, S. A. Gevorgyan, and J. Alstrup, J. Mater. Chem., 19, 5442 (2009).
T. Someya, T. Sekitani, S. Iba, Y. Kato, H. Kawaguchi, and T. Sakurai, Proc. Natl. Acad. Sci. U.S.A., 101, 9966 (2004).
X. H. Wu, Y. Ma, G. Q. Zhang, Y. L. Chu, J. Du, Y. Zhang, Z. Li, Y. R. Duan, Z. Y. Fan, and J. Huang, Adv. Funct. Mater., 25, 2138 (2015).
H. T. Yi, M. M. Payne, J. E. Anthony, and V. Podzorov, Nat. Commun., 3, 1259 (2012).
B. Nketia-Yawson and Y. Y. Noh, Macromol. Res., 25, 489 (2017).
S. Holliday, R. S. Ashraf, A. Wadsworth, D. Baran, S. A. Yousaf, C. B. Nielsen, C. H. Tan, S. D. Dimitrov, Z. R. Shang, N. Gasparini, M. Alamoudi, F. Laquai, C. J. Brabec, A. Salleo, J. R. Durrant, and I. McCulloch, Nat. Commun., 7, 11585 (2016).
Z. D. Seibers, T. P. Le, Y. Lee, E. D. Gomez, and S. M. Kilbey, ACS Appl. Mater. Interface., 10, 2752 (2018).
G. Y. Zhang, R. C. Huber, A. S. Ferreira, S. D. Boyd, C. K. Luscombe, S. H. Tolbert, and B. J. Schwartz, J. Phys. Chem., 118, 18424 (2014).
M. J. Minkler and B. S. Beckingham, Mater. Today Commun., 20, 100547 (2019).
R. Mauer, M. Kastler, and F. Laquai, Adv. Funct. Mater., 20, 2085 (2010).
X. B. Shen, V. V. Duzhko, and T. P. Russell, Adv. Energy Mater., 3, 263 (2013).
C. Poelking and D. Andrienko, Macromolecule., 46, 8941 (2013).
K. Balakrishnan, A. Datar, T. Naddo, J. L. Huang, R. Oitker, M. Yen, J. C. Zhao, and L. Zang, J. Am. Chem. Soc., 128, 7390 (2006).
C. Lee, H. Kang, W. Lee, T. Kim, K. H. Kim, H. Y. Woo, C. Wang, and B. J. Kim, Adv. Mater., 27, 2466 (2015).
T. Liu, L. J. Huo, S. Chandrabose, K. Chen, G. C. Han, F. Qi, X. Y. Meng, D. J. Xie, W. Ma, Y. P. Yi, J. M. Hodgkiss, F. Liu, J. Wang, C. L. Yang, and Y. M. Sun, Adv. Mater., 30, 1707353 (2018).
C. K. Mai, R. A. Schlitz, G. M. Su, D. Spitzer, X. J. Wang, S. L. Fronk, D. G. Cahill, M. L. Chabinyc, and G. C. Bazan, J. Am. Chem. Soc., 136, 13478 (2014).
D. Chandran and K. S. Lee, Macromol. Res., 21, 272 (2013).
S. H. Wang, D. Fazzi, Y. Puttisong, M. J. Jafari, Z. H. Chen, T. Ederth, J. W. Andreasen, W. M. M. Chen, A. Facchetti, and S. Fabiano, Chem. Mater., 31, 3395 (2019).
Y. X. Li, D. H. Lee, J. Lee, T. L. Nguyen, S. Hwang, M. J. Park, D. H. Choi, and H. Y. Woo, Adv. Funct. Mater., 27, 1701942 (2017).
H. S. Ryu, M. J. Kim, Y. W. Lee, S.-H. Lee, T. J. Shin, J. H. Cho, and H. Y. Woo, ACS Appl. Mater. Interface., 11, 47170 (2019).
M. J. Zhang, X. Guo, W. Ma, H. Ade, and J. H. Hou, Adv. Mater., 26, 5880 (2014).
E. A. Trifonova, N. M. Ankudinov, A. A. Mikhaylov, D. A. Chusov, Y. V. Nelyubina, and D. S. Perekalin, Angew. Chem. Int. Ed., 57, 7714 (2018).
J. Choi, K. H. Kim, H. Yu, C. Lee, H. Kang, I. Song, Y. Kim, J. H. Oh, and B. J. Kim, Chem. Mater., 27, 5230 (2015).
S.-S. Sun and L. R. Dalton, Introduction to Organic Electronic and Optoelectronic Materials and Devices, CRC Press, Boca Raton, 2016.
B. E. Warren, X-ray Diffraction, Courier Corporation, Mineola, 1990.
R.-J. Roe and R. Roe, Methods of X-ray and Neutron Scattering in Polymer Science, Oxford University Press, New York, 2000.
Z. G. Zhang, B. Y. Qi, Z. W. Jin, D. Chi, Z. Qi, Y. F. Li, and J. Z. Wang, Energy Environ. Sci., 7, 1966 (2014).
A. K. K. Kyaw, D. H. Wang, V. Gupta, W. L. Leong, L. Ke, G. C. Bazan, and A. J. Heeger, ACS Nan., 7, 4569 (2013).
M. Lenes, M. Morana, C. J. Brabec, and P. W. M. Blom, Adv. Funct. Mater., 19, 1106 (2009).
J. Roncali, Macromol. Rapid Commun., 28, 1761 (2007).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Acknowledgments: This work was supported by the National Research Foundation (NRF) of Korea (Grants NRF-2019R1A2C2085290, 2019R1A6A1A11044070, 2019M1A2A2065614, 2015M1A2A2057506).
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Lee, Y.W., Pak, K., Park, S.Y. et al. Regioisomeric Polythiophene Derivatives: Synthesis and Structure-Property Relationships for Organic Electronic Devices. Macromol. Res. 28, 772–781 (2020). https://doi.org/10.1007/s13233-020-8097-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-020-8097-x