Abstract
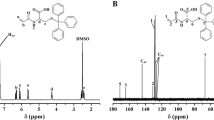
We synthesized block copolymers containing sulfamate linkage in the hydrophobic block that is cleavable to pH change. 2-(trimethylsilyloxy)ethyl methacrylate (HEMATMS) was polymerized from poly(ethylene oxide) (PEO) macroinitiator via atom transfer radical polymerization (ATRP), resulting in PEO-b-PHEMATMS with different length of PHEMATMS blocks. TMS groups were deprotected in the presence of potassium fluoride (KF), and in situ transformed to sulfamate groups by reacting with sulfamoyl chloride. The resulting block copolymer was further functionalized with trityl groups, yielding in PEO-b-PHEMA-tritylsulfamate. Molecular weight and molecular weight distribution were determined by gel permeation chromatography (GPC), and degree of polymerization (DP) of PHEMATMS blocks was measured by 1H NMR spectroscopy. These block copolymers were self-assembled as micelles in water and dissociated into unimers by lowering pH of the solutions, which was demonstrated by dynamic light scattering (DLS). We also showed that pyrene, a model hydrophobic dye, was loaded efficiently into the micelles and released upon pH-triggered disruption of micelles.

Similar content being viewed by others
References
A. Rosler, G. W. M. Vandermeulen, and H.-A. Klok, Adv. Drug Deliv. Rev., 53, 95 (2001).
H.-I. Lee, W. Wu, J. K. Oh, L. Mueller, G. Sherwood, L. Peteanu, T. Kowalewski, and K. Matyjaszewski, Angew. Chem., Int. Ed., 46, 2453 (2007).
N. Rapoport, Prog. Polym. Sci., 32, 962 (2007).
X. Hu and X. Jing, Expert Opin. Drug Deliv., 6, 1079 (2009).
F. D. Jochum and P. Theato, Chem. Commun., 46, 6717 (2010).
J.-M. Schumers, C.-A. Fustin, and J.-F. Gohy, Macromol. Rapid Commun., 31, 1588 (2010).
N. Zhang, P. R. Wardwell, and R. A. Bader, Pharmaceutics, 5, 329 (2013).
I. Roy and N. G. Munishwar, Chem. Biol., 10, 1161 (2003).
D. Fournier, R. Hoogenboom, H. M. L. Thijs, R. M. Paulus, and U. S. Schubert, Macromolecules, 40, 915 (2007).
A. K. Bajpai, S. K. Shukla, S. Bhanu, and S. Kankane, Prog. Polym. Sci., 33, 1088 (2008).
D. Roy, J. N. Cambre, and B. S. Sumerlin, Prog. Polym. Sci., 35, 278 (2010).
Y. Liu, W. Wang, J. Yang, C. Zhou, and J. Sun, Asian J. Pharm. Sci., 8, 159 (2013).
I.-K. Park, K. Singha, R. B. Arote, Y.-J. Choi, W. J. Kim, and C.-S. Cho, Macromol. Rapid Commun., 31, 1122 (2010).
R. Tang, W. Ji, and C. Wang, Macromol. Chem. Phys., 212, 1185 (2011).
H.-I. Lee, J. R. Boyce, A. Nese, S. S. Sheiko, and K. Matyjaszewski, Polymer, 49, 5490 (2008).
S.-H. Jung, H.-Y. Song, Y. Lee, H. M. Jeong, and H.-I. Lee, Macromolecules, 44, 1628 (2011).
S. Dai, P. Ravi, and K. C. Tam, Soft Matter, 4, 435 (2008).
J.-S. Wang and K. Matyjaszewski, J. Am. Chem. Soc., 117, 5614 (1995).
K. Matyjaszewski and J. Xia, Chem. Rev., 101, 2921 (2001).
H.-I. Lee, J. Pietrasik, and K. Matyjaszewski, Macromolecules, 39, 3914 (2006).
D. S. Fischer, S. K. Chander, L. W. L. Woo, J. C. Fenton, A. Purohit, M. J. Reed, and B. V. L. Potter, J. Steroid Biochem., 84, 343 (2003).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, H., Lee, Hi. pH-responsive polymeric micelles from sulfamate-conjugated block copolymers. Macromol. Res. 23, 129–133 (2015). https://doi.org/10.1007/s13233-015-3013-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-015-3013-5