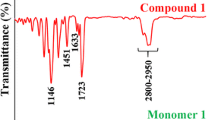
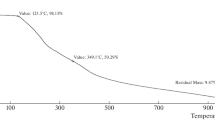
Abstract
A new strategy for the synthesis of dimethacrylate-based macromonomers on isosorbide via a ring opening reaction is suggested. For this purpose, ethylene glycol derivatives (mono, di, and tri) were firstly monobrominated, and their hydroxyl groups were protected as tetrahydropyranyl (THP) ethers. Then, the reaction of isosorbide with obtained alkylation reagents were carried out in the presence of NaH/N,N-dimethylformamide (DMF) to provide alkylated isosorbide derivatives. Deprotection of tetrahydropyranyl groups was accomplished under acidic conditions at room temperature and series of new diols from isosorbide were thus prepared in high yields. Eventually, the reaction of new diols with glycidyl methacrylate, which afforded new dimethacrylates containing an isosorbide skeleton, was linked to mono, di, and tri-ethylene glycol moieties. The chemical structures of all samples as representatives were characterized by Fourier transform infrared (FTIR), 13C and 1H nuclear magnetic resonance (NMR) spectroscopy.

Similar content being viewed by others
References
H. R. Kricheldorf, Rev. Macromol. Chem. Phys., 7, 599 (1997).
D. Abenhaim, A. Loupy, L. Munnier, R. Tamion, F. Marsais, and G. Queguiner, Carbohydr. Res., 261, 255 (1994).
D. J. Claffey, M. F. Casey, and P. A. Finan, Carbohydr. Res., 339, 2433 (2004).
P. Stoss and R. Hemmer, Adv. Carbohydr. Chem. Biochem., 49, 92 (1991).
M. Rose and R. Palkovits, ChemSusChem, 5, 167 (2012).
Q. Lin, J. Pasatta, and T. E. Long, J. Polym. Sci. Part A: Polym. Chem., 41, 2512 (2003).
B. Zhang, Y. Zheng, and H. Lu, Liq. Cryst., 32, 357 (2005).
N. N. Chavan, Mater. Sci. Appl., 2, 1520 (2011).
O. N. Van-Buu, A. Aupoix, and G. Vo-Thanh, Tetrahedron, 65, 2260 (2009).
C. Carcedo, A. Dervisi, A. I. Fallis, L. Ooi, and A. K. M. Malik, Chem. Commun., 10, 1236 (2004).
S. Kumar and U. Ramachandran, Tetrahedron, 61, 4141 (2005).
S. Chatti, M. Bortolussi, and A. Loupy, Tetrahedron Lett., 41, 3367 (2000).
J. Wang, C. Zhang, Z. Qu, Y. Hou, B. Chen, and P. Wu, J. Chem. Res., 1, 294 (1999).
S. Naik, R. Gopinath, and B. K. Patel, Tetrahedron Lett., 42, 7679 (2001).
Y. J. Jung, E. S. Park, J. H. Lee, and C. M. Yoon, Bull. Korean Chem. Soc., 23, 791 (2002).
F. Badalassi, G. Klein, P. Crotti, and J. L. Reymond, Eur. J. Org. Chem., 12, 2557 (2004).
P. S. Méndez, R. E. Cachau, G. Seoane, and O. N. Ventura, THEOCHEM. 904, 21 (2009).
F. Osterkamp, H. Wehlan, U. Koert, M. Wiesner, P. Raddatz, and S. L. Goodman, Tetrahedron, 55, 10713 (1999).
S. E. Denmark, P. A. Barsanti, G. L. Beutner, and T. W. Wilson, Adv. Synth. Catal., 349, 567 (2007).
P. Chiniwalla, Y. Bai, E. Elce, R. Shick, W. C. Mc-Dougal, S. A. B. Allen, and P. A. Kohl, J. Appl. Polym. Sci., 89, 568 (2003).
M. Ghaemy, M. Heidaripour, and M. Barghamadi, J. Appl. Polym. Sci., 106, 1917 (2007).
M. S. Sheela, K. T. Selvy, V. K. Krishnan, and S. N. Pal, J. Appl. Polym. Sci., 42, 561 (1991).
G. Sahin, A. Z. Albayrak, Z. Sarayli, and D. Avci, J. Polym. Sci. Part A: Polym. Chem., 44, 6775 (2006).
C. Domingo, R. W. Arcs, A. López-Macipe, R. Osorio, R. Rodr guez-Clemente, J. Murtra, M. A. Fanovich, and M. Toledano, J. Biomed. Mater. Res., 56, 297 (2001).
N. A. Chowdhury, K. Wakasa, R. Priyawan, and M. Yamaki, J. Mater. Sci. Mater. Med., 6, 400 (1995).
K. V. Kalliyana, P. P. Lizymol, and P. N. Sindhu, J. Appl. Polym. Sci., 74, 735 (1999).
A. M. Atta, R. A. El-Ghazawy, R. K. Farag, and A. A. Abdel-Azim, React. Funct. Polym., 66, 931 (2006).
A. M. Atta, R. A. El-Ghazawy, R. K. Farag, and A. A. Abdel-Azim, J. Polym. Res., 13, 257 (2006).
M. H. Zhou and W. Cho, Polym. Int., 50, 1193 (2001).
A. M. Atta and K. F. Arndt, Polym. Int., 50, 1360 (2001).
J. E. Mark and B. Erman, in Performance of Plastics, W. Brostow, Ed., Hanser, Munich-Cincinnati, 2000, Chap. 17.
Z. Czech, Polym. Int., 52, 347 (2003).
M. Jaymand, Polym. J., 43, 901 (2011).
D. Bogdal, J. Pielichowski, and A. Boron, J. Appl. Polym. Sci., 66, 2333 (1997).
R. Kluger and A. Alagic, Bioorg. Chem., 32, 451 (2004).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Vazifehasl, Z., Hemmati, S., Zamanloo, M. et al. Synthesis and characterization of novel diglycidyl methacrylate-based macromonomers on isosorbide for dental composites. Macromol. Res. 21, 427–434 (2013). https://doi.org/10.1007/s13233-013-1038-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-013-1038-1