Abstract
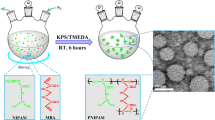
A unique and rapid method was used to synthesize thermosensitive self-crosslinked nano (N-isopropylacrylamidevinylpyrrolidone-acrylamide) (NIPAAm-VP-AAm) terpolymers. Nanohydrogels were obtained within 30 min during a polymerization reaction with a convenient yield due to the high dilution of aqueous solution at an elevated temperature. Furthermore, hydrogen peroxide (H2O2) was employed for the first time as a safe initiator without any toxic segments for the thermosensitive polymer synthesis. Different nanohydrogels were obtained by modulating the molar ratio of monomers to initiators and their influences on the composition ratios, thermoresponsive behavior, size distribution, phase separation, and nanohydrogel drug release were investigated. As the synthetic route is crosslinker free, the obtained nanohydrogels can be introduced as a non-toxic efficient drug delivery system (DDS). The possibility of free radical formation on the PNIPAAm chains, which leads to the crosslinked structure, has been investigated theoretically by quantum mechanical calculations. The potential energy surface of the reaction was examined by changing the distance between the OH radical and the H atoms. Considering these calculations, the reaction can proceed from all pathways as it is observed experimentally. The average size of nanohydrogels, as revealed by dynamic light scattering (DLS) and transmission electronic microscopy (TEM), is <50 nm. Finally, naltrexone as an opiate antagonist was selected as a model drug for the investigation of nanohydrogel drug delivery capabilities. Nanohydrogels show sustained naltrexone release beyond 3 months without any initial burst release.

Similar content being viewed by others
References
J. Zhang, H. Chen, L. Xu, and Y. Gu, J. Control. Release, 34, 131 (2008).
Y. Lu and M. Ballauff, Prog. Polym. Sci., 36, 767 (2011).
A. Sosnik, A. M. Carcaboso, and D. A. Chiappetta, Recent Pat. Biomed. Eng., 1, 43 (2008).
O. C. Farokhzad and R. Langer, ACS Nano, 3, 16 (2009).
V. P. Torchilin, Adv. Drug Deliv. Rev., 58, 1532 (2006).
V. Mittal, N. B. Matsko, A. Butte, and M. Morbidelli, Eur. Polym. J., 43, 4868 (2007).
F. J. Xu, J. Li, S. J. Yuan, Z. X. Zhang, E. T. Kang, and K. G. Neoh, Biomacromolecules, 9, 331 (2008).
C. Choi, S. Y. Chae, and J. W. Nah, Polymer, 47, 4571 (2006).
G. Fundueanu, M. Constantin, and P. Ascenzi, Biomaterials, 29, 2767 (2008).
H. L. Wei, K. Yao, Z. Yang, H. J. Chu, J. Zhu, C. C. Ma, and Z. X. Zhao, Macromol. Res., 19, 294 (2011)
B. Massoumi and A. Entezami, Eur. Polym. J., 37, 1015 (2001).
M. K. Jaiswal, R. Banerjee, P. Pradhan, and D. Bahadur, Colloids Surf. B: Biointerface, 81, 185 (2010).
L. Starovoytovaa, J. Spevaceka, and M. Lavsky, Polymer, 46, 677 (2005).
A. K. Bajpai, S. K. Shukla, S. Bhanu, and S. Kankane, Prog. Polym. Sci., 33, 1088 (2008).
X. Lia, W. Liua, G. Yea, B. Zhanga, D. Zhua, K. Yaoa, Z. Liub, and X. Sheng, Biomaterials, 26, 7002 (2005).
X. Z. Zhang, D. Q. Wu, and C. C. Chu, Biomaterials, 25, 3793 (2004).
L. M. Geever, M. J. D. Nugent, J. E. Kennedy, J. G. Lyons, and C. L. Higginbotham, Eur. Polym. J., 42, 69 (2006).
F. Eeckman, A. J. Moes, and K. Amighi, Eur. Polym. J., 40, 873 (2004).
A. Gutowska, Y. H. Bae, J. Feijen, and S. W. Kim, J. Control. Release, 22, 95 (1992).
J. Spevacek, Curr. Opin. Colloid Interface Sci., 14, 184 (2009).
H. Kubot and A. Fukuda, J. Appl. Polym. Sci., 65, 1313 (1998).
S. Nayak and L. A. Lyon, Angew. Chem. Int. Ed., 44, 7686 (2005).
J. Gao and B. J. Frisken, Langmuir, 19, 5217 (2003).
W. E. Hennink and C. F. Nostrum, Adv. Drug Deliv. Rev., 54, 13 (2002).
X. J. Loh, P. Peh, S. Liao, and C. Sng, and J. Li, J. Control. Release, 143, 175 (2010).
D. N. Robinson and N. A. Peppas, Macromolecules, 35, 3668 (2002).
J. Gao and B. J. Frisken, Langmuir, 19, 5212 (2003).
R. Salehi, N. Arsalani, S. Davaran, and A. A. Entezami, J. Biomed. Mater. Res. A, 89A, 919 (2009).
R. Salehi, S. Davaran, M. R. Rashidi, and A. A. Entezami, J. Appl. Polym. Sci., 111, 1905 (2009).
R. Salehi, K. Nowruzi, A. A. Entezami, V. Asgharzadeh, and S. Davaran, Polym. Adv. Technol., 4, 416 (2009).
X. Qiao and Z. Zhang, J. Photochem. Photobiol. A, 190, 15 (2007).
M. J. Frisch, Gaussian 03 (Revision B.03), Gaussian. Inc, Pittsburgh, 2003.
L. M. Geever, D. M. Devine, M. J. D. Nugent, J. E. Kennedy, J. G. Lyons, A. Hanley, and C. L. Higginbotham, Eur. Polym. J., 42, 2540 (2006).
A. Larsson, D. Kuckling, and M. Schonhoff, Colloids Surf. A, 190, 185 (2001).
M. Andersson and S. L. Maunu, Colloid Polym. Sci., 285, 293 (2006).
H. Kourilov, L. Hanykov, and J. Spevcek, Eur. Polym. J., 45, 2935 (2009).
X. Huang and C. S. Brazel, J. Control. Release, 73, 121 (2001).
C. Choi, S. Y. Chae, and J. W. Nah, Polymer, 47, 4571 (2006).
W. F. Lee and R. J. Chiu, J. Mater. Sci. Eng. C, 20, 161 (2002).
C. H. Alarcon, S. Pennadam, and C. Alexander, Chem. Soc. Rev., 34, 276 (2005).
O. Mashinchian, R. Salehi, G. Dehghan, A. Aganejad, S. Davaran, and Y. Omidi, Int. J. Drug Deliv., 2, 278 (2010).
L. E. Bromberg and E. S. Ron, Adv. Drug Deliv. Rev., 31, 197 (1998).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fathi, M., Entezami, A.A., Ebrahimi, A. et al. Synthesis of thermosensitive nanohydrogels by crosslinker free method based on N-isopropylacrylamide: Applicable in the naltrexone sustained release. Macromol. Res. 21, 17–26 (2013). https://doi.org/10.1007/s13233-012-0181-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-012-0181-4