Abstract
Background
Genetic and epigenetic factors play significant roles in the aetio-pathogenesis of pre-eclampsia (PE). The effects may vary across racial and geographical boundaries. The role of epigenetic modification in pre-eclampsia was studied among African populations in Lagos, Nigeria.
Aim and Objectives
This study aimed to determine the pattern of Methylene tetrahydrofolate reductase gene (MTHFR) CpG island methylation in pre-eclampsia, and evaluate associated covariates.
Methodology
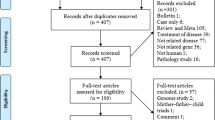
This study was an observational, cross-sectional, study conducted at the Lagos University Teaching Hospital and the Lagos State Island Maternity Hospital. A total of 400 pregnant women consisting of 200 pregnant women diagnosed with pre-eclampsia (study group) and 200 pregnant normotensive and apparently healthy women (control group) were recruited for the study. Demographic and clinical histories were obtained through questionnaires. The DNA Methylation status of the CpG Island in promoter region of the MTHFR gene was assessed using bisulphite conversion and methylation specific PCR method. The biochemical parameters measured in the study were: red cell folate, vitamin B12, plasma homocysteine (Hcy) and methylene tetrahydrofolate reductase enzyme level.
Results
Homozygous MTHFR CpG island hypomethylation pattern was significantly associated with pre-eclampsia (χ2 = 22.96; p = 0.000), Mean values of plasma homocysteine in PE women with homozygous hypomethylation (26.1 ± 9.1 umol/L) were significantly higher than (20.1 ± 4.2 umol/L) observed in PE subjects with homozygous hypermethylation (p = 0.008). Homozygous CpG island hypomethylated pattern of the MTHFR promoter region, was associated with the lowest median MTHFR enzyme level (72.8 ± 39.8 pmol/L) compared with heterozygous methylated pattern (91.3 ± 60.9 pmol/L; p = 0.047) and homozygous methylated pattern (82.3 ± 31.0 pmol/L; 0.047). Red cell folate and Vitamin B12 levels were not significantly associated with CpG island methylation status.
Conclusion
Epigenetic modification plays significant role in the pathogenesis of pre-eclampsia.
Similar content being viewed by others
References
Tranquilli AL, Dekker G, Magee L. The classification, diagnosis and management of the hypertensive disorders of pregnancy: a revised statement from the ISSHP. Pregnancy Hypertens. 2014;4(2):97–104.
Gandhi S, Rao V. Socio-demographic and other risk factors of pre-eclampsia at a tertiary care hospital, Karnataka: case control study. J Clin Diagn Res. 2014;8(9):JC01–4. https://doi.org/10.7860/jcdr/2014/10255.4802.
Soomro S, Kumar R, Lakhan H, et al. Risk factors for pre-eclampsia and eclampsia disorders in tertiary care center in Sukkur, Pakistan. Cureus. 2019;11(11):e6115. https://doi.org/10.7759/cureus.6115.
Das S, Das R, Bajracharya R, et al. Incidence and risk factors of pre-eclampsia in the paropakar maternity and women’s hospital, Nepal: a retrospective study. Int J Environ Res Public Health. 2019;16(19):3571. https://doi.org/10.3390/ijerph16193571.
Kongwattanakul K, Saksiriwuttho P, Chaiyarach S, et al. Incidence, characteristics, maternal complications, and perinatal outcomes associated with preeclampsia with severe features and HELLP syndrome. Int J Womens Health. 2018;10:371–7.
Oladapo OT, Adetoro OO, Ekele BA, et al. When getting there is not enough: a nationwide cross-sectional study of 998 maternal deaths and 1451 near-misses in public tertiary hospitals in a low-income country. BJOG. 2015;123(6):928–38.
Pisal H, Dangat K, Randhir K, et al. Higher maternal plasma folate, vitamin B12 and homocysteine levels in women with preeclampsia. J Hum Hypertens. 2019;33:393–9.
Ge J, Wang F, Zhang B, et al. Correlation between MTHFR gene methylation and pre-eclampsia, and its clinical significance. Genet Mol Res. 2015;14(3):8021–8.
Wen SW, Guo Y, Rodger M, et al. Folic acid supplementation in pregnancy and the risk of pre-eclampsia-a cohort study. PLoS ONE. 2016;11(2):e0149818.
Harika R, Faber M, Samuel F, et al. Micronutrient status and dietary intake of iron, vitamin a, iodine, folate and zinc in women of reproductive age and pregnant women in Ethiopia, Kenya, Nigeria and South Africa: a systematic review of data from 2005 to 2015. Nutrients. 2017;9(10):1096–110.
Mousa AA, Archer K, Cappello R, et al. DNA methylation is altered in maternal blood vessels of women with preeclampsia. Reproduct Sci. 2012;19(12):1332–42.
Kristen RY, Christine LC, Ruth P, et al. DNA methylation profiles in pre-eclampsia and healthy control placentas. Am J Physiol- Heart Circ Physiol. 2016;310(10):H1295–303.
Li WX, Dai SX, Zheng JJ, et al. Homocysteine metabolism gene polymorphisms (MTHFR C677T, MTHFR A1298C, MTR A2756G and MTRR A66G) jointly elevate the risk of folate deficiency. Nutrients. 2015;7(8):6670–87.
Li Z, Ye R, Zhang L, et al. Folic acid supplementation during early pregnancy and the risk of gestational hypertension and preeclampsia. Hypertension. 2013;61(4):873–9.
Fernàndez-Roig S, Lai SC, Murphy MM, et al. Vitamin B12 deficiency in the brain leads to DNA hypomethylation in the TCblR/CD320 knockout mouse. Nutr Metab (Lond). 2012;18(9):41–52.
Bomba-Opoń D, Hirnle M, Kalinka J, et al. Folate supplementation during the preconception period, pregnancy and puerperium. Pol Soc Gynecol Obstet Guidel Ginekol Polska. 2017;88(11):633–6.
Author information
Authors and Affiliations
Contributions
Osunkalu Vincent: Conceptualization, data analysis and manuscript writing. Idowu Taiwo: Conceptualization, Genetic analysis and study design, literature review. Makwe Christain: Article review, literature review, project supervision. Abiola Ann Ogbenna: Article review, literature review, project supervision Quao Rachel: statistical analysis and editing. Anorlu Rose: Article review, literature review, project supervision.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare no conflict of interest whatsoever.
Human and Animal Rights
Research involving Human participants only. The study has been performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helenski and its amendments or comparable ethical standards.
Informed Consent
Study was approved by the Human Research and Ethical Committee of the Lagos University Teaching Hospital, Nigeria. Written and oral informed consent were obtained from all participants before recruitment into the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Osunkalu, Vincent Oluseye is an Associate Professor of Haematology and Blood Transfusion at the College of Medicine of the University of Lagos, Lagos, Nigeria. Idowu Taiwo, is an Associate Professor of Cell Biology and Genetics from the University of Lagos, Lagos, Nigeria. Makwe Christian is a Senior Lecturer in the Department of Obstetrics and Gynaecology at the University of Lagos, Lagos, Nigeria. Anorlu Rose is a Professor from the Department of Obstetrics and Gynaecology at the College of Medicine, University of Lagos, Lagos, Nigeria. Quao Rachel Abiana is a Graduate from the Physiology Department at the College of Medicine, University of Lagos, Lagos, Nigeria.
Rights and permissions
About this article
Cite this article
Osunkalu, V.O., Taiwo, I.A., Makwe, C.C. et al. Epigenetic Modification in Methylene Tetrahydrofolate Reductase (MTHFR) Gene of Women with Pre-eclampsia. J Obstet Gynecol India 71, 52–57 (2021). https://doi.org/10.1007/s13224-020-01374-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13224-020-01374-w