Abstract
Purpose
To evaluate the role of single-step universal screening in first trimester and its effectiveness.
Materials and methods
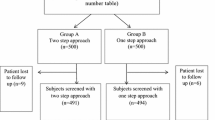
Three thousand women attending antenatal clinic of Pt JNM Medical College, Raipur, were screened with 75 gm OGTT in their first trimester irrespective of their last meal, and those who were screened negative were again subjected to OGTT at 24–28 weeks. The women were followed throughout pregnancy till delivery. Any maternal or perinatal complications were noted.
Result
Overall incidence of GDM was 5.2 %. About 61.54 % women screened positive in first trimester. At 24–28 weeks, 38.46 % women were diagnosed with GDM. Women diagnosed in first trimester showed significantly low incidence of cesarean section rate (20.83 %), PIH (2.08 %), and macrosomia (14.44 %), in comparison to women diagnosed later in pregnancy.
Conclusion
The role of first trimester screening was found effective in reducing adverse maternal and perinatal outcomes in GDM.


Similar content being viewed by others
References
International Diabetes Federation. Policy briefing. Diabetes in pregnancy: protecting maternal health. Brussels: IDF.; 2011.
Kayal A, Anjana RM, Mohan V. Gestational diabetes—an update from India. Diabetes Voice. 2013;58(2):30–4.
Wahi P, Dogra V, Jandial K. Prevalence of gestational diabetes mellitus GDM and its outcomes in Jammu Region. JAPI. 2011;59:227–30.
Vambergue A, Fajarde I. Consequences of gestational and pre-gestational diabetes on placental function and birth weight. World J Diabetes. 2011;2(11):196–203.
Rajput R, Yadav Y, Nanda S, et al. Prevalence of gestational diabetes mellitus & associated risk factors at a tertiary care hospital in Haryana. Indian J Med Res. 2013;137:728–33.
Anjana RM, Pradeepa R, Deepa M, et al. ICMR-INDIAB Collaborative study group, Prevalence of diabetes and prediabetes (Impaired fasting glucose and/or impaired glucose tolerance) in urban and rural India phase 1 result of Indian council of Medical Research-INdiaDIABates (ICMR-INDIAB) study. Diabetologia. 2011;54:3022–7.
Plasencia W, Garcia R, Pereira S, et al. Criteria for screening and diagnosis of gestational diabetes mellitus in the first trimester of pregnancy. Fetal Diagn Ther. 2011;30:108–15.
Seshiah V, Banerjee S, Balaji V, et al. Consensus evidence-based guidelines for management of gestational diabetes mellitus in India. Suppl JAPI. 2014;62:55–62.
International Association of Diabetes and Pregnancy Study Groups. Recommendations on the diagnosis and classification of Hyperglycemia in pregnancy. Diabetes Care. 2010;33:3.
WHO. Guidelines diagnostic criteria and classification of hyperglycemia first detected in pregnancy. 2013.
NICE. Guidelines diabetes in pregnancy: management of diabetes and its complications from preconception to postnatal period nic.org.uk/guidence/ng3, 25 Feb 2015.
Huynh J, Ratnaik S, Bartalotta C. Challenging the glucose challenge test. Aust N Z J Obstet Gynarcol. 2011;51:22–5.
Savvidou M, Nelson MS, Makgoba M, et al. First—trimester prediction of diabetes mellitus: examining the potential of combining maternal characteristics and laboratory measures. Diabetes. 2010;59(12):3017–22.
Black MH, Sacks DA, Xiang AH, et al. Clinical outcome of pregnancies complicated by mild gestational diabetes differ by combinations of abnormal oral glucose tolerance test values. Diabetes Care. 2010;33(12):2524–30.
Landon MB, Mele L, Spong CY, et al. The relationship between maternal glycemia and perinatal outcome. Am Coll Obstet Gynecol. 2011;117:218–24.
O’Sullivan EP, Avalos G, O’Reilly M, et al. Atlantic diabetes in pregnancy (DIP); the prevalence and outcomes of gestational diabetes mellitus using new diagnostic criteria. Diabetologia. 2011;54(7):1670–5.
Falavigna M, Prestes I, Schmidt MI, et al. Impact of gestational diabetes mellitus screening strategies on perinatal outcomes: a simulation study. Diabetes Res Clin Pract. 2013;99(3):358–65.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Abha Singh and Avinashi declare they have no conflict of interest.
Ethical Approval
As this was prospective observational study, not an interventional or case–control study, informed consent with human subjects for being included in the study was not necessary; however, all procedures followed were in accordance with ethical standard of the responsible committee on human experiments (institutional and international) and with the Helsinki Declaration of 1975, as revised in 2008(5).
Additional information
Dr. Abha Singh is a Director, Professor, & HOD in the Department of Obstetrics & Gynaecology at Pt. J.N.M. Medical College. Dr. Avinashi Kujur is an Assistant Professor in the Department of Obstetrics & Gynaecology at Pt. J.N.M. Medical College.
Rights and permissions
About this article
Cite this article
Singh, A., Kujur, A. Single-Step First Trimester Screening “Sooner the Better”. J Obstet Gynecol India 66 (Suppl 1), 77–81 (2016). https://doi.org/10.1007/s13224-015-0785-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13224-015-0785-7