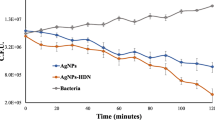
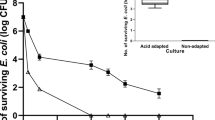
Abstract
Houttuynia Cordata Thunb (HCT; Saururaceae), known as ‘Eosungcho’ in Korea, has been traditionally used for antibiotic treatment and has proved its effect through clinical demonstrations. In spite of it s being used quite frequently in traditional prescriptions because of its superior antibiotic effect, HCT’s molecular mechanisms on its antibiotic effect have not been understood. Hence, this is the first study about HCT’s antibiotic mechanisms at the molecular level. HCT was extracted to acquire the fractions, then inoculated to confirm its antibiotic effect toward the bacteria Escherichia coli O157:H7 (E. coli O157:H7; ATCC 43894) through the disc diffusion method. The antibacterial efficacy of HCT was measured by diameter of cleared zone. Thereafter, we isolated the RNA of affected cells and analyzed it’s expressions through the microarray. To confirm the accuracy of the acquired data, RT-PCR was carried out. Results from this study indicate that E. coli O157:H7 exposures to HCT fractions, a number of genes that have been related to the synthesis of bacterial cell wall are downregulated, while some of the cell wall synthesis inhibitory genes are upregulated. Both results indicate that HCT fractions inhibit cell wall synthesis as like as β-lactam antibiotics. Furthermore, a few genes that have a critical role to DNA replication are downregulated. These genes, including folA, are closely related to the folate biosynthesis. Remarkable abnormal regulations that show antibiotic resistance activity of the E. coli O157: H7 are found, and are seem to be closely linked to the multidrug efflux pumps, especially upregulation of marRAB operon. The results of this study improve our understanding of the mode of action of HCT on E. coli O157:H7 and may show the usefulness of HCT fractions in the antibacterial treatment.
Similar content being viewed by others
References
Ahn, B. Literature review on the pharmaceutical effects and utilization of Houttyunia cordata Thunb. J. East Asian Soc. Dietary Life 12, 1–6 (2002).
Lee, J.S. et al. Suppressive effects of Houttuynia cordata Thunb (Saururaceae) extract on Th2 immune response. J. Ethnopharmacol. 117, 34–40 (2008).
Lau, K.M. et al. Immunomodulatory and anti-SARS activities of Houttuynia cordata. J. Ethnopharmacol. 118, 79–85 (2008).
Sherman, P.M., Ossa, J.C. & Wine, E. Bacterial infections: new and emerging enteric pathogens. Curr. Opin. Gastroenterol. 26, 1–4 (2010).
La Ragione, R.M., Best, A., Woodward, M.J. & Wales, A.D. Escherichia coli O157:H7 colonization in small domestic ruminants. FEMS Microbiol. Rev. 33, 394–410 (2009).
Chase-Topping, M., Gally, D., Low, C., Matthews, L. & Woolhouse, M. Super-shedding and the link between human infection and livestock carriage of Escherichia coli O157. Nat. Rev. Microbiol. 6, 904–912 (2008).
Tarr, P.I., Gordon, C.A. & Chandler, W.L. Shiga-toxinproducing Escherichia coli and haemolytic uraemic syndrome. Lancet 365, 1073–1086 (2005).
Holtz, L.R., Neill, M.A. & Tarr, P.I. Acute bloody diarrhea: a medical emergency for patients of all ages. Gastroenterology 136, 1887–1898 (2009).
Jenke, C. et al. Phylogenetic analysis of enterohemorrhagic Escherichia coli O157, Germany, 1987–2008. Emerg. Infect. Dis. 16, 610–616 (2010).
U.S.FDA. Immediate Release. P06-197 (12 Dec 2006).
Walsh, C. Molecular mechanisms that confer antibacterial drug resistance. Nature 406, 775–781 (2000).
Blumberg, P.M. & Strominger, J.L. Interaction of penicillin with the bacterial cell: penicillin-binding proteins and penicillin-sensitive enzymes. Bacteriol. Rev. 38, 291–335 (1974).
Lovering, A.L., Gretes, M. & Strynadka, N.C. Structural details of the glycosyltransferase step of peptidoglycan assembly. Curr. Opin. Struct. Biol. 18, 534–543 (2008).
Onoe, H., Matsumoto, A., Hashimoto, K., Yamano, Y. & Morishima, I. Peptidoglycan recognition protein (PGRP) from eri-silkworm, Samia cynthia ricini; protein purification and induction of the gene expression. Comp. Biochem. Physiol. B. Biochem. Mol. Biol. 147, 512–519 (2007).
Stenbak, C.R. et al. Peptidoglycan molecular requirements allowing detection by the Drosophila immune deficiency pathway. J. Immunol. 173, 7339–7348 (2004).
Mor, A., Delfour, A. & Nicolas, P. Identification of a D-alanine-containing polypeptide precursor for the peptide opioid, dermorphin. J. Biol. Chem. 266, 6264–6270 (1991).
Barrett, D. et al. Analysis of glycan polymers produced by peptidoglycan glycosyltransferases. J. Biol. Chem. 282, 31964–31971 (2007).
Wiedemann, B., Pfeifle, D., Wiegand, I. & Janas, E. beta-Lactamase induction and cell wall recycling in gram-negative bacteria. Drug Resist Updat. 1, 223–226 (1998).
Hirano, T., Minamino, T. & Macnab, R.M. The role in flagellar rod assembly of the N-terminal domain of Salmonella FlgJ, a flagellum-specific muramidase. J. Mol. Biol. 312, 359–369 (2001).
Allen, M.J., White, G.F. & Morby, A.P. The response of Escherichia coli to exposure to the biocide polyhexamethylene biguanide. Microbiology 152, 989–1000 (2006).
Feher, K., Pristovsek, P., Szilagyi, L., Ljevakovic, D. & Tomasic, J. Modified glycopeptides related to cell wall peptidoglycan: conformational studies by NMR and molecular modelling. Bioorg. Med. Chem. 11, 3133–3140 (2003).
Reynolds, P.E., Structure, biochemistry and mechanism of action of glycopeptide antibiotics. Eur. J. Clin. Microbiol. Infect. Dis. 8, 943–950 (1989).
Korat, B., Mottl, H. & Keck, W. Penicillin-binding protein 4 of Escherichia coli: molecular cloning of the dacB gene, controlled overexpression, and alterations in murein composition. Mol. Microbiol. 5, 675–684 (1991).
Kishida, H. et al. Crystal structure of penicillin binding protein 4 (dacB) from Escherichia coli, both in the native form and covalently linked to various antibiotics. Biochemistry 45, 783–792 (2006).
Lessard, I.A. & Walsh, C.T. VanX, a bacterial Dalanyl-D-alanine dipeptidase: resistance, immunity, or survival function? Proc. Natl. Acad. Sci. USA 96, 11028–11032 (1999).
Scholte, A.A., Eubanks, L.M., Poulter, C.D. & Vederas, J.C. Synthesis and biological activity of isopentenyl diphosphate analogues. Bioorg. Med. Chem. 12, 763–770 (2004).
Shaw, P.J. & Hills, G.J. The three-dimensional structure of the cell wall glycoprotein of Chlorogonium elongatum. J. Cell Sci. 68, 271–284 (1984).
Besong, G.E. et al. A de novo designed inhibitor of D-Ala-D-Ala ligase from E. coli. Angew. Chem. Int. Ed. Engl. 44, 6403–6406 (2005).
Frick, D.N., Townsend, B.D. & Bessman, M.J. A novel GDP-mannose mannosyl hydrolase shares homology with the MutT family of enzymes. J. Biol. Chem. 270, 24086–24091 (1995).
Fohrer, J., Hennig, M. & Carlomagno, T. Influence of the 2′-hydroxyl group conformation on the stability of A-form helices in RNA. J. Mol. Biol. 356, 280–287 (2006).
Sousa, M.M., Krokan, H.E. & Slupphaug, G. DNA-uracil and human pathology. Mol. Aspects. Med. 28, 276–306 (2007).
Pytel, D., Slupianek, A., Ksiazek, D., Skorski, T. & Blasiak, J. [Uracil-DNA glycosylases]. Postepy. Biochem. 54, 362–370 (2008).
Fenech, M. Folate, DNA damage and the aging brain. Mech. Ageing. Dev. (2010).
Bailey, S.W. & Ayling, J.E. The extremely slow and variable activity of dihydrofolate reductase in human liver and its implications for high folic acid intake. Proc. Natl. Acad. Sci. USA 106, 15424–15429 (2009).
Wegkamp, A., van Oorschot, W., de Vos, W.M. & Smid, E.J. Characterization of the role of para-aminobenzoic acid biosynthesis in folate production by Lactococcus lactis. Appl. Environ. Microbiol. 73, 2673–2681 (2007).
Pages, J.M., Masi, M. & Barbe, J. Inhibitors of efflux pumps in Gram-negative bacteria. Trends. Mol. Med. 11, 382–389 (2005).
Bavro, V.N. et al. Assembly and channel opening in a bacterial drug efflux machine. Mol. Cell 30, 114–121 (2008).
Morita, Y., Sobel, M.L. & Poole, K. Antibiotic inducibility of the MexXY multidrug efflux system of Pseudomonas aeruginosa: involvement of the antibioticinducible PA5471 gene product. J. Bacteriol. 188, 1847–1855 (2006).
Lewis, K., Hooper, D.C. & Ouellette, M. Multidrug resistance pumps provide broad defense — MDR pumps expel a broad array of otherwise toxic molecules, including many antibiotics. ASM News 63, 605–610 (1997).
Asako, H., Nakajima, H., Kobayashi, K., Kobayashi, M. & Aono, R. Organic solvent tolerance and antibiotic resistance increased by overexpression of marA in Escherichia coli. Appl. Environ. Microbiol. 63, 1428–1433 (1997).
Fralick, J.A. Evidence that TolC is required for functioning of the Mar/AcrAB efflux pump of Escherichia coli. J. Bacteriol. 178, 5803–5805 (1996).
Martin, R.G. & Rosner, J.L. Binding of purified multiple antibiotic-resistance repressor protein (MarR) to mar operator sequences. Proc. Natl. Acad. Sci. USA 92, 5456–5460 (1995).
Martin, R.G., Jair, K.W., Wolf, R.E., Jr. & Rosner, J.L. Autoactivation of the marRAB multiple antibiotic resistance operon by the MarA transcriptional activator in Escherichia coli. J. Bacteriol. 178, 2216–2223 (1996).
McMurry, L.M., George, A.M. & Levy, S.B. Active efflux of chloramphenicol in susceptible Escherichia coli strains and in multiple-antibiotic-resistant (Mar) mutants. Antimicrob. Agents. Chemother. 38, 542–546 (1994).
Okusu, H., Ma, D. & Nikaido, H. AcrAB efflux pump plays a major role in the antibiotic resistance phenotype of Escherichia coli multiple-antibiotic-resistance (Mar) mutants. J. Bacteriol. 178, 306–308 (1996).
White, D.G., Goldman, J.D., Demple, B. & Levy, S.B. Role of the acrAB locus in organic solvent tolerance mediated by expression of marA, soxS, or robA in Escherichia coli. J. Bacteriol. 179, 6122–6126 (1997).
Cohen, S.P., McMurry, L.M. & Levy, S.B. marA locus causes decreased expression of OmpF porin in multiple-antibiotic-resistant (Mar) mutants of Escherichia coli. J. Bacteriol. 170, 5416–5422 (1988).
Cohen, S.P., Hachler, H. & Levy, S.B. Genetic and functional analysis of the multiple antibiotic resistance (mar) locus in Escherichia coli. J. Bacteriol. 175, 1484–1492 (1993).
Rosner, J.L., Chai, T.J. & Foulds, J. Regulation of ompF porin expression by salicylate in Escherichia coli. J. Bacteriol. 173, 5631–5638 (1991).
Viveiros, M. et al. Antibiotic stress, genetic response and altered permeability of E. coli. PLoS One 2, e365 (2007).
Vecchione, J.J., Alexander, B., Jr. & Sello, J.K. Two distinct major facilitator superfamily drug efflux pumps mediate chloramphenicol resistance in Streptomyces coelicolor. Antimicrob. Agents. Chemother. 53, 4673–4677 (2009).
Mizuno, T., Kato, M., Jo, Y.L. & Mizushima, S. Interaction of OmpR, a positive regulator, with the osmoregulated ompC and ompF genes of Escherichia coli. Studies with wild-type and mutant OmpR proteins. J. Biol. Chem. 263, 1008–1012 (1988).
Han, X. et al. Escherichia coli genes that reduce the lethal effects of stress. BMC Microbiol. 10, 35 (2010).
Itou, J., Eguchi, Y. & Utsumi, R. Molecular mechanism of transcriptional cascade initiated by the EvgS/EvgA system in Escherichia coli K-12. Biosci. Biotechnol. Biochem. 73, 870–878 (2009).
Kato, A. et al. Transcription of emrKY is regulated by the EvgA-EvgS two-component system in Escherichia coli K-12. Biosci. Biotechnol. Biochem. 64, 1203–1209 (2000).
Nishino, K. & Yamaguchi, A. Overexpression of the response regulator evgA of the two-component signal transduction system modulates multidrug resistance conferred by multidrug resistance transporters. J. Bacteriol. 183, 1455–1458 (2001).
Nishino, K., Senda, Y. & Yamaguchi, A. The AraCfamily regulator GadX enhances multidrug resistance in Escherichia coli by activating expression of mdtEF multidrug efflux genes. J. Infect. Chemother. 14, 23–29 (2008).
Higashi, K. et al. Identification of a spermidine excretion protein complex (MdtJI) in Escherichia coli. J. Bacteriol. 190, 872–878 (2008).
Author information
Authors and Affiliations
Corresponding authors
Additional information
These authors contributed equally to this work
Rights and permissions
About this article
Cite this article
Kim, K.S., Park, Y.J., Jung, HJ. et al. Global transcriptome analysis of the Escherichia coli O157 response to Houttuynia Cordata Thunb. BioChip J 4, 237–246 (2010). https://doi.org/10.1007/s13206-010-4312-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13206-010-4312-8