Abstract
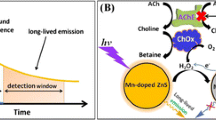
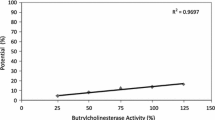
The method to detect the organophosphorus (OP) pesticide was developed using an acetylcholine esterase (AChE) and choline oxidase (ChOx) bienzyme system. AChE, which is inhibited by the OP pesticide, was enzymatically linked with ChOx in solution as well as on electrodes. By employing acetylcholine as the substrate for AChE, we supplied the enzyme product choline to ChOx in order to generate electrocatalyzed current. For electrochemical signaling with AChE/ChOx, ferrocene (Fc)-mediated bioelectrocatalysis through a flavin adenine dinucleotide (FAD) cofactor of ChOx was verified experimentally. An immobilization method for ChOx on a gold electrode was designed to generate amplified and controlled signal by using poly-L-lysine charge interaction and glutaraldehyde cross-linking. Using diazinon-oxon (DZN) as a detection target among the OP pesticides, we successfully conducted an electrochemical detection on the basis of AChE inhibition mediated by the ChOx bioelectrocatalysis.
Similar content being viewed by others
References
Nagatani, N. et al. Rapid and sensitive visual detection of residual pesticides in food using acetylcholinesterase-based disposable membrane chips. Food Control 18, 914–920 (2007).
Zakir Hossain, S.M. et al. Development of a bioactive paper sensor for detection of neurotoxins using piezoelectric inkjet printing of sol-gel-derived bioinks. Anal. Chem. 81, 5474–5483 (2009).
Du, D., Ding, J., Tao, Y. & Chen, X. Application of chemisorption/desorption process of thiocholine for pesticide detection based on acetylcholinesterase biosensor. Sens. Actuators B: Chem. 134, 908–912 (2008).
Jeanty, G. & Marty, J.L. Detection of paraoxon by continuous flow system based enzyme sensor. Biosens. Bioelectron. 13, 213–218 (1998).
Arduini, F. et al. Electrocatalytic oxidation of thiocholine at chemically modified cobalt hexacyanoferrate screen-printed electrodes. J. Electroanal. Chem. 626, 66–74 (2009).
Liu, G., Riechers, S.L., Mellen, M.C. & Lin, Y. Sensitive electrochemical detection of enzymatically generated thiocholine at carbon nanotube modified glassy carbon electrode. Electrochem. Commun. 7, 1163–1169 (2005).
He, P., Davies, J., Greenway, G. & Haswell, S.J. Measurement of acetylcholinesterase inhibition using bienzymes immobilized monolith micro-reactor with integrated electrochemical detection. Anal. Chim. Acta 659, 9–14 (2010).
Riklin, A. & Willner, I. Glucose and acetylcholine sensing multilayer enzyme electrodes of controlled enzyme layer thickness. Anal. Chem. 67, 4118–4126 (1995).
Willner, I., Katz, E. & Willner, B. Electrical contact of redox enzyme layers associated with electrodes: routes to amperometric biosensors. Electroanalysis 9, 965–977 (1997).
Florou, A.B. et al. Electrocatalytic oxidation of NADH in flow analysis by graphite electrode modified with 2,6-dichlorophenolindophenol salts. Electroanalysis 10, 1261–1268 (1998).
Pandey, P.C., Upadhyay, S., Pathak, H.C., Pandey, C.M.D. & Tiwari, I. Acetylthiocholine/acetylcholine and thiocholine/choline electrochemical biosensors/sensors based on an organically modified sol-gel glass enzyme reactor and graphite paste electrode. Sens. Actuators B: Chem. 62, 109–116 (2000).
Xin, Q. & Wightman, R.M. Enzyme modified amperometric sensors for choline and acetylcholine with tetrathiafulvalene tetracyanoquinodimethane as the electron-transfer mediator. Anal. Chim. Acta 341, 43–51 (1997).
Pandey, P.C., Aston, R.W. & Weetall, H.H. Tetracyanoquinodimethane mediated glucose sensor based on a self-assembling alkanethiol/phospholipid bilayer. Biosens. Bioelectron. 10, 669–674 (1995).
Mukherjee, J. & Kirchhoff, J.R. Electrocatalytic microelectrode detectors for choline and acetylcholine following separation by capillary electrophoresis. Anal. Chem. 81, 6996–7002 (2009).
Mao, L. & Yamamoto, K. Glucose and choline on-line biosensors based on electropolymerized Meldola’s blue. Talanta 51, 187–195 (2000).
Upadhyay, S. et al. Immobilization of acetylcholinesterase-choline oxidase on a gold-platinum bimetallic nanoparticles modified glassy carbon electrode for the sensitive detection of organophosphate pesticides, carbamates and nerve agents. Biosens. Bioelectron. 25, 832–838 (2009).
Alfonta, L., Katz, E. & Willner, I. Sensing of acetylcholine by a tricomponent-enzyme layered electrode using faradaic impedance spectroscopy, cyclic voltammetry, and microgravimetric quartz crystal microbalance transduction methods. Anal. Chem. 72, 927–935 (2000).
Chen, Q., Kobayashi, Y., Takesita, H., Hoshi, T. & Anzai, J. Avidin-biotin system-based enzyme multilayer membranes for biosensor application: optimization of loading of choline esterase and choline oxidase in the bienzyme membrane for acetylcholine biosensors. Electroanalysis 10, 94–97 (1998).
Lin, Y., Lu, F. & Wang, J. Disposable carbon nanotube modified screen-printed biosensor for amperometric detection of organophosphorus pesticides and nerve agents. Electroanalysis 16, 145–149 (2004).
Fan, F. & Gadda, G. On the catalytic mechanism of choline oxidase. J. Am. Chem. Soc. 127, 2067–2074 (2005).
Gadda, G., Fan, F. & Hoang, J.V. On the contribution of the positively charged headgroup of choline to substrate binding and catalysis in the reaction catalyzed by choline oxidase. Arch. Biochem. Biophys. 451, 182–187 (2006).
Yoon, H.C. & Kim, H.-S. Multilayered assembly of dendrimers with enzymes on gold: thickness-controlled biosensing Interface. Anal. Chem. 72, 922–926 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, J.H., Han, Y.D., Song, S.Y. et al. Biosensor for organophosphorus pesticides based on the acetylcholine esterase inhibition mediated by choline oxidase bioelectrocatalysis. BioChip J 4, 223–229 (2010). https://doi.org/10.1007/s13206-010-4310-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13206-010-4310-x