Abstract
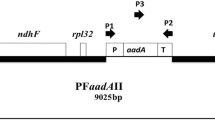
A protocol has been established for genetic transformation of the chloroplasts in two new cultivars of tomato (Solanum lycopersicum L.) grown in India and Australia: Pusa Ruby and Yellow Currant. Tomato cv. Green Pineapple was also used as a control that has previously been used for establishing chloroplast transformation by other researchers. Selected tomato cultivars were finalized from ten other tested cultivars (Green Pineapple excluded) due to their high regeneration potential and better response to chloroplast transformation. This protocol was set up using a chloroplast transformation vector (pRB94) for tomatoes that is made up of a synthetic gene operon. The vector has a chimeric aadA selectable marker gene that is controlled by the rRNA operon promoter (Prrn). This makes the plant or chloroplasts resistant to spectinomycin and streptomycin. After plasmid-coated particle bombardment, leaf explants were cultured in 50 mg/L selection media. Positive explant selection from among all the dead-appearing (yellow to brown) explants was found to be the major hurdle in the study. Even though this study was able to find plastid transformants in heteroplasmic conditions, it also found important parameters and changes that could speed up the process of chloroplast transformation in tomatoes, resulting in homoplasmic plastid-transformed plants.




Similar content being viewed by others
Data availability
The entire dataset is available as supplemental material to this publication.
Abbreviations
- Prrn:
-
rRNA Pomoter
- IAA:
-
Indole 3-acetic Acid
- BAP:
-
N6-benzylamino Purine
- LCY:
-
Lycopene Cyclase
- BHY:
-
β-Carotene Hydroxylase
- BKT:
-
β-Carotene Ketolase
- PRM:
-
Plant Regeneration Media
- CTAB:
-
Cetyltrimethylammonium Bromide
- GP:
-
Green Pineapple
- YC:
-
Yellow Currant
- PR:
-
Pusa Ruby
References
Avila EM, Day A (2014) Stable plastid transformation of Petunia. In: Maliga P (ed) Chloroplast biotechnology. Springer, Berlin, pp 277–293
Bouvier F, Camara B (2007) The role of plastids in ripening fruits. In: Wise RR, Hoober JK (eds) The structure and function of plastids. Springer, Dordrecht, pp 419–432
Cheng L, Li H-P, Qu B, Huang T, Tu J-X, Fu T-D, Liao Y-C (2010) Chloroplast transformation of rapeseed (Brassica napus) by particle bombardment of cotyledons. Plant Cell Rep 29(4):371–381
Dubald M, Tissot G, Pelissier B (2014) Plastid transformation in soybean. In: Maliga P (ed) Chloroplast biotechnology. Springer, Berlin, pp 345–354
Farooq N, Nawaz MA, Mukhtar Z, Ali I, Hundleby P, Ahmad N (2019) Investigating the in vitro regeneration potential of commercial cultivars of Brassica. Plants (Basel, Switzerland) 8(12):558. https://doi.org/10.3390/plants8120558
Harada H, Maoka T, Osawa A, Hattan J-I, Kanamoto H, Shindo K, Otomatsu T, Misawa N (2014) Construction of transplastomic lettuce (Lactuca sativa) dominantly producing astaxanthin fatty acid esters and detailed chemical analysis of generated carotenoids. Transgenic Res 23(2):303–315
Joyard J, Teyssier E, Miege C, Berny-Seigneurin D, Maréchal E, Block MA, Dorne A-J, Rolland N, Ajlani G, Douce R (1998) The biochemical machinery of plastid envelope membranes. Plant Physiol 118(3):715–723
Kumar S, Dhingra A, Daniell H (2004a) Plastid-expressed betaine aldehyde dehydrogenase gene in carrot cultured cells, roots, and leaves confers enhanced salt tolerance. Plant Physiol 136(1):2843–2854
Kumar S, Dhingra A, Daniell H (2004b) Stable transformation of the cotton plastid genome and maternal inheritance of transgenes. Plant Mol Biol 56(2):203–216. https://doi.org/10.1007/s11103-004-2907-y
Lu Y, Rijzaani H, Karcher D, Ruf S, Bock R (2013) Efficient metabolic pathway engineering in transgenic tobacco and tomato plastids with synthetic multigene operons. Proc Natl Acad Sci 110(8):E623–E632
Maliga P, Tungsuchat-Huang T (2014) Plastid transformation in Nicotiana tabacum and Nicotiana sylvestris by biolistic DNA delivery to leaves. In: Maliga P (ed) Chloroplast biotechnology: methods and protocols. Humana Press, New Jersey, pp 147–163
McBride KE, Svab Z, Schaaf DJ, Hogan PS, Stalker DM, Maliga P (1995) Amplification of a chimeric bacillus gene in chloroplasts leads to an extraordinary level of an insecticidal protein in tobacco. Bio/technology 13(4):362–365
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15(3):473–497
Okumura S, Sawada M, Park YW, Hayashi T, Shimamura M, Takase H, Tomizawa K-I (2006) Transformation of poplar (Populus alba) plastids and expression of foreign proteins in tree chloroplasts. Transgenic Res 15(5):637–646
Ruf S, Bock R (2014) Plastid transformation in tomato. In: Maliga P (ed) Chloroplast biotechnology. Springer, Berlin, pp 265–276
Ruf S, Bock R (2021) Plastid transformation in tomato: a vegetable crop and model species. Methods Mol Biol 2317:217–228. https://doi.org/10.1007/978-1-0716-1472-3_11
Ruf S, Hermann M, Berger IJ, Carrer H, Bock R (2001) Stable genetic transformation of tomato plastids and expression of a foreign protein in fruit. Nat Biotechnol 19(9):870
Ruf S, Kroop X, Bock R (2021) Chloroplast transformation in Arabidopsis. Curr Protoc 1(4):e103. https://doi.org/10.1002/cpz1.103
Sikdar S, Serino G, Chaudhuri S, Maliga P (1998) Plastid transformation in Arabidopsis thaliana. Plant Cell Rep 18(1–2):20–24
Singh A, Verma S, Bansal K (2010) Plastid transformation in eggplant (Solanum melongena L.). Transgenic Res 19(1):113–119
Svab Z, Maliga P (1993) High-frequency plastid transformation in tobacco by selection for a chimeric aadA gene. Proc Natl Acad Sci U S A 90(3):913–917. https://doi.org/10.1073/pnas.90.3.913
Tanwar N, Rookes JE, Cahill DM, Lenka SK (2023a) Carotenoid pathway engineering in tobacco chloroplast using a synthetic operon. Mol Biotechnol 65:1923–1934
Tanwar N, Arya SS, Rookes JE, Cahill DM, Lenka SK, Bansal KC (2023b) Prospects of chloroplast metabolic engineering for developing nutrient-dense food crops. Crit Rev Biotechnol 43(7):1001–1018
Tseng M-J, Yang M-T, Chu W-R, Liu C-W (2014) Plastid transformation in cabbage (Brassica oleracea L. var. capitata L.) by the biolistic process. Methods Mol Biol 1132:355–366
Verma D, Samson NP, Koya V, Daniell H (2008) A protocol for expression of foreign genes in chloroplasts. Nat Protoc 3(4):739–758. https://doi.org/10.1038/nprot.2007.522
Viitanen PV, Devine AL, Khan MS, Deuel DL, Van Dyk DE, Daniell H (2004) Metabolic engineering of the chloroplast genome using the Escherichia coli ubi C gene reveals that chorismate is a readily abundant plant precursor for p-hydroxybenzoic acid biosynthesis. Plant Physiol 136(4):4048–4060
Wang Y, Wei Z, Xing S (2018) Stable plastid transformation of rice, a monocot cereal crop. Biochem Biophys Res Commun 503(4):2376–2379
Wurbs D, Ruf S, Bock R (2007) Contained metabolic engineering in tomatoes by expression of carotenoid biosynthesis genes from the plastid genome. Plant J 49(2):276–288
Zhou F, Badillo-Corona JA, Karcher D, Gonzalez-Rabade N, Piepenburg K, Borchers AMI, Maloney AP, Kavanagh TA, Gray JC, Bock R (2008) High-level expression of human immunodeficiency virus antigens from the tobacco and tomato plastid genomes. Plant Biotechnol J 6(9):897–913
Acknowledgements
We thank Prof. Dr. Ralph Bock (Max Planck Institute for molecular plant physiology) for providing pRB94 chloroplast specific vector and Deakin University Australia for partially supporting this research and providing postgraduate research scholarship to NT.
Funding
Deakin University Australia is acknowledged for partially supporting this research and providing postgraduate research scholarship to the first author.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tanwar, N., Mahto, B.K., Rookes, J.E. et al. Chloroplast transformation in new cultivars of tomato through particle bombardment. 3 Biotech 14, 120 (2024). https://doi.org/10.1007/s13205-024-03954-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-024-03954-3