Abstract
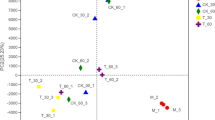
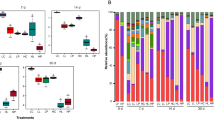
Alfalfa (Medicago sativa L.) is a vital source of forage protein for ruminants, yet its ensiling poses challenges due to high buffering capacity and low water-soluble carbohydrates (WSC). This study investigated the impact of sodium diacetate (SDA) on alfalfa silage quality and aerobic stability. SDA was applied at four different rates to wilted alfalfa on a fresh basis: 0 g/kg, 3 g/kg, 5 g/kg, and 7 g/kg, and silages were ensiled in laboratory-scale silos for 45 days, followed by 7 days of aerobic exposure. A 16S rRNA gene sequencing assay using GenomeLab™ GeXP was performed to determine the relationship between dominant isolated lactic acid bacteria species and fermentation characteristics and aerobic stability on silage. The results showed that Lentilolactobacillus brevis, Pediococcus pentosaceus and Enterococcus faecium were the most prevalent bacteria when silos were opened, whereas Weissella paramesenteroides, Bacillus cereus, B. megaterium and Bacillus spp. were most prevalent bacteria after 7 days of aerobic exposure. Dry matter, pH, and WSC content were not affected by SDA, but doses above 5 g/kg induced a homofermentative process, which increased lactic acid concentration and lactic acid to acetic acid ratio, decreased yeast count during aerobic exposure, and improved aerobic stability. These findings offer useful information for optimizing SDA usage in silage, assuring improved quality and longer storage, and thereby improving animal husbandry and sustainable feed practices.





Similar content being viewed by others
Data availability
Not applicable.
References
Anonymous (1986) The analysis of agricultural material; Reference Book. HMSO, London, pp 427–428
AOAC (1990) Official methods of analysis. In: 15th ed. Association of Official Analytical Chemist, Washington DC
Ashbell G, Weinberg ZG, Azrieli A, Hen Y, Horev B (1991) A simple system to study the aerobic determination of silages. Can Agric Eng 34:171–175
Auerbach H, Nadeau E (2020) Effects of additive type on fermentation and aerobic stability and its interaction with air exposure on silage nutritive value. Agronomy 10:1229. https://doi.org/10.3390/agronomy10091229
Bağcık C, Koç F, Erten K, Esen S, Palangi V, Lackner M (2022) Lentilactobacillus buchneri preactivation affects the mitigation of methane emission in corn silage treated with or without urea. Fermentation 8:747. https://doi.org/10.3390/fermentation8120747
Bai J, Xu D, Xie D, Wang M, Li Z, Guo X (2020) Effects of antibacterial peptide-producing Bacillus subtilis and Lactobacillus buchneri on fermentation, aerobic stability, and microbial community of alfalfa silage. Bioresour Technol 315:123881. https://doi.org/10.1016/j.biortech.2020.123881
Beasley SS, Saris PEJ (2004) Nisin-producing Lactococcus lactis strains from human milk. Appl Environ Microbiol 70:5051–5053. https://doi.org/10.1128/AEM.70.8.5051-5053.2004
Bernardi A, Härter CJ, Rabelo CHS, Silva AWL, Reis RA (2019) A meta-analysis examining lactic acid bacteria inoculants for maize silage: effects on fermentation, aerobic stability, nutritive value and livestock production. Grass Forage Sci 74:596–612. https://doi.org/10.1111/gfs.12452
Blajman JE, Vinderola G, Paez RB, Signorini ML (2020) The role of homofermentative and heterofermentative lactic acid bacteria for alfalfa silage: a meta-analysis. J Agric Sci 158:107–118. https://doi.org/10.1017/S0021859620000386
Borreani G, Tabacco E, Schmidt RJ, Holmes BJ, Muck RE (2018) Silage review: factors affecting dry matter and quality losses in silages. J Dairy Sci 101:3952–3979. https://doi.org/10.3168/jds.2017-13837
Chen L, Li J, Dong Z, Shao T (2019) Effects of lactic acid bacteria inoculants and fibrolytic enzymes on the fermentation quality, in vitro degradability, ruminal variables and microbial communities of high-moisture alfalfa silage. Grassl Sci 65:216–225. https://doi.org/10.1111/grs.12240
Dai T, Dong D, Wang S, Zong C, Yin X, Jia Y, Shao T (2022) The effectiveness of chemical additives on fermentation profiles, aerobic stability and in vitro ruminal digestibility of total mixed ration ensiled with Napier grass and wet distillers’ grains in southeast China. Ital J Anim Sci 21:979–989. https://doi.org/10.1080/1828051X.2022.2078234
Driehuis F, Wilkinson JM, Jiang Y, Ogunade I, Adesogan AT (2018) Silage review: animal and human health risks from silage. J Dairy Sci 101:4093–4110. https://doi.org/10.3168/jds.2017-13836
Duniere L, Xu S, Long J, Elekwachi C, Wang Y, Turkington K, Forster R, McAllister TA (2017) Bacterial and fungal core microbiomes associated with small grain silages during ensiling and aerobic spoilage. BMC Microbiol 17:1–16. https://doi.org/10.1186/s12866-017-0947-0
Esen S, Okuyucu B, Koç F, Özdüven ML (2022a) Determination of nutritional quality and aerobic stability of sorghum, maize, and sorghum-maize mixture silages. J Tekirdag Agric Fac 19:61–69. https://doi.org/10.33462/jotaf.889657
Esen S, Cabi E, Koç F (2022b) Effect of freeze-dried kefir culture inoculation on nutritional quality, in vitro digestibility, mineral concentrations, and fatty acid composition of white clover silages. Biomass Convers Biorefin. https://doi.org/10.1007/s13399-022-03422-z
Guo G, Shen C, Liu Q, Zhang SL, Wang C, Chen L, Xu QF, Wang YX, Huo WJ (2019) Fermentation quality and in vitro digestibility of first and second cut alfalfa (Medicago sativa L.) silages harvested at three stages of maturity. Anim Feed Sci Technol 257:1–9. https://doi.org/10.1016/j.anifeedsci.2019.114274
He L, Wang C, Xing Y, Zhou W, Pian R, Chen X, Zhang Q (2020) Ensiling characteristics, proteolysis and bacterial community of high-moisture corn stalk and stylo silage prepared with Bauhinia variegate flower. Bioresour Technol 296:122336. https://doi.org/10.1016/j.biortech.2019.122336
He Q, Zhou W, Chen X, Zhang Q (2021) Chemical and bacterial composition of Broussonetia papyrifera leaves ensiled at two ensiling densities with or without Lactobacillus plantarum. J Clean Prod 329:129792. https://doi.org/10.1016/j.jclepro.2021.129792
Herrmann C, Heiermann M, Idler C (2011) Effects of ensiling, silage additives and storage period on methane formation of biogas crops. Bioresour Technol 102:5153–5161. https://doi.org/10.1016/j.biortech.2011.01.012
Jiang D, Li B, Zheng M, Niu D, Zuo S, Xu C (2020) Effects of Pediococcus pentosaceus on fermentation, aerobic stability and microbial communities during ensiling and aerobic spoilage of total mixed ration silage containing alfalfa (Medicago sativa L.). Grassl Sci 66:215–224. https://doi.org/10.1111/grs.12272
Koc F, Coskuntuna L (2003) The comparison of the two different methods on the determination of organic acids in silage fodders. J Anim Prod 44:37–47
Koç F, Ağma Okur A, Okur E (2020) Sodyum diesatat ve sodyum benzoat ilavesinin yüksek nemli mısır silajlarının aerobik stabilite özellikleri üzerine etkileri. Ege Üniv Ziraat Fak Derg 57:289–301. https://doi.org/10.20289/zfdergi.611010
Kung L Jr, Shaver RD, Grant RJ, Schmidt RJ (2018) Silage review: interpretation of chemical, microbial, and organoleptic components of silages. J Dairy Sci 101:4020–4033. https://doi.org/10.3168/jds.2017-13909
Li P, Ji S, Hou C, Tang H, Wang Q, Shen Y (2016) Effects of chemical additives on the fermentation quality and N distribution of alfalfa silage in south of China. Anim Sci J 87:1472–1479. https://doi.org/10.1111/asj.12600
Li D, Wang Y, Zhang Y, Lin Y, Yang F (2018a) Evaluation of lactic acid bacteria isolated from alfalfa for silage fermentation. Grassl Sci 64:190–198. https://doi.org/10.1111/grs.12198
Li X, Tian J, Zhang Q, Jiang Y, Wu Z, Yu Z (2018b) Effects of mixing red clover with alfalfa at different ratios on dynamics of proteolysis and protease activities during ensiling. J Dairy Sci 101:8954–8964. https://doi.org/10.3168/jds.2018-14763
Li P, Zhang Y, Gou W, Cheng Q, Bai S, Cai Y (2019) Silage fermentation and bacterial community of bur clover, annual ryegrass and their mixtures prepared with microbial inoculant and chemical additive. Anim Feed Sci Technol 247:285–293. https://doi.org/10.1016/j.anifeedsci.2018.11.009
Li R, Jiang D, Zheng M, Tian P, Zheng M, Xu C (2020) Microbial community dynamics during alfalfa silage with or without clostridial fermentation. Sci Rep 10:17782. https://doi.org/10.1038/s41598-020-74958-1
Liu B, Huan H, Gu H, Xu N, Shen Q, Ding C (2019) Dynamics of a microbial community during ensiling and upon aerobic exposure in lactic acid bacteria inoculation-treated and untreated barley silages. Bioresour Technol 273:212–219. https://doi.org/10.1016/j.biortech.2018.10.041
Liu G, Nie R, Liu Y, Mehmood A (2022) Combined antimicrobial effect of bacteriocins with other hurdles of physicochemic and microbiome to prolong shelf life of food: a review. Sci Total Environ 825:154058. https://doi.org/10.1016/j.scitotenv.2022.154058
Lv H, Pian R, Xing Y, Zhou W, Yang F, Chen X, Zhang Q (2020) Effects of citric acid on fermentation characteristics and bacterial diversity of Amomum villosum silage. Bioresour Technol 307:123290. https://doi.org/10.1016/j.biortech.2020.123290
McDonald P, Henderson N, Heron S (1991) The biochemistry of silage, 2nd edn. Chalcombe Publications, Marlow
Minitab (2014) Minitab I: statistical software for windows, release 17. Minitab Incorporation, USA
Mu L, Wang Q, Cao X, Zhang Z (2022) Effects of fatty acid salts on fermentation characteristics, bacterial diversity and aerobic stability of mixed silage prepared with alfalfa, rice straw and wheat bran. J Sci Food Agric 102:1475–1487. https://doi.org/10.1002/jsfa.11482
Mugabe W, Yuan X, Li J, Dong Z, Shao T (2019) Effects of hexanoic acid, Lactobacillus plantarum and their combination on the fermentation characteristics of Napier grass. Anim Feed Sci Technol 253:135–140. https://doi.org/10.1016/j.anifeedsci.2019.05.005
Ni K, Wang X, Lu Y, Guo L, Li X, Yang F (2020) Exploring the silage quality of alfalfa ensiled with the residues of astragalus and hawthorn. Bioresour Technol 297:122249. https://doi.org/10.1016/j.biortech.2019.122249
Oladosu Y, Rafii MY, Abdullah N, Magaji U, Hussin G, Ramli A, Miah G (2016) Fermentation quality and qdditives: a case of rice straw silage. Biomed Res Int. https://doi.org/10.1155/2016/7985167
Olaimat AN, Al-Holy MA, Shahbaz HM, Al-Nabulsi AA, Abu Ghoush MH, Osaili TM, Ayyash MM, Holley RA (2018) Emergence of antibiotic resistance in Listeria monocytogenes isolated from food products: a comprehensive review. Compr Rev Food Sci Food Saf 17:1277–1292. https://doi.org/10.1111/1541-4337.12387
Playne MJ, Mc Donald P (1966) The buffering constituent of herbage and of silage. J Sci Food Agric 17:264–268. https://doi.org/10.1002/jsfa.2740170609
Queiroz OCM, Ogunade IM, Weinberg Z, Adesogan AT (2018) Silage review: foodborne pathogens in silage and their mitigation by silage additives. J Dairy Sci 101:4132–4142. https://doi.org/10.3168/jds.2017-13901
Ren H, Sun W, Yan Z, Zhang Y, Wang Z, Song B, Zheng Y, Li J (2021) Bioaugmentation of sweet sorghum ensiling with rumen fluid: Fermentation characteristics, chemical composition, microbial community, and enzymatic digestibility of silages. J Clean Prod 294:126308. https://doi.org/10.1016/j.jclepro.2021.126308
Santos WP, Carvalho BF, Ávila CLS, Júnior GSD, Pereira MN, Rosane Freitas Schwan RF (2015) Glycerin as an additive for sugarcane silage. Ann Microbiol 65:1547–1556. https://doi.org/10.1007/s13213-014-0993-x
Sarteshnizi FR, Seifdavati J, Abdi-benemar H, Salem AZM, Sharifi RS, Mlambo V (2018) The potential of rumen fluid waste from slaughterhouses as an environmentally friendly source of enzyme additives for ruminant feedstuffs. J Clean Prod 195:1026–1031. https://doi.org/10.1016/j.jclepro.2018.05.268
Seale DR, Pahlow G, Spoelstra SF, Lindgren S, Dellaglio F, Lowe JF (1986) Methods for the microbiological analysis of silage. In: Proceedings of the Eurobac Conference, Uppsala, Sweden, August 12–16, pp 147–164
Soundharrajan I, Park HS, Rengasamy S, Sivanesan R, Choi KC (2021) Application and future prospective of lactic acid bacteria as natural additives for silage production—a review. Appl Sci 11:8127. https://doi.org/10.3390/app11178127
Stanaćev V, Vik D (2002) The feed sugar minimum as a precondition of good quality silage. Acta Agric Serb 7:41–48
Tabacco E, Righi F, Quarantelli A, Borreani G (2011) Dry matter and nutritional losses during aerobic deterioration of corn and sorghum silages as influenced by different lactic acid bacteria inocula. J Dairy Sci 94:1409–1419. https://doi.org/10.3168/jds.2010-3538
Tamura K, Stecher G, Kumar S (2021) MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 38:3022–3027. https://doi.org/10.1093/molbev/msab120
Ulger I, Buyukkılıc Beyzi S, Kaliber M, Konca Y (2020) Chemical, nutritive, fermentation profile and gas production of citrus pulp silages, alone or combined with maize silage. South Afr J Anim Sci 50:161–169. https://doi.org/10.4314/sajas.v50i1.17
Wang M, Franco M, Cai Y, Yu Z (2020a) Dynamics of fermentation profile and bacterial community of silage prepared with alfalfa, whole-plant corn and their mixture. Anim Feed Sci Technol 270:114702. https://doi.org/10.1016/j.anifeedsci.2020.114702
Wang S, Zhao J, Dong Z, Li J, Kaka NA, Shao T (2020b) Sequencing and microbiota transplantation to determine the role of microbiota on the fermentation type of oat silage. Bioresour Technol 309:123371. https://doi.org/10.1016/j.biortech.2020.123371
Wang M, Chen M, Bai J, Zhang J, Su R, Franco M, Ding Z, Zhang X, Zhang Y, Guo X (2022a) Ensiling characteristics, in vitro rumen fermentation profile, methane emission and archaeal and protozoal community of silage prepared with alfalfa, sainfoin and their mixture. Anim Feed Sci Technol 284:115154. https://doi.org/10.1016/j.anifeedsci.2021.115154
Wang S, Li J, Zhao J, Dong Z, Shao T (2022b) Exploring the ensiling characteristics and bacterial community of red clover inoculated with the epiphytic bacteria from temperate gramineous grasses. J Appl Microbiol 132:177–188. https://doi.org/10.1111/jam.15234
Weinberg ZG (2008) Preservation of forage crops by solid-state lactic acid fermentation-ensiling. Current developments in solid-state fermentation, 1st edn. Springer, New York, pp 443–467
Wen AY, Yuan XJ, Wang J, Desta ST, Shao T (2017) Effects of four short-chain fatty acids or salts on dynamics of fermentation and microbial characteristics of alfalfa silage. Anim Feed Sci Technol 223:141–148. https://doi.org/10.1016/j.anifeedsci.2016.11.017
Williams SD, Shinners KJ (2012) Farm-scale anaerobic storage and aerobic stability of high dry matter sorghum as a biomass feedstock. Biomass Bioenergy 46:309–316. https://doi.org/10.1016/j.biombioe.2012.08.010
Yuan X, Wen A, Desta ST, Wang J, Shao T (2017) Effects of sodium diacetate on the fermentation profile, chemical composition and aerobic stability of alfalfa silage. Asian- Australas. J Anim Sci 30:804–810. https://doi.org/10.5713/ajas.16.0773
Zhao J, Dong Z, Li J, Chen L, Bai Y, Jia Y, Shao T (2018) Ensiling as pretreatment of rice straw: the effect of hemicellulase and Lactobacillus plantarum on hemicellulose degradation and cellulose conversion. Bioresour Technol 266:158–165. https://doi.org/10.1016/j.biortech.2018.06.058
Zhao J, Tao X, Wang S, Li J, Shao T (2021) Effect of sorbic acid and dual-purpose inoculants on the fermentation quality and aerobic stability of high dry matter rice straw silage. J Appl Microbiol 30:1456–1465. https://doi.org/10.1111/jam.14882
Zheng M, Niu D, Zuo S, Mao P, Meng L, Xu C (2018) The effect of cultivar, wilting and storage period on fermentation and the clostridial community of alfalfa silage. Ital J Anim Sci 17:336–346. https://doi.org/10.1080/1828051X.2017.1364984
Zhou W, Pian R, Yang F, Chen X, Zhang Q (2021) The sustainable mitigation of ruminal methane and carbon dioxide emissions by co-ensiling corn stalk with Neolamarckia cadamba leaves for cleaner livestock production. J Clean Prod 311:127680. https://doi.org/10.1016/j.jclepro.2021.127680
Funding
No funding was received to assist with the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Conceptualization, SE and FK; Methodology, SE, FK and RI; Software, SE and RI; Validation, FK and RI; Formal analysis, FK and RI; Investigation, SE and FK; Data curation, FK; Writing-original draft, SE; Writing-review and editing, SE, FK and RI; Supervision, FK. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Ethical approval
Not applicable.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Esen, S., Koç, F. & Işık, R. Effect of sodium diacetate on fermentation, aerobic stability, and microbial diversity of alfalfa silage. 3 Biotech 14, 10 (2024). https://doi.org/10.1007/s13205-023-03853-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-023-03853-z